Clear Sky Science · en
ASCT2 palmitoylation regulated by JNK1-ZDHHC14 axis orchestrates glutamine metabolism and NSCLC progression
Fueling the Fire of Lung Cancer
Many fast-growing lung cancers are addicted to glutamine, a common amino acid that serves as a key fuel and building block. This study uncovers how tumor cells fine-tune a major glutamine “gate” on their surface and how disrupting this control system could slow cancer growth. Understanding this hidden fuel valve helps explain why some tumors are so hard to starve and points to new drug combinations that may cut off their energy supply more effectively.
A Cellular Gate for Glutamine
Glutamine enters cells mainly through a transporter protein called ASCT2, which sits in the outer membrane and shuttles glutamine inside. Non-small cell lung cancer (NSCLC), the most common form of lung cancer, often relies heavily on glutamine to support rapid division and survival. High ASCT2 levels in tumors are linked to aggressive disease and poor patient outcomes. Yet ASCT2 itself is constantly being made, modified and destroyed, raising the question: what decides how much of this gatekeeper remains on the cell surface at any given time?
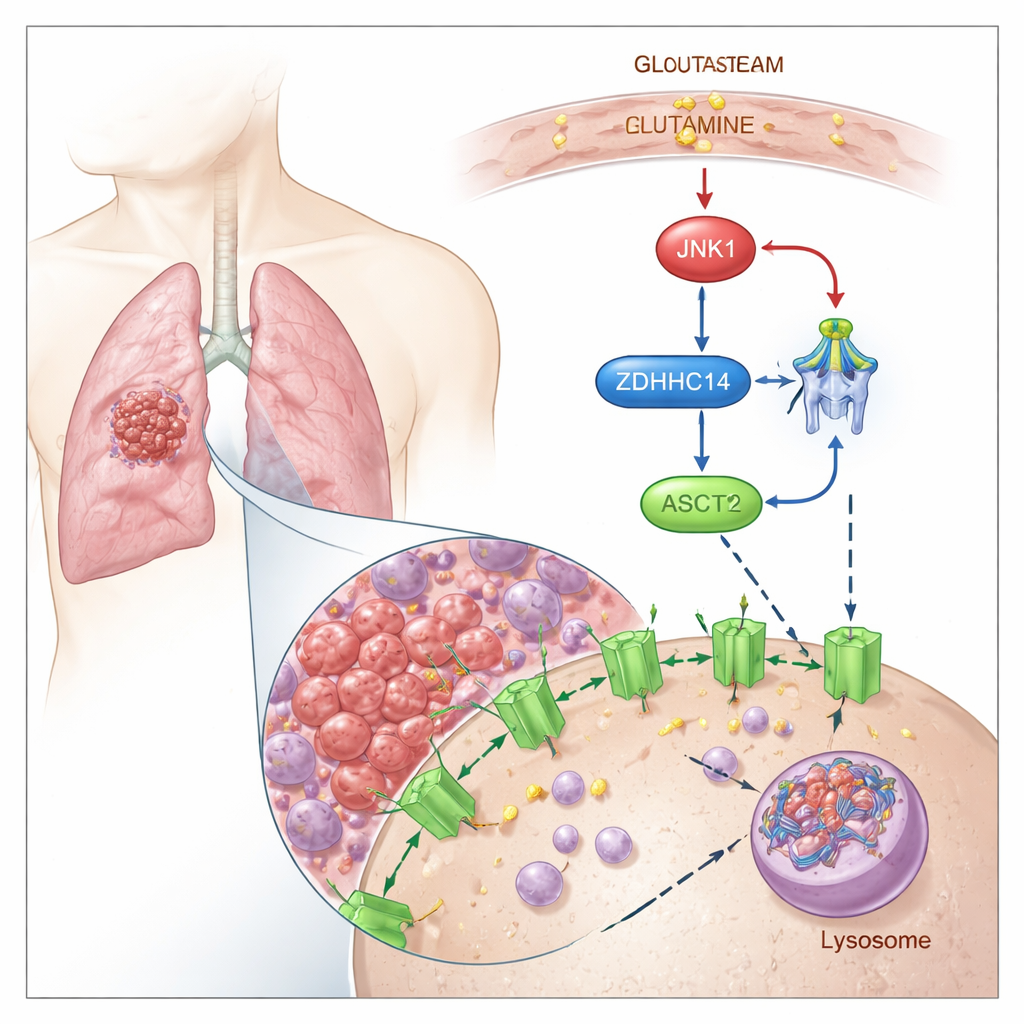
Tagging ASCT2 for Breakdown
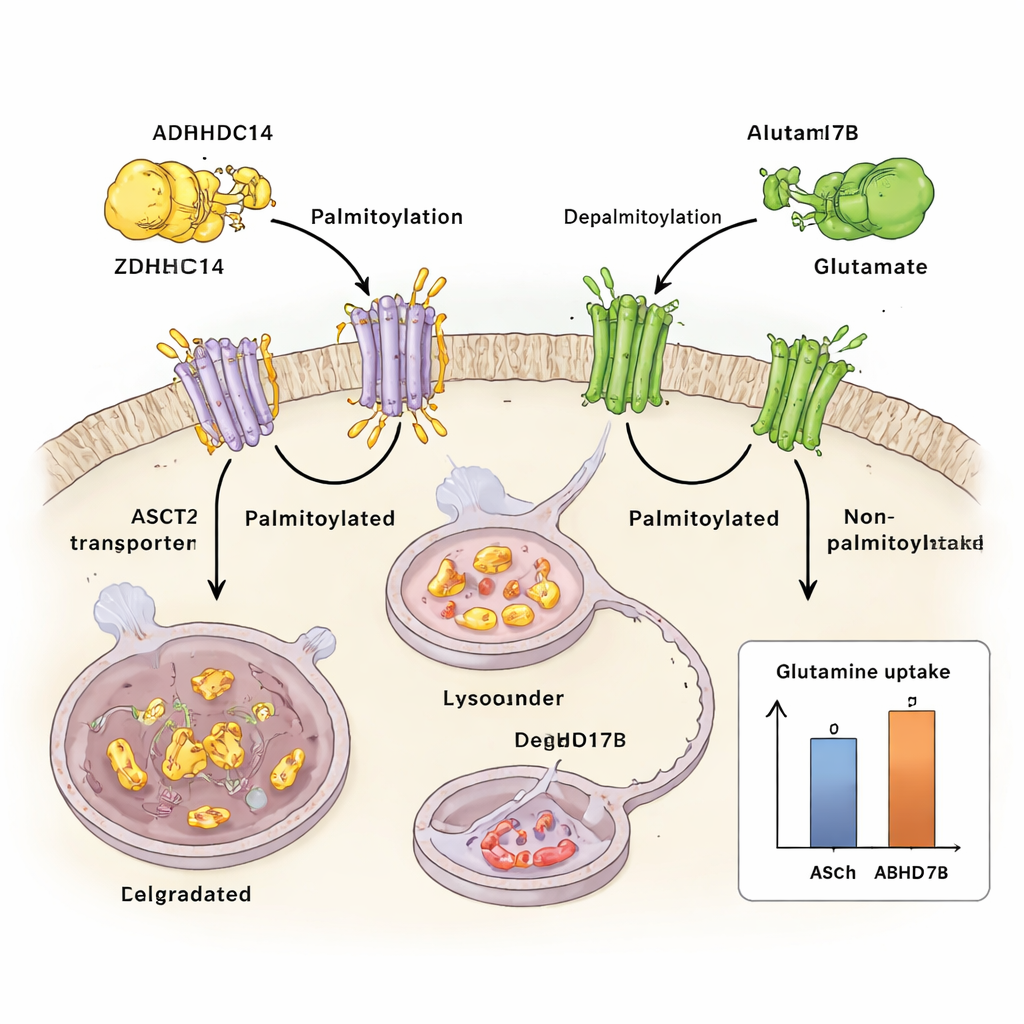
The researchers show that ASCT2 is controlled by a chemical “tag” called palmitoylation, in which a fatty acid is attached to specific cysteine building blocks in the protein. They find that an enzyme named ZDHHC14 adds this lipid tag to two conserved sites on ASCT2 (Cys39 and Cys48). Once tagged, ASCT2 is rerouted from the cell surface into cellular recycling centers called lysosomes, where it is broken down. Blocking palmitoylation keeps ASCT2 from being tagged and degraded, leading to higher ASCT2 protein levels and boosted glutamine uptake, without changing the underlying gene activity.
A Counter-Enzyme That Protects the Gate
Opposing ZDHHC14 is another enzyme, ABHD17B, which removes the fatty tag from ASCT2. When ABHD17B is active, ASCT2 is less palmitoylated, more stable, and more likely to be recycled back to the cell surface instead of sent to the lysosome. This back-and-forth tagging and untagging acts like a finely tuned dial on glutamine flow: ZDHHC14 pushes ASCT2 toward destruction, while ABHD17B rescues it and sustains glutamine transport. In lung cancer cells, shifting this balance toward less palmitoylation increases glutamine uptake and supports tumor growth in dishes and in mice.
How Nutrient Stress Rewires the System
The team then examined what happens when tumor cells run low on glutamine. Under glutamine deprivation, a stress-sensitive signaling protein called JNK1 switches on. JNK1 physically binds to ZDHHC14 and adds a phosphate group at a specific site (Thr440), marking ZDHHC14 for its own lysosomal degradation. As ZDHHC14 levels drop, ASCT2 receives fewer palmitoylation tags, escapes breakdown, and builds up at the cell surface—precisely when the cell most needs to scavenge scarce glutamine. Mutating this single JNK1-sensitive site stabilizes ZDHHC14, restores ASCT2 tagging, and reduces glutamine uptake, underscoring how tightly nutrient stress is wired into this control loop.
Translating Mechanism into Therapy
Because ASCT2 is so important for tumor metabolism, drugs that block it, such as the experimental compound V9302, are being explored as cancer treatments. This study reveals that JNK1 signaling can counteract ASCT2 destruction, effectively protecting the glutamine gate. In cell and mouse models of NSCLC, combining a JNK inhibitor with V9302 more strongly reduced glutamine uptake, tumor cell growth, and tumor size than either drug alone. Patient samples and public datasets further show that low ZDHHC14 and high ASCT2 levels are common in NSCLC and associated with worse survival, suggesting that this molecular pattern could help identify patients most likely to benefit from targeting this pathway.
What This Means for Patients
In everyday terms, this work shows that certain lung cancers control their favorite fuel by adjusting a molecular valve on their surface. A trio of players—JNK1, ZDHHC14, and ABHD17B—decides whether the glutamine gate ASCT2 is tagged for destruction or kept open. When glutamine is scarce, the system shifts to preserve ASCT2 and keep fuel flowing, helping tumors survive in tough conditions. By jointly blocking ASCT2 and the JNK pathway that protects it, researchers were able to starve cancer cells more effectively in laboratory and animal models. While more testing is needed, this phosphorylation–palmitoylation axis offers a promising new angle for therapies aimed at cutting off the nutrient lifelines of glutamine-hungry lung tumors.
Citation: Chen, X., Ke, Z., Wei, S. et al. ASCT2 palmitoylation regulated by JNK1-ZDHHC14 axis orchestrates glutamine metabolism and NSCLC progression. Cell Discov 12, 13 (2026). https://doi.org/10.1038/s41421-026-00870-z
Keywords: glutamine metabolism, non-small cell lung cancer, ASCT2 transporter, protein palmitoylation, JNK signaling