Clear Sky Science · en
The molecular mechanisms and potential therapeutic implications of the crosstalk between DNA methylation and metabolic reprogramming in thyroid cancer
Why this research matters beyond the lab
Thyroid cancer is one of the fastest-rising cancers worldwide. While many cases are treatable, the most aggressive forms often stop responding to radioactive iodine, a mainstay therapy, leaving patients with few good options. This review article explains how two invisible forces inside cells—chemical tags on DNA and changes in how tumor cells use fuel—team up to drive thyroid cancer growth and treatment resistance. Understanding this hidden partnership could open the door to more precise and durable treatments.
How cancer cells rewrite their instruction manual
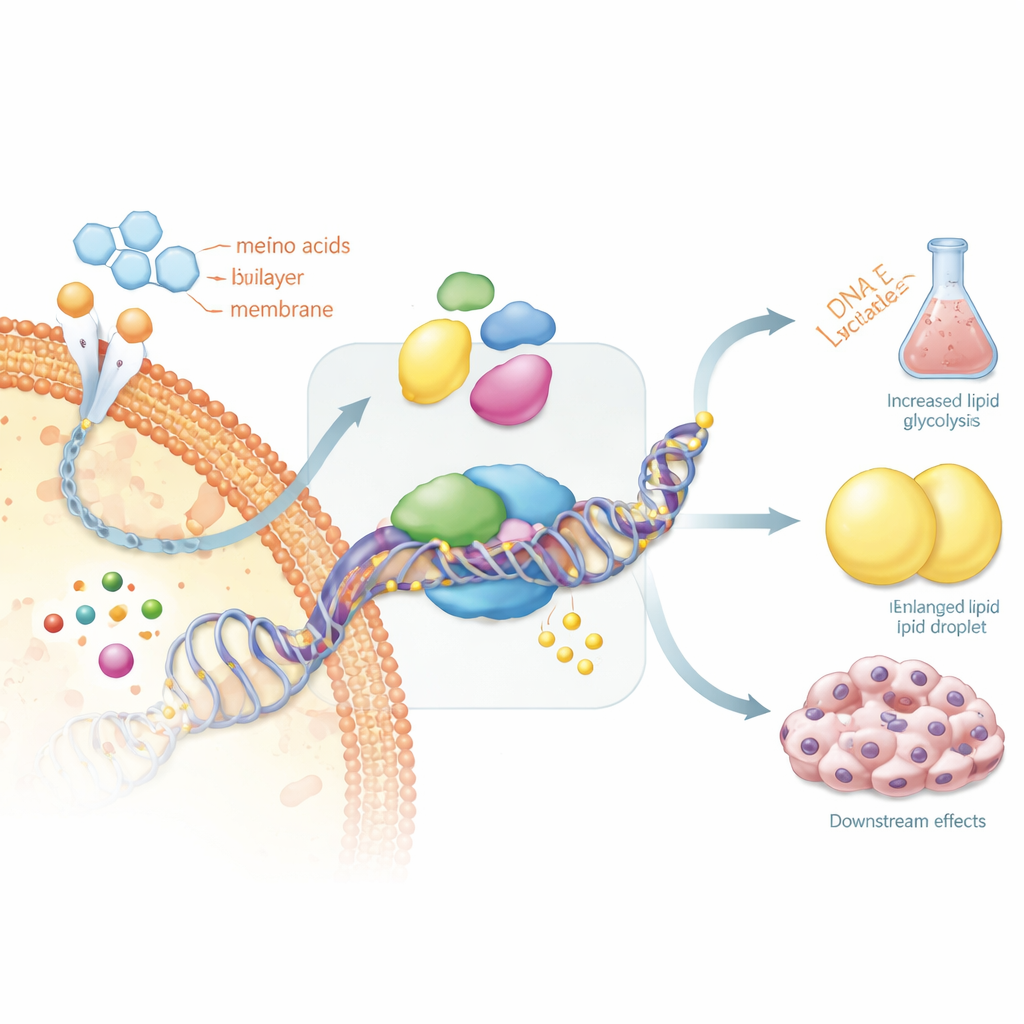
Every cell carries the same DNA, but chemical tags called methyl groups can switch genes on or off without changing the genetic code itself. In thyroid cancer, this DNA “markup” is profoundly distorted. Large stretches of the genome lose methyl tags, which can destabilize chromosomes, while key brakes on growth and thyroid function become over-tagged and silenced. These include tumor suppressor genes and thyroid-specific genes that normally help cells take up iodine. The result is a genetic instruction book where many safety chapters are blacked out, encouraging uncontrolled growth and loss of normal thyroid identity.
How cancer cells rewire their fuel lines
At the same time, thyroid tumor cells overhaul how they get and use energy. Instead of mainly burning fuel efficiently in mitochondria, they lean heavily on fast but wasteful sugar breakdown, generating acid by-products that help them evade the immune system. They also ramp up the making and burning of fats, and re-route amino acids such as serine and glutamine to support rapid division. These shifts are not random: they are steered by cancer-driving signals and create a flexible metabolic network that lets tumor cells survive low oxygen, resist drugs, and grow in new locations.
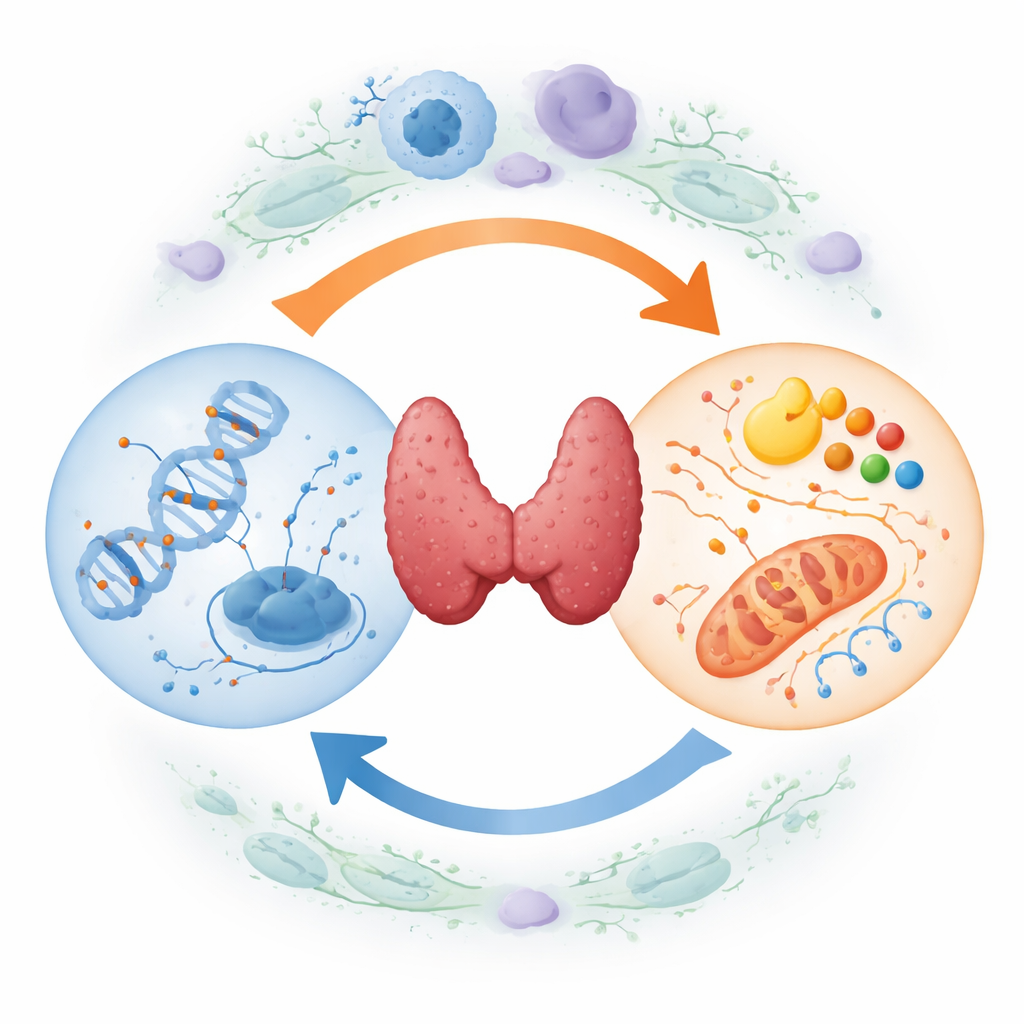
Two-way traffic between DNA tags and cell metabolism
A central message of the article is that DNA marking and fuel use are not separate stories—they form a self-reinforcing loop. On one side, the altered metabolism of tumor cells produces specific small molecules that act as switches for the enzymes placing or removing methyl tags on DNA. When levels of these molecules change, they can push the genome toward more or less methylation at sensitive sites. On the other side, DNA methylation can directly silence or boost genes that control sugar use, fat handling, and amino acid transport. Over time, this two-way traffic locks in a cancer-friendly state in which abnormal DNA markings and distorted metabolism constantly strengthen one another.
Why some thyroid cancers stop drinking iodine
This feedback loop has very practical consequences for patients. Radioactive iodine therapy works only if tumor cells keep the molecular machinery needed to pull iodine inside. The review highlights that key thyroid genes responsible for this, including the thyroid-stimulating hormone receptor and the sodium–iodide transporter, are often shut down by heavy methyl tagging of their control regions. Once this happens, cancer cells “forget” they are thyroid cells and no longer absorb iodine, making standard therapy fail. The authors propose that carefully reversing these DNA marks, or blocking the metabolic pathways that support them, could reawaken iodine uptake and restore sensitivity to treatment.
New treatment ideas built on an integrated view
Seeing thyroid cancer as a disease of a “DNA methylation–metabolism axis” leads to new therapeutic strategies. Rather than relying on single drugs, the authors argue for combination approaches that hit several parts of the loop at once—epigenetic drugs that loosen harmful DNA tags, metabolic drugs that cut off key fuel routes, and signal-blocking drugs that turn down the main cancer-driving pathways. Adding modern immunotherapies and advanced drug delivery systems, such as smart nanoparticles, could further tip the balance by reshaping the tumor’s immune environment while limiting side effects. Early laboratory and clinical data suggest that such coordinated attacks may slow disease, restore radioactive iodine uptake in some patients, and delay resistance.
What this means for future care
For a lay reader, the takeaway is that aggressive thyroid cancers may be curable not by finding a single magic bullet, but by understanding and disrupting the vicious cycle between DNA markings and cell metabolism. By mapping this axis in detail and tracking these changes in blood and tumor samples, doctors may one day match patients to tailored combinations of epigenetic, metabolic, targeted, and immune therapies. If successful, this integrated approach could turn today’s most stubborn thyroid cancers into conditions that are once again treatable—and possibly prevent them from becoming so aggressive in the first place.
Citation: Zhang, T., Han, H., Zhang, Y. et al. The molecular mechanisms and potential therapeutic implications of the crosstalk between DNA methylation and metabolic reprogramming in thyroid cancer. Cell Death Discov. 12, 110 (2026). https://doi.org/10.1038/s41420-026-02981-8
Keywords: thyroid cancer, DNA methylation, cancer metabolism, radioiodine resistance, targeted therapy