Clear Sky Science · en
AdipoR1–AMPK axis suppresses breast cancer across molecular subtypes via multimodal cell death pathways, including ferroptosis and apoptosis
Why fat cells matter for breast cancer
Most of us think of body fat as a passive energy store, but fat cells are active factories that release hormones and signaling molecules into the bloodstream. This study explores how one of those fat‑derived signals, acting through a molecule called AdipoR1 on breast cancer cells, can slow tumor growth. The work is important because it suggests a new way to treat many forms of breast cancer by harnessing communication between fat tissue and tumors, potentially boosting the effects of existing drugs.
Looking across many cancers
The researchers began by mining large public cancer databases that contain genetic information from thousands of patient tumors. They focused on about three dozen genes involved in signals coming from fat tissue and the cell structures that receive those signals. When they compared tumors from 31 different organs with healthy tissue, one pattern stood out: breast cancers showed especially strong changes in these fat‑related signals. In particular, the receptor AdipoR1 was frequently present at higher‑than‑normal levels in breast tumors, regardless of whether they were hormone‑driven, HER2‑positive, or the more aggressive triple‑negative type. Tissue samples from more than 600 breast cancers confirmed that AdipoR1 protein was widely present in tumor cells.
Testing a fat‑signal drug on cancer cells
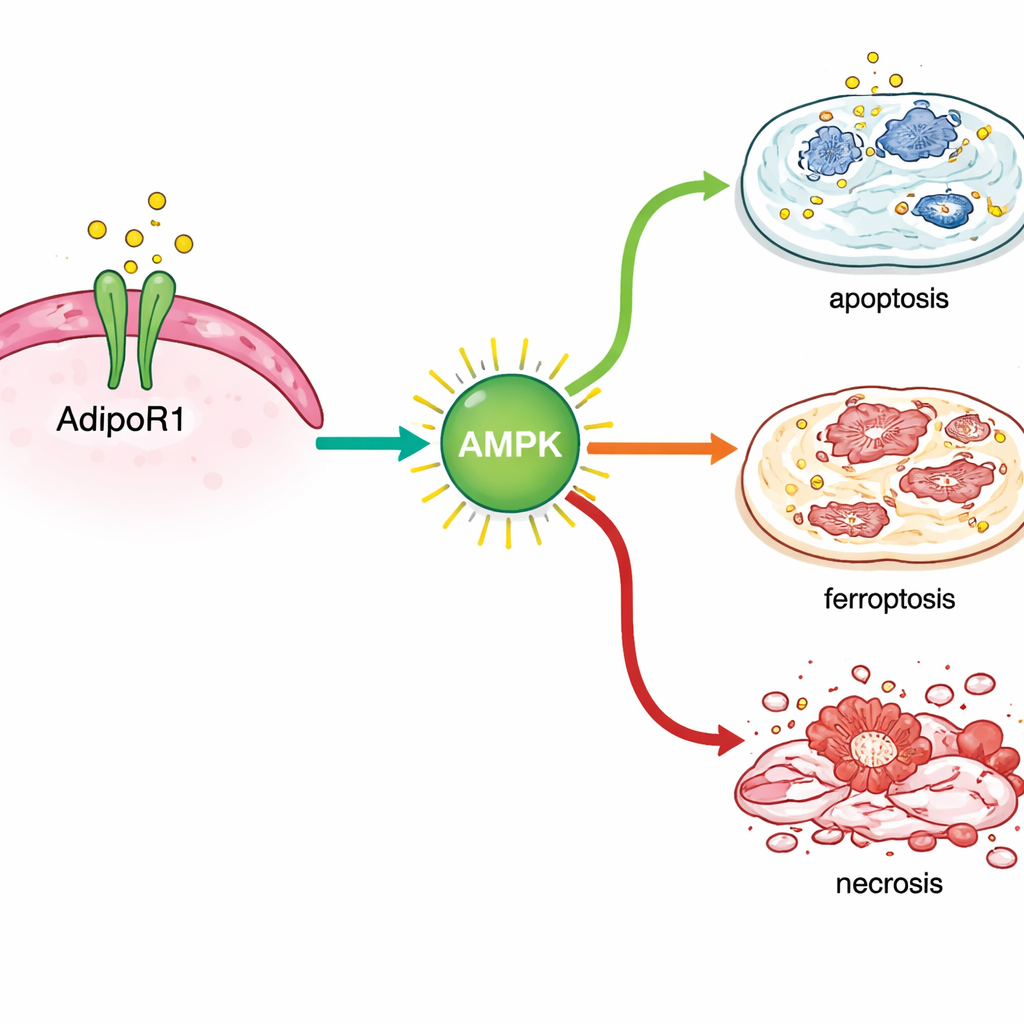
To see what AdipoR1 actually does inside tumors, the team turned to breast cancer cell lines grown in the lab, representing several common subtypes. They treated these cells with AdipoRon, a small molecule that switches on AdipoR1. AdipoRon rapidly triggered a known energy‑sensing switch in cells called AMPK, showing that the signal was getting through. As the dose of AdipoRon increased, cancer cells divided more slowly, moved less in wound‑healing tests, and many underwent programmed self‑destruction. When the researchers deliberately reduced AdipoR1 levels in the cells, AdipoRon lost much of its power, whereas boosting AdipoR1 made the drug more effective, tying the effect directly to this receptor.
Multiple ways to push cancer cells toward death
By analyzing gene activity after treatment, the scientists found that AdipoRon turned on stress responses inside breast cancer cells. It activated genes linked to standard programmed cell death as well as those involved in a more recently recognized form of iron‑dependent cell destruction called ferroptosis. Key markers of this process rose at both the RNA and protein level, especially in hormone‑sensitive breast cancer cells, and chemical readouts of damaged cell lipids supported this finding. At the same time, AdipoRon reduced the levels of proteins that normally help cancer cells survive, including DNA repair helpers (BRCA1 and BRCA2), the estrogen receptor that drives growth in many tumors, and TROP2, a surface molecule tied to aggressive behavior.
Working with, not against, current treatments
The team then asked whether activating AdipoR1 could complement standard drugs. In hormone‑sensitive breast cancer cells, AdipoRon combined with tamoxifen produced greater growth suppression than either drug alone. In triple‑negative lines, AdipoRon enhanced the effects of common chemotherapy agents such as paclitaxel and doxorubicin. Moving to mice bearing hormone‑sensitive breast tumors, the researchers gave AdipoRon by mouth. Tumors in treated animals grew more slowly and contained larger dead zones when examined under the microscope. Importantly, the mice did not show obvious side effects in behavior, weight, or liver health over the course of treatment.
What this could mean for patients
Taken together, the findings suggest that turning on AdipoR1 in breast cancer cells engages several overlapping routes to cell death while weakening key survival systems, and that this approach works across different genetic forms of the disease. Rather than replacing current therapies, AdipoR1‑targeting drugs like AdipoRon could act as add‑ons that make tumors more vulnerable to hormone therapy, chemotherapy, or future targeted agents. Before this strategy can reach the clinic, researchers will need to develop more potent and selective AdipoR1 activators, clarify which patients’ tumors express the receptor most strongly, and carefully evaluate safety. Still, this work points to an intriguing idea: the signals from our own fat tissue could be redirected to help fight breast cancer instead of fueling it.
Citation: Sato, S., Yamanaka, T., Komori, Y. et al. AdipoR1–AMPK axis suppresses breast cancer across molecular subtypes via multimodal cell death pathways, including ferroptosis and apoptosis. Cell Death Dis 17, 384 (2026). https://doi.org/10.1038/s41419-026-08583-7
Keywords: breast cancer, adipokines, AdipoR1, cell death pathways, cancer metabolism