Clear Sky Science · en
PRDM1 restricts bladder cancer progression and enhances chemosensitivity by suppressing OTUD6A-mediated deubiquitination of CDC6
Why this research matters for patients
Bladder cancer is common and often treated with chemotherapy drugs such as gemcitabine and cisplatin. Yet many tumors either do not respond well or quickly become resistant, leaving patients with few options. This study uncovers a built-in protective brake inside bladder cells, a protein called PRDM1, that slows tumor growth and helps chemotherapy work better. Understanding how this brake functions — and how tumors switch it off — could open new ways to predict treatment response and design smarter therapies.
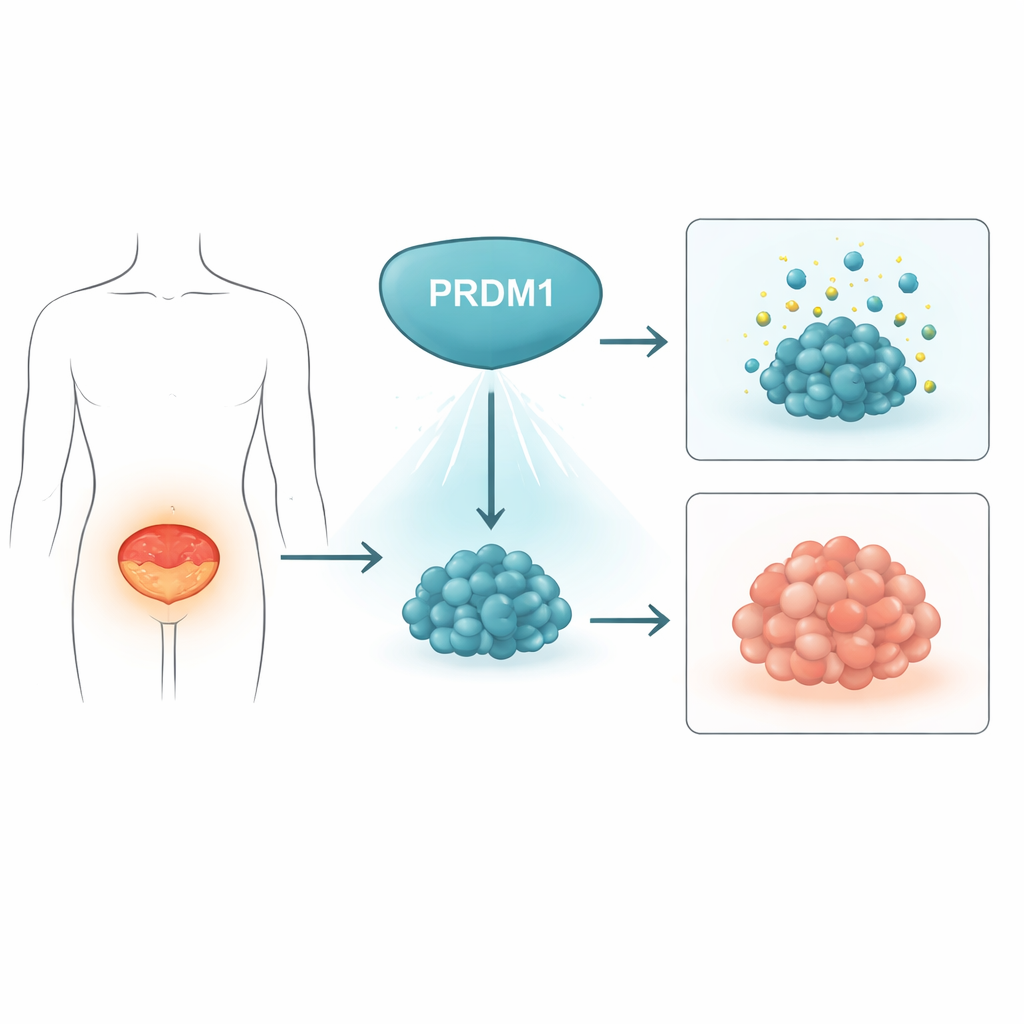
A natural brake on bladder tumors
The researchers first asked whether PRDM1, a gene known to control how immune cells develop, might also influence bladder cancer. By examining 48 pairs of human bladder tumors and nearby normal tissue, they found that PRDM1 protein levels were consistently lower in tumors than in healthy bladder lining. Large public cancer datasets showed the same pattern at the RNA level. In a mouse model in which bladder cancer is induced over time by a chemical in the drinking water, PRDM1 levels dropped progressively as normal tissue turned cancerous. Together, these data suggest that PRDM1 normally acts as a brake on tumor formation and is switched off as bladder cancer develops.
Slowing cancer cell growth and cell division
To explore what PRDM1 actually does inside bladder cancer cells, the team used genetic tools to turn the gene down or up. When they reduced PRDM1 in bladder cancer cell lines, the cells multiplied faster, formed more colonies, and grew larger tumors in mice. Completely deleting PRDM1 using CRISPR gene editing had similar effects. In contrast, forcing cells to make more PRDM1 slowed their growth in dishes and led to much smaller tumors in animal models, along with lower levels of the cell division marker Ki-67. Detailed analysis of the cell cycle showed that extra PRDM1 caused a delay as cells attempted to move from mitosis into the next growth phase, indicating that the protein helps keep cell division in check.
Making chemotherapy hit harder
Because chemotherapy success depends heavily on how well drugs damage tumor DNA and trigger cell death, the authors investigated whether PRDM1 affects drug sensitivity. Cells lacking PRDM1 were harder to kill with gemcitabine or cisplatin, requiring higher doses to achieve the same effect, whereas cells with extra PRDM1 became more sensitive. PRDM1-deficient cells showed less DNA damage and fewer dying cells after treatment, while PRDM1-overexpressing cells displayed more DNA breaks and higher levels of apoptotic markers. The study also examined a key DNA damage signaling route known as the ATR–Chk1 pathway, which helps cells survive chemotherapy. Loss of PRDM1 boosted this survival signaling, whereas increasing PRDM1 dampened it. In mouse tumor models, tumors with high PRDM1 responded better to gemcitabine, and restoring PRDM1 in drug-resistant cells partly reversed resistance.
Uncovering a three-step control pathway
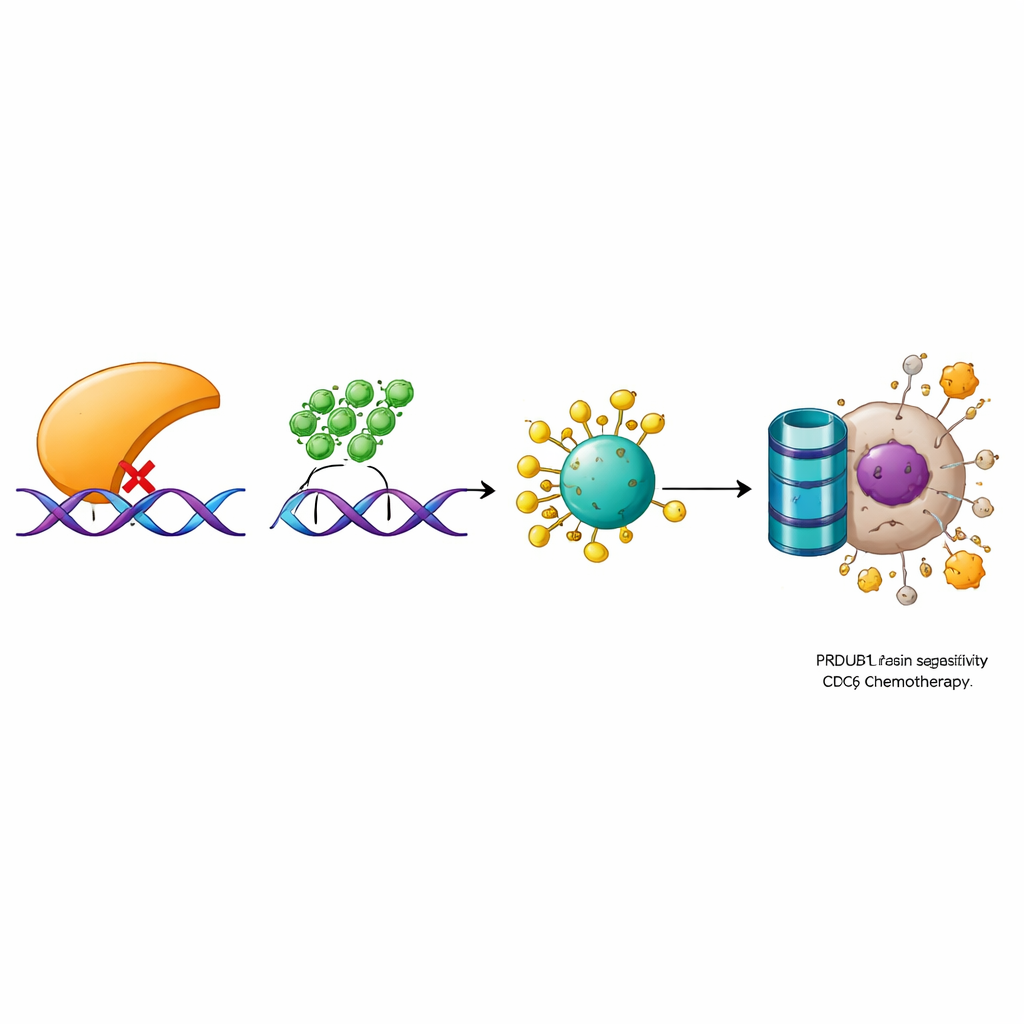
Diving deeper, the scientists identified a molecular chain that connects PRDM1 to cell growth and drug response. In earlier work, they had shown that an enzyme called OTUD6A stabilizes another protein, CDC6, which is involved in starting DNA replication and in activating DNA damage signaling. Here they discovered that PRDM1 does not change CDC6’s genetic message but instead promotes its destruction by the cell’s protein disposal machinery. PRDM1 reduces OTUD6A levels, leading to more chemical tags on CDC6 that mark it for breakdown. When OTUD6A or CDC6 were experimentally lowered, they counteracted the growth advantage and drug resistance caused by low PRDM1. Conversely, raising OTUD6A or CDC6 could undo the benefits of high PRDM1. Analyses of patient samples confirmed this relationship: tumors generally had low PRDM1 but high OTUD6A and CDC6, and these proteins’ levels were tightly correlated.
What this could mean for future treatments
Overall, the study outlines a clear pathway in which PRDM1 restrains bladder cancer and boosts chemotherapy response by turning down OTUD6A, which in turn allows the cell to break down CDC6. Without this brake, CDC6 accumulates, cell division speeds up, and tumor cells become more adept at surviving DNA-damaging drugs. For patients, these findings suggest that measuring PRDM1, OTUD6A, and CDC6 levels in tumors could help predict how well chemotherapy will work. In the longer term, drugs that restore PRDM1 activity or block OTUD6A or CDC6 might be developed to re-sensitize resistant bladder cancers and improve treatment outcomes.
Citation: Cui, J., Chen, S., Liu, X. et al. PRDM1 restricts bladder cancer progression and enhances chemosensitivity by suppressing OTUD6A-mediated deubiquitination of CDC6. Cell Death Dis 17, 247 (2026). https://doi.org/10.1038/s41419-026-08498-3
Keywords: bladder cancer, chemoresistance, PRDM1, CDC6, OTUD6A