Clear Sky Science · en
OTUD4 deubiquitination stabilizes EGFR and activates the PI3K/AKT pathway to promote the invasiveness of triple-negative breast cancer
Why this research matters for breast cancer
Triple-negative breast cancer is one of the toughest forms of breast cancer to treat because it lacks the usual hormone and growth markers that many drugs target. This study uncovers a hidden "protector" molecule that helps cancer cells keep a powerful growth switch turned on. By understanding this unseen support system, scientists may find new ways to cut off the signals that drive tumor growth and spread.
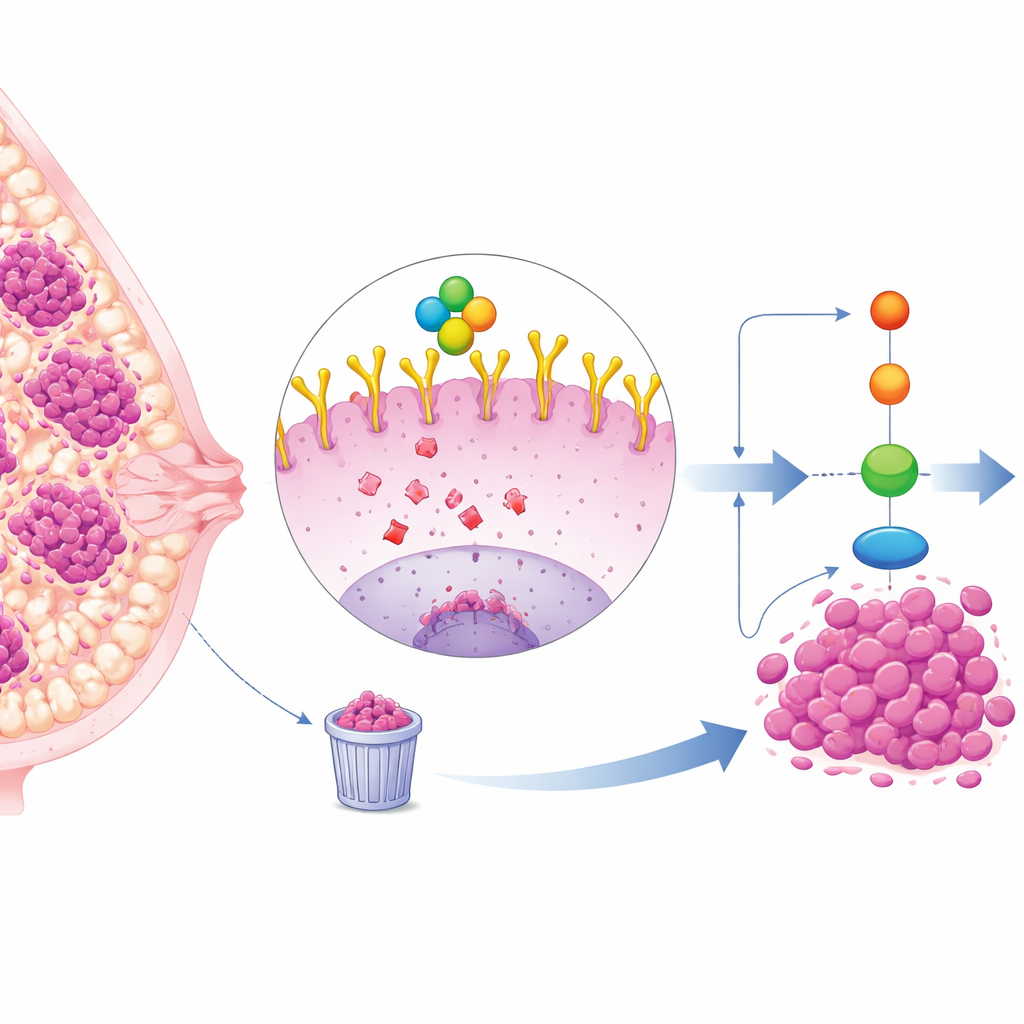
A hard-to-treat breast cancer subtype
Triple-negative breast cancer (TNBC) makes up roughly a quarter of breast cancer cases and tends to grow and spread faster than other subtypes. Because TNBC cells do not carry estrogen, progesterone, or HER2 receptors, standard targeted therapies are less effective, leaving chemotherapy and surgery as the main options. Many TNBC tumors, however, display high levels of a cell-surface protein called EGFR, which acts like an antenna to receive growth and survival signals. High EGFR levels are linked with poor outcomes, but drugs that directly block EGFR have had disappointing results in the clinic, suggesting there are deeper layers of regulation at work.
Finding a hidden helper of tumor growth
The researchers focused on OTUD4, an enzyme that removes small protein tags called ubiquitin from other proteins. These tags often act as disposal signals, marking proteins to be broken down, so removing them can stabilize and protect the tagged protein. Using large cancer databases and tissue samples from patients, the team showed that OTUD4 is present at higher levels in TNBC tumors and cell lines than in normal breast tissue. Patients whose tumors had more OTUD4 tended to have worse survival, suggesting OTUD4 behaves like an oncogene, a gene that helps drive cancer progression.
How OTUD4 makes cancer cells more aggressive
To test what OTUD4 actually does in TNBC cells, the scientists reduced its levels in two widely studied TNBC cell lines. When OTUD4 was knocked down, cancer cells grew more slowly, formed fewer colonies, and were less able to migrate in wound-healing and Transwell assays, all signs of reduced aggressiveness. When OTUD4 was instead boosted, the opposite happened: cells proliferated faster and moved more readily, reinforcing the idea that OTUD4 is a driver of malignant behavior. In mice, tumors formed from cells lacking OTUD4 grew more slowly and showed reduced markers of cell division and growth signals, confirming these effects in living organisms.
A molecular shield that protects EGFR
Digging into the mechanism, the team used protein interaction screens and biochemical assays to identify EGFR as a direct binding partner of OTUD4. They found that OTUD4 attaches to a specific region of EGFR and removes K48-linked ubiquitin chains, the type of tag that normally marks proteins for destruction by the cell’s recycling machinery. When OTUD4 was reduced, EGFR degraded more quickly, but its gene activity stayed the same, showing that OTUD4 acts after EGFR is made, not at the DNA or RNA level. Blocking the cell’s protein disposal system restored EGFR levels, underscoring that the key step is protection from breakdown. With more stable EGFR on the cell surface, a major internal signal route, the PI3K/AKT/mTOR pathway, stays switched on, driving cell growth and survival.
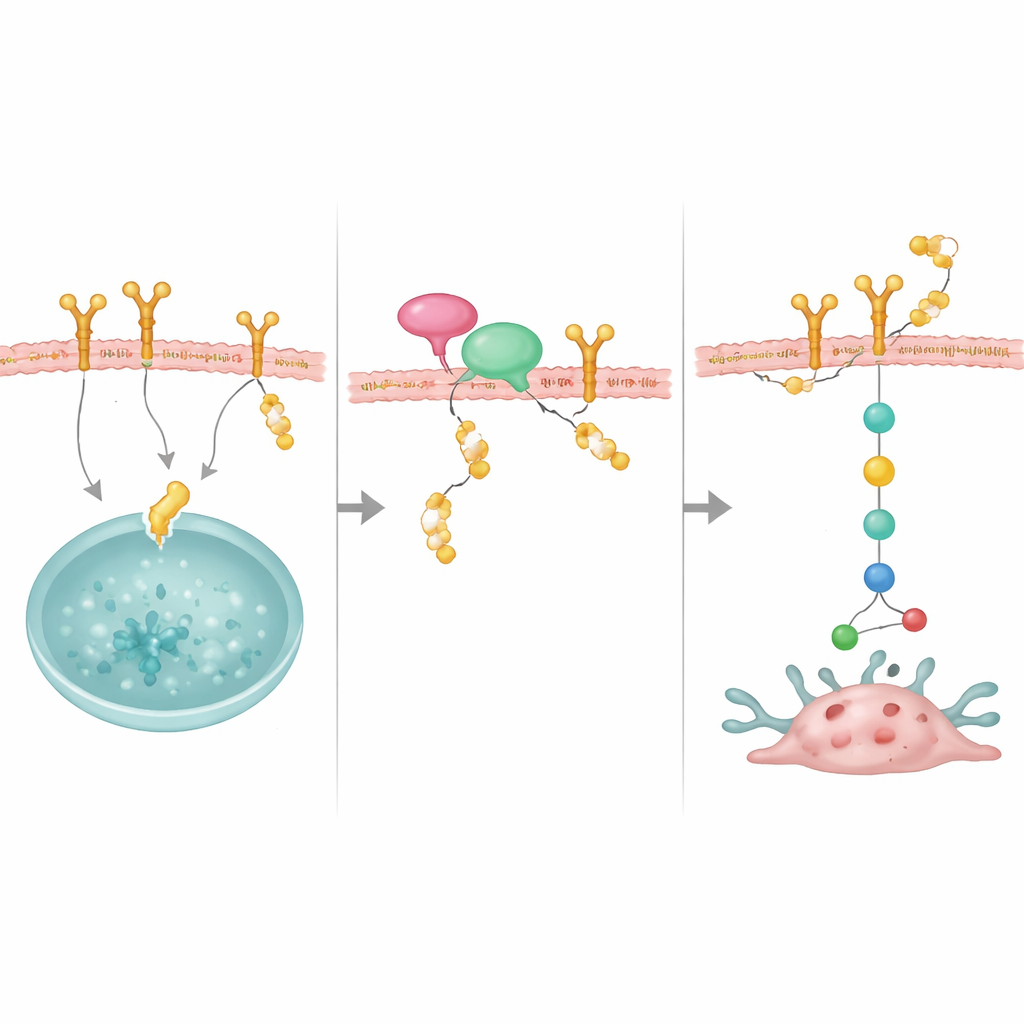
An adaptor that brings the protector to its target
The study also uncovered an important partner, NRP1, a membrane protein previously linked to aggressive breast cancers. NRP1 binds both OTUD4 and EGFR and helps bring OTUD4 into position to rescue EGFR from destruction. When NRP1 was reduced, EGFR became less stable and its growth signals weakened, even though OTUD4 levels did not change. Overproducing OTUD4 could partly overcome the loss of NRP1, suggesting that NRP1 acts like a docking platform that recruits OTUD4 to EGFR. Together, these molecules form a stabilizing complex that keeps EGFR plentiful at the cell surface.
What this means for future treatment
By revealing how OTUD4 and NRP1 work together to shield EGFR from being broken down, this study identifies a new control point for a key growth signal in triple-negative breast cancer. Instead of trying to block EGFR’s activity at its active site, future therapies might aim to disrupt the protective actions of OTUD4 or its recruitment by NRP1, lowering EGFR levels and dampening the PI3K/AKT pathway that fuels tumor growth. While more work is needed to find safe and effective OTUD4 inhibitors and to understand possible escape routes used by cancer cells, the findings open up promising avenues for more precise treatments against this challenging breast cancer subtype.
Citation: Ren, Y., Zhou, F., Tan, Z. et al. OTUD4 deubiquitination stabilizes EGFR and activates the PI3K/AKT pathway to promote the invasiveness of triple-negative breast cancer. Cell Death Dis 17, 245 (2026). https://doi.org/10.1038/s41419-026-08482-x
Keywords: triple-negative breast cancer, EGFR signaling, OTUD4, PI3K AKT pathway, protein degradation