Clear Sky Science · en
Spatial lipidomic and neuron-specific transcriptomic signatures in the nucleus accumbens reveal phospholipid dyshomeostasis in depression-related maladaptations
Why Brain Fats Matter for Mood
Depression is often explained in terms of brain chemicals like serotonin, but our brains are also packed with fats that quietly keep nerve cells working. This study looks at how specific brain fats in a key reward center go out of balance under stress and genetic risk, and how that imbalance may help drive low mood, loss of pleasure, and social withdrawal in depression.
A Closer Look at the Brain’s Reward Hub
The researchers focused on the nucleus accumbens, a small but crucial region that helps us feel pleasure, stay motivated, and cope with stress. Changes in this area have been linked to depression in people, and deep brain stimulation there can ease symptoms in some severe cases. The team used mice lacking a protein called p11, which has been tied to depression-like behavior and is naturally enriched in this reward hub, and combined this genetic vulnerability with chronic restraint stress to mimic real-life pressure.
When Stress and Vulnerability Combine
In behavioral tests, mice missing p11 showed classic signs that mirror core symptoms of depression. They spent less time engaging in social interaction, were less interested in sweetened water (a readout of pleasure and reward), and were more immobile in a test that reflects passive coping or despair, even though their basic movement was unchanged. Adding repeated stress made several of these traits worse, suggesting that p11 normally helps the brain resist the emotional toll of ongoing adversity.
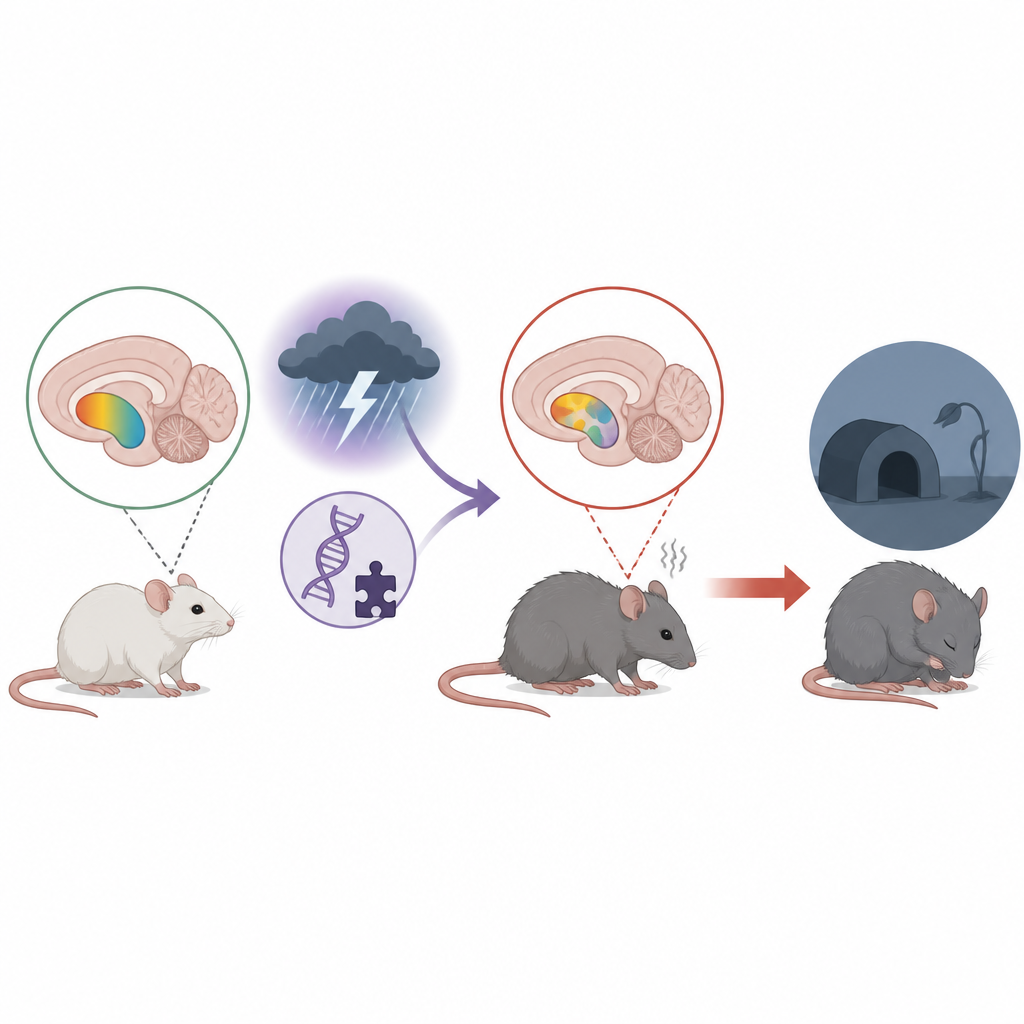
Brain Fats Fall Out of Balance
To see what was happening inside the brain, the team mapped lipid molecules directly in slices of the nucleus accumbens using a form of mass spectrometry imaging, and paired this with high-resolution RNA profiling of neurons in the same region. They found that mice with p11 loss, especially when stressed, had lower levels of several phospholipids, a major class of fats that form the outer skin of cells and help control how signals are passed along. The most consistent changes were in phosphatidylethanolamine (PE) and related “ether” PE species, which together make up a large share of brain membrane fats. Levels of specific PE and ether PE molecules tracked with behavior: animals with more of these fats tended to drink more sucrose and spent less time immobile, hinting at a direct link between membrane composition and mood-related actions.
Gene Shifts in Neurons Reveal a Lipid Pathway Under Strain
Next, the authors zoomed in on neurons within the nucleus accumbens and examined the activity of genes that build, remodel, or break down PE. In the core part of this region, they detected coordinated changes in several enzymes. Some genes that normally support PE production were reduced, while others that direct PE toward breakdown or toward ether-linked forms were increased. These shifts again lined up with behavior: for example, higher levels of one enzyme involved in ether lipid production were associated with worse social and stress-coping scores, whereas higher levels of a transport protein tied to lipid balance went together with better reward seeking and less immobility. This pattern suggests that chronic stress and p11 loss push the PE system away from its sweet spot in vulnerable neurons.
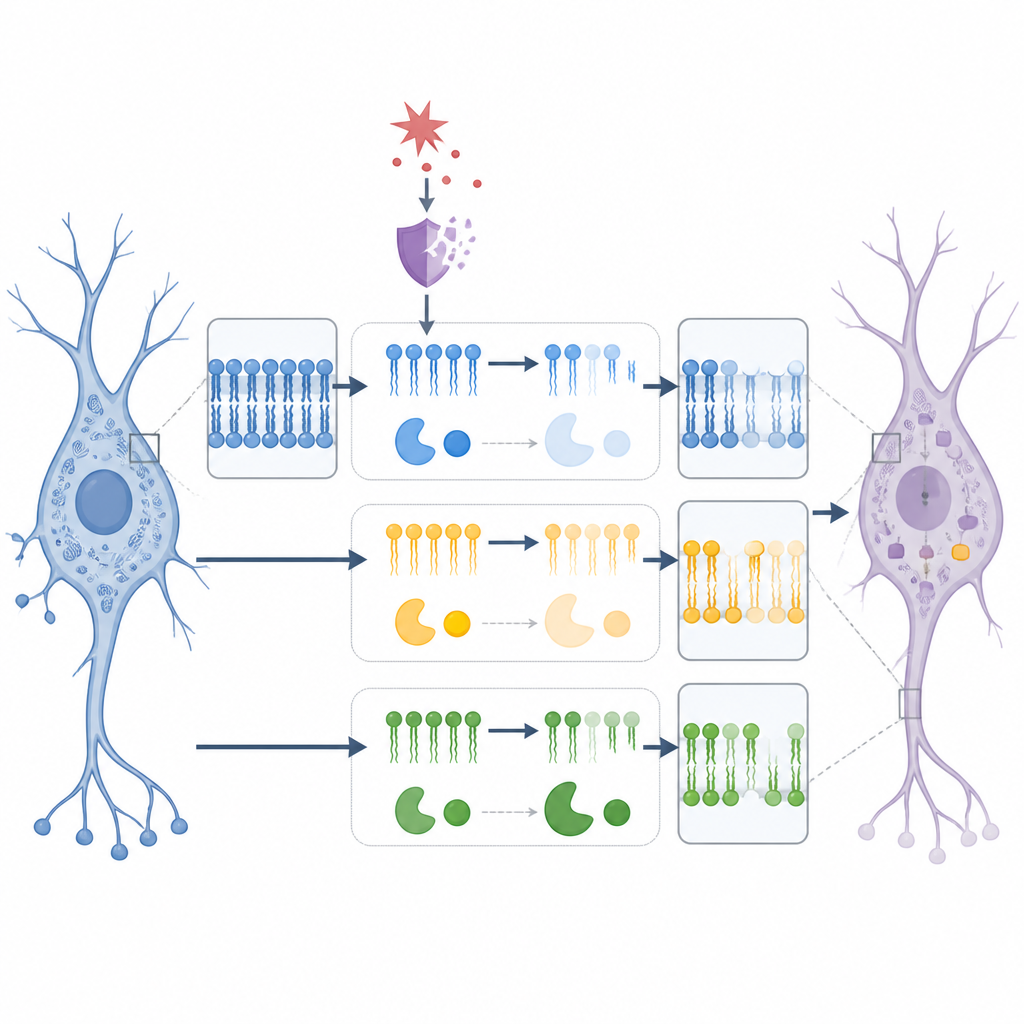
Disrupting Lipids Alone Can Trigger Low Mood Signs
To test whether disturbing phospholipids is enough to affect mood-related behavior, the researchers gave mice a drug called chelerythrine, known to interfere with the synthesis of choline- and ethanolamine-containing phospholipids. After a week of treatment, both normal and p11-deficient mice showed reduced social interaction and lower preference for sucrose, without changes in basic activity. Imaging confirmed that this drug shifted multiple phospholipid species in the nucleus accumbens, including further drops in PE-related fats. Several of these drug-induced lipid changes also tracked with measures of pleasure and social behavior, strengthening the idea that altered membrane fats contribute directly to emotional outcomes.
What This Means for Understanding Depression
This work suggests that depression-like behavior in mice arises not only from changes in classic brain chemicals but also from subtle, spatially precise disruptions in the fats that build neuronal membranes in a key reward circuit. In particular, phosphatidylethanolamine and its related pathways in nucleus accumbens neurons appear especially sensitive to stress and to loss of the p11 protein. While these experiments were done in animals, they echo lipid changes seen in people with depression and indicate that restoring healthy phospholipid balance in vulnerable brain regions could become a future avenue for new treatments.
Citation: Camargo, A., Kaya, I., Sturchio, A. et al. Spatial lipidomic and neuron-specific transcriptomic signatures in the nucleus accumbens reveal phospholipid dyshomeostasis in depression-related maladaptations. Transl Psychiatry 16, 243 (2026). https://doi.org/10.1038/s41398-026-04063-w
Keywords: depression, nucleus accumbens, brain lipids, phosphatidylethanolamine, stress