Clear Sky Science · en
Targeting the p53 cancer mutants Y220C, Y220N, and Y220S with the small-molecule stabilizer rezatapopt
Why this matters for future cancer treatments
Cancer often disarms p53, a protein sometimes called the cell’s “guardian,” by mutating it so it can no longer keep dangerous cells in check. One particular change in p53, known as Y220C, is common in solid tumors and has become a test case for drugs that aim to repair broken p53 rather than kill cells outright. This study asks a practical, patient-focused question: can a promising experimental drug, rezatapopt, that is already in clinical trials for Y220C also help people whose tumors carry closely related but rarer mutations at the same position in p53?
A drug designed to patch a crack in p53
Many p53 mutations make the protein flop open and lose its precise shape. In the Y220C mutation, the change creates a small cavity on the protein surface—a defect that chemists realized could be “plugged” with tailored small molecules to stabilize p53. Rezatapopt is one such molecule, engineered by building on earlier chemical series that bind into this cavity and act like a molecular brace. The authors focus on two other mutations at the same amino-acid position, Y220S and Y220N, which also carve out a similar pocket but destabilize p53 even more than Y220C. If rezatapopt could also patch these variants, it might extend this precision-medicine approach to thousands more patients each year. 
Measuring how well rezatapopt grips and steadies mutant p53
To test this, the team purified the DNA-binding core of p53 with Y220C, Y220S, or Y220N changes and exposed each to rezatapopt (and close chemical cousins). Using heat-based assays, they showed that all three mutant proteins became more stable when the drug bound, with Y220C and Y220S effectively regaining wild-type–like sturdiness. By contrast, Y220N recovered only part of its lost stability. A second technique that measures the heat released on binding revealed why: rezatapopt attaches very tightly to Y220C (around a few tens of nanomolar), somewhat less strongly to Y220S, and more weakly still to Y220N, though all are within ranges medicinal chemists consider promising.
Seeing the molecular patch at atomic detail
High-resolution X-ray crystal structures provided a visual explanation. In all three mutants, rezatapopt nestles into the mutation-made cleft with a conserved pose: its central scaffold fills the cavity, one end reaches deep into the pocket with a cluster of fluorine atoms, and the other end interacts with a nearby protein loop. The drug makes a network of contacts, including a strategically placed fluorine atom that engages the protein backbone. For Y220S, these interactions are preserved with only minor shifts, allowing strong stabilization. For Y220N, however, binding the drug forces the asparagine side chain into the oily core of the protein in an energetically unfavorable way, and some contacts with the drug’s fluorinated “anchor” are lost. This strained fit explains both the weaker binding and the incomplete rescue of stability. 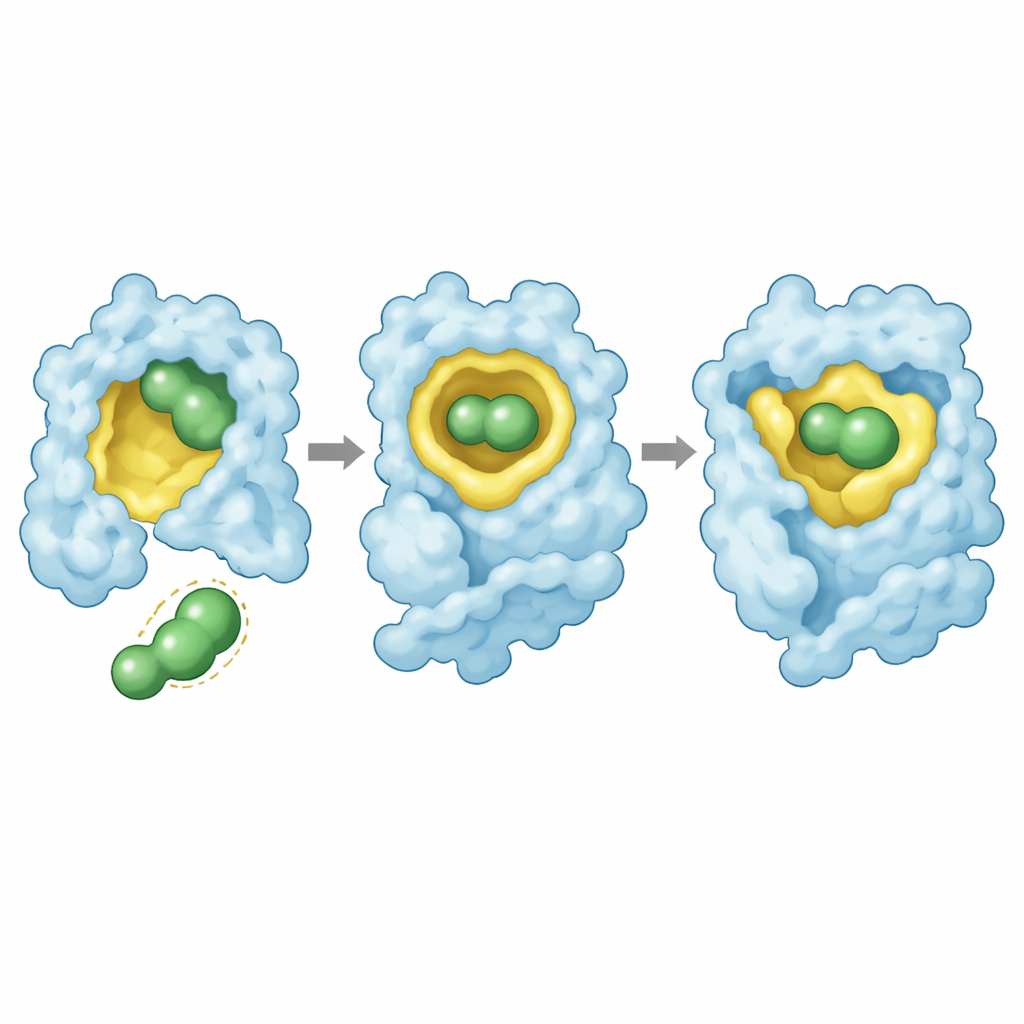
From protein repair to cell behavior
The crucial test is whether such stabilization revives p53’s protective role inside living cells. The researchers engineered lung cancer cells lacking natural p53 to express either the Y220C, Y220S, Y220N, or Y220H variants. When treated with rezatapopt, cells bearing Y220C showed a powerful activation of classic p53 target genes involved in halting the cell cycle and triggering cell death. Y220S cells displayed a very similar pattern—recovery of the folded, active form of p53, strong gene activation, slowed proliferation, and increased cell death—but only at drug concentrations more than ten times higher than those needed for Y220C. Y220N cells, by comparison, showed no clear target-gene activation at tolerated doses and remained largely in the misfolded state, although slight late-onset growth effects hint at only partial functional rescue.
What this means for patients and future drug design
For people with tumors carrying the Y220S mutation, these findings are cautiously encouraging: rezatapopt can, in principle, restore p53’s protective functions, but only at substantially higher doses, which may be difficult to achieve safely in patients. For Y220N, the current drug appears insufficient to fully repair p53 under realistic conditions. Still, the structural blueprints in this work outline why the current design underperforms and how future molecules might be adjusted to engage all three mutant side chains without forcing them into unfavorable positions. In other words, creating a true “pan-Y220” p53 repair drug looks challenging but achievable, and would modestly expand the number of patients who might benefit from this emerging strategy of fixing, rather than replacing, the body’s own tumor suppressor.
Citation: Mavridi, D., Funk, J.S., Balourdas, DI. et al. Targeting the p53 cancer mutants Y220C, Y220N, and Y220S with the small-molecule stabilizer rezatapopt. Cell Death Dis 17, 268 (2026). https://doi.org/10.1038/s41419-026-08492-9
Keywords: p53 reactivation, rezatapopt, Y220C mutation, cancer precision medicine, protein stabilization