Clear Sky Science · en
Role of lipid rafts in the FGFR2c-mediated oncogenic signaling by involvement of TRPA1 channel in pancreatic ductal adenocarcinoma cells
Why small islands in cell membranes matter
Pancreatic cancer is one of the deadliest cancers, in part because its cells are unusually good at breaking away from the original tumor and invading nearby tissues. This study looks at tiny “islands” in the outer skin of cancer cells, made of cholesterol and fats, and asks a simple question with big implications: do these islands help switch on a growth signal that makes pancreatic cancer more aggressive, and can disrupting them slow the process down?
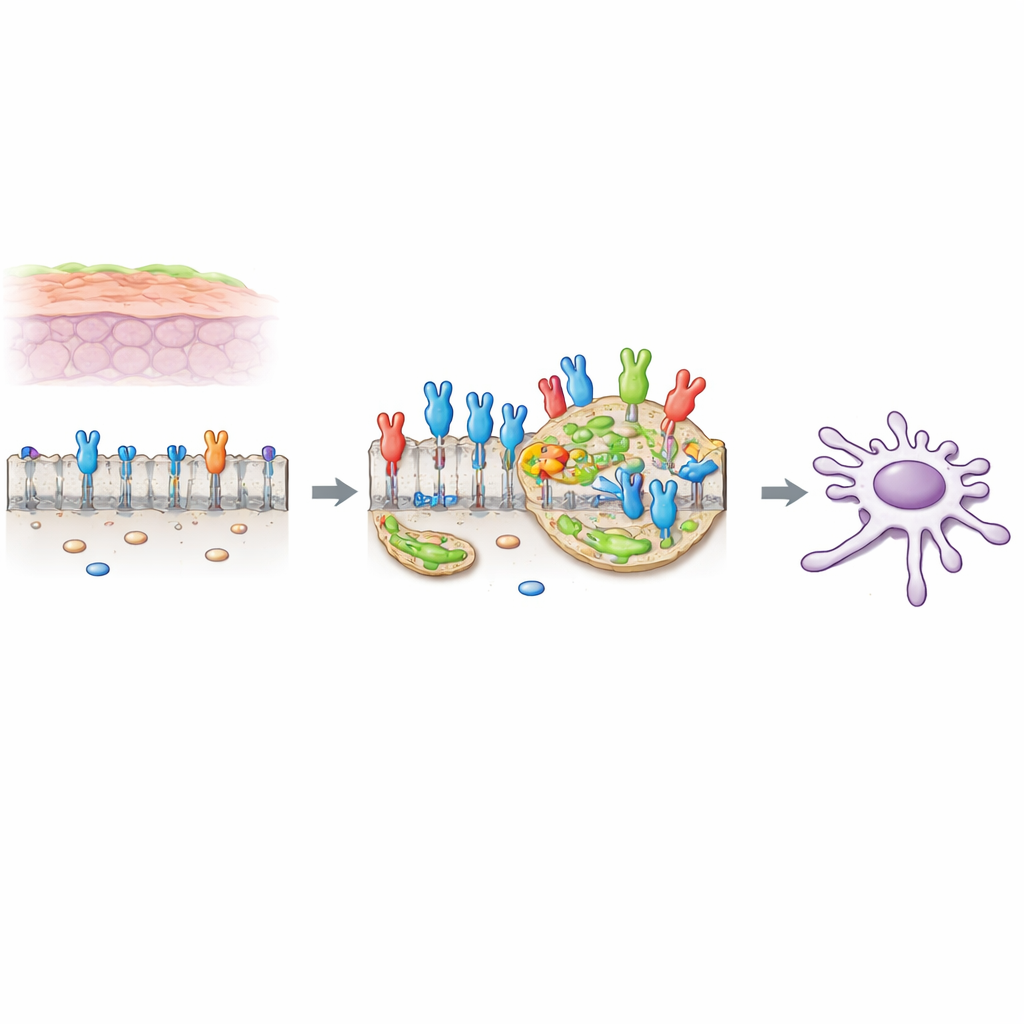
A cancer-promoting switch on the cell surface
Many cells carry switches on their surface that respond to growth signals. One of these switches, called FGFR2c, is a variant normally found in more flexible, mobile cells, but it becomes abnormally abundant in pancreatic ductal adenocarcinoma (PDAC) cells. When this switch is turned on by its partner molecule in the environment, it drives changes known as epithelial–mesenchymal transition (EMT), which make cells less attached and more able to move and invade. Earlier work from the authors showed that, in PDAC cells, FGFR2c activates a chain of internal messengers involving a protein called PKCε and several classic growth pathways, boosting cell survival, movement and invasion.
Cholesterol-rich islands as signal amplifiers
The outer membrane of a cell is not uniform. It contains small, cholesterol-rich patches called lipid rafts, which act as miniature platforms where signaling molecules can cluster and communicate more efficiently. The researchers found that when FGFR2c is activated by its external signal, more of the receptor moves into these lipid rafts. Using fluorescent markers and biochemical fractionation, they showed that activated FGFR2c concentrates in these patches, while its distribution outside the rafts changes from smooth to dotted patterns, consistent with receptor clustering on these islands.
Breaking the islands weakens cancer traits
To test whether these lipid islands are essential for the cancer-promoting effects of FGFR2c, the team used a compound (methyl-β-cyclodextrin) that selectively removes cholesterol from the membrane and destabilizes lipid rafts without killing the cells. In PDAC cells rich in FGFR2c, disrupting the rafts strongly reduced activation of key signaling molecules downstream of the receptor and blunted the EMT program: genes and proteins associated with a mobile, invasive state decreased, while epithelial features were partly restored. The same treatment also lowered levels of invasion-linked proteins such as MCL-1, SRC and matrix-degrading enzymes, and it reduced the ability of cancer cells to move and penetrate a gel that mimics surrounding tissue. Cells with very little FGFR2c showed almost no response, underscoring how central this receptor–raft partnership is in these tumors.
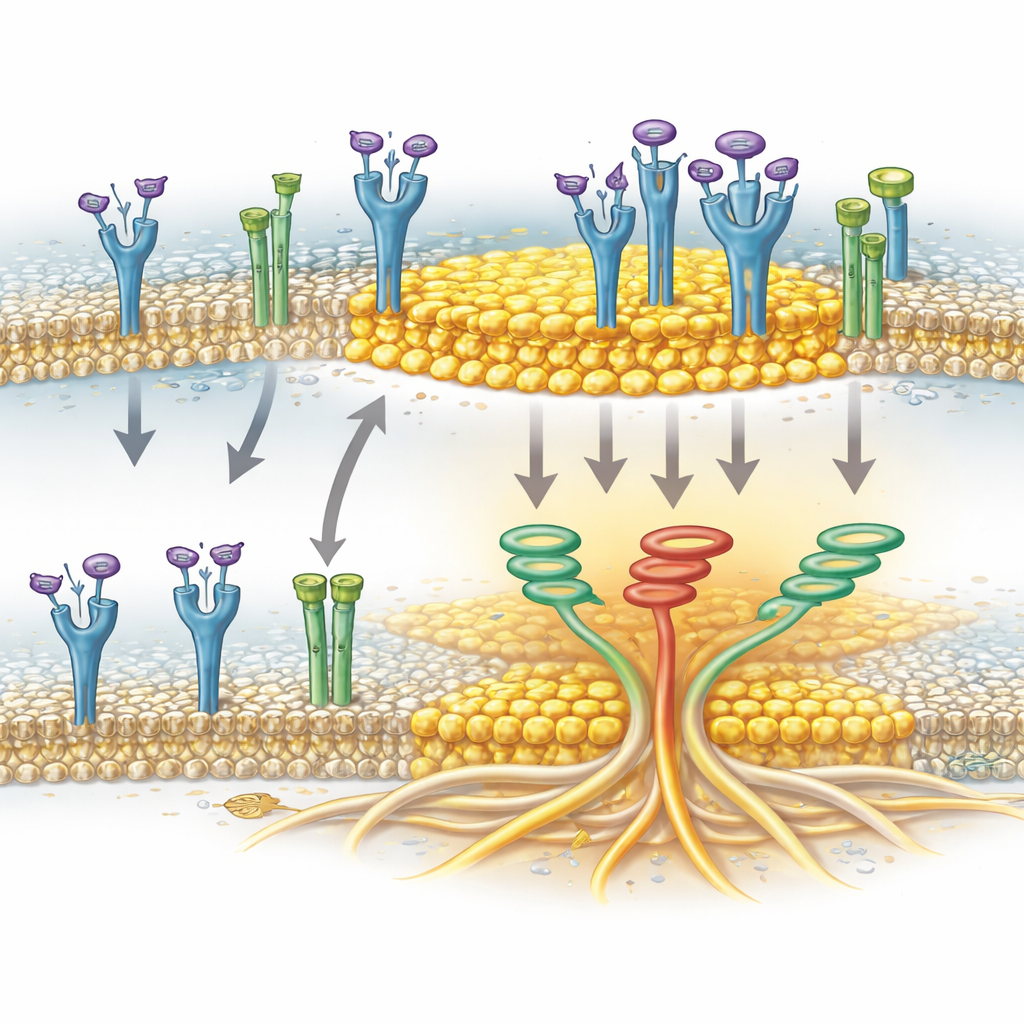
An ion channel as a helper for clustering
The study also identifies a surprising helper: TRPA1, a channel protein best known for sensing irritants and oxidative stress in nerves. In pancreatic cancer cells, TRPA1 is present at the membrane and can reside in lipid rafts. The authors showed that TRPA1 physically associates with FGFR2c when the receptor is activated and that this association coincides with FGFR2c’s enrichment in raft fractions. When TRPA1 levels were reduced, activated FGFR2c no longer accumulated efficiently in the raft regions, even though the rafts themselves remained intact. This indicates that TRPA1 is not just a passive passenger but actively helps escort or stabilize FGFR2c within these cholesterol-rich platforms, where the receptor can more effectively trigger the invasive signaling network.
What this means for future treatments
Taken together, the work paints a picture in which FGFR2c, TRPA1 and lipid rafts form a cooperative unit that strengthens the invasive behavior of pancreatic cancer cells. FGFR2c provides the growth signal, lipid rafts provide the physical stage where signaling complexes assemble, and TRPA1 helps bring the receptor onto that stage. For a lay reader, the key takeaway is that not only the cancer-causing molecules themselves, but also the tiny membrane landscapes they occupy, can determine how dangerous a tumor becomes. By targeting FGFR2c, TRPA1, or the stability of these cholesterol-rich islands—alone or in combination—future therapies may be able to weaken the invasive machinery of pancreatic cancer cells in a more precise, tumor-specific way.
Citation: Mancini, V., Manganelli, V., Garofalo, T. et al. Role of lipid rafts in the FGFR2c-mediated oncogenic signaling by involvement of TRPA1 channel in pancreatic ductal adenocarcinoma cells. Cell Death Dis 17, 259 (2026). https://doi.org/10.1038/s41419-026-08513-7
Keywords: pancreatic cancer, lipid rafts, FGFR2c, TRPA1 channel, cell invasion