Clear Sky Science · en
K48 and K63 linkage-competed ubiquitination of BECN1 promotes circPDE4D-mediated autophagy in chronic obstructive pulmonary disease
Why this research matters for people with lung disease
Chronic obstructive pulmonary disease (COPD) makes it hard to breathe and is a major cause of illness and death worldwide. Current treatments can ease symptoms but do little to stop the slow damage to lung tissue. This study uncovers a hidden self‑protection system inside lung cells—built from unusual circular genetic fragments and protein tags—that can calm inflammation, remove damaged material, and improve lung function. Understanding this built‑in defense could open doors to new diagnostic tests and therapies for people living with COPD.
A closer look at the lung’s cleanup crew
Our cells constantly tidy themselves through a process called self‑eating, or autophagy, in which unwanted or damaged components are packed into small bubbles and broken down. In COPD, this cleanup process often goes wrong, contributing to ongoing inflammation and tissue destruction. The authors focused on a key autophagy switch called BECN1 and on tiny circular RNA molecules that influence how strongly this switch is turned on. They discovered that one specific circular RNA, called circPDE4D, is consistently reduced in blood and airway cells from people with COPD and in cells and mice exposed to cigarette smoke, a main cause of the disease. Lower levels of circPDE4D were linked to poorer lung function, suggesting that this molecule might serve as both a disease marker and a protective factor.
How a circular RNA calms stressed lung cells
To test circPDE4D’s role, the researchers experimentally increased or decreased it in human airway and lung cells. Raising circPDE4D levels helped cells survive stress, boosted their capacity for autophagy, and encouraged the formation of stress granules—temporary holding areas that help cells ride out harsh conditions. At the same time, this boost reduced signals of inflammation, including the inflammatory molecules IL‑1β and IL‑6. When circPDE4D was lowered, the opposite happened: more cell death, less autophagy, weaker stress‑response structures, and higher inflammation. In mice chronically exposed to cigarette smoke, delivering extra circPDE4D directly to the lungs lessened tissue damage, reduced scarring and mucus‑producing cells, lowered inflammatory signals, and measurably improved lung‑function readings.
The hidden chain of molecular middlemen
CircPDE4D does not act alone. It works by soaking up a tiny regulatory molecule, miR545‑3p, that would otherwise suppress a helpful enzyme named SMURF1. This enzyme adds small “ubiquitin” tags to proteins, marks that can either send proteins for destruction or fine‑tune their activity. In COPD cells and patient samples, SMURF1 levels were also reduced, but they rose again when circPDE4D was restored. When miR545‑3p was added back or SMURF1 was blocked, the benefits of circPDE4D—better cell survival, stronger autophagy, more stress granules, and lower inflammation—were largely lost. These experiments place SMURF1 at the center of a chain: circPDE4D restrains miR545‑3p, which frees SMURF1 to support the cell’s protective responses.
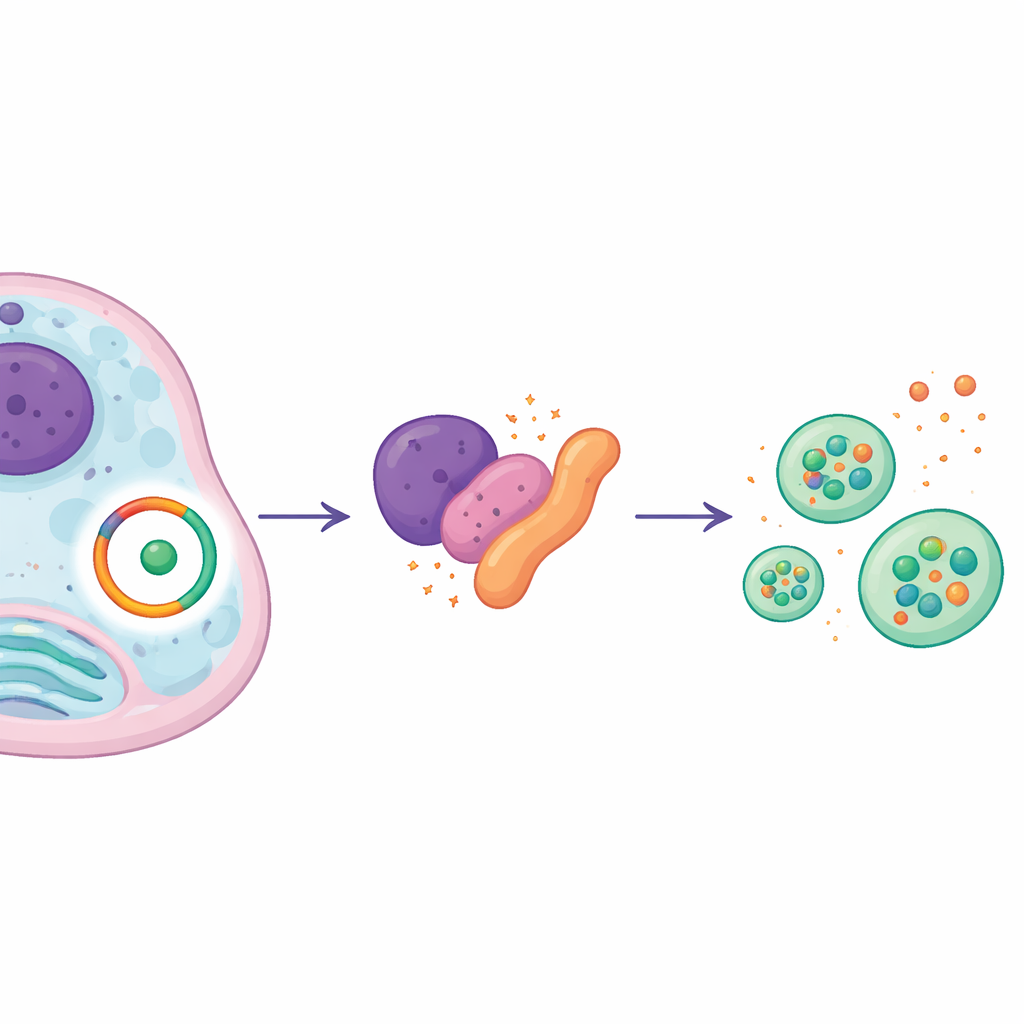
Fine‑tuning the master switch for cellular cleanup
Digging deeper, the team found that SMURF1 physically binds to BECN1, the core autophagy switch, and changes how it is tagged with ubiquitin. One type of tag (K48‑linked) acts like a disposal signal, steering BECN1 toward destruction, while another (K63‑linked) supports its stability and function. SMURF1 tipped this balance by reducing the destructive tags and increasing the protective ones on BECN1, effectively extending the life of this cleanup switch inside cells. Strikingly, when circPDE4D was removed, SMURF1 could no longer reshape these tags on BECN1, and autophagy weakened. This shows that circPDE4D is required for SMURF1 to properly stabilize BECN1 and keep the cell’s cleanup machinery running.
What this could mean for future COPD care
Together, the findings outline a self‑reinforcing protective loop in lung cells: circPDE4D blocks miR545‑3p, which allows SMURF1 to adjust the tags on BECN1 so that it stays active and supports autophagy, stress handling, and lower inflammation. When this loop is weakened, as seen in COPD, lungs are left more vulnerable to cigarette smoke and other irritants. By highlighting circPDE4D as both a potential blood‑based marker and a molecular switch that can restore the cell’s cleanup and anti‑inflammatory systems, this work points toward new strategies to diagnose, monitor, and perhaps one day treat COPD by nudging the cell’s own defenses back into balance.
Citation: Chen, TT., Wang, MY., Kang, JY. et al. K48 and K63 linkage-competed ubiquitination of BECN1 promotes circPDE4D-mediated autophagy in chronic obstructive pulmonary disease. Cell Death Dis 17, 321 (2026). https://doi.org/10.1038/s41419-026-08582-8
Keywords: COPD, autophagy, circular RNA, lung inflammation, ubiquitination