Clear Sky Science · en
Bulk and single-cell transcriptomic brain data identify overlapping processes and cell-types with human AUD and mammalian models of alcohol use
Why this research matters to everyday life
Alcohol use disorder is not just about willpower; it reflects deep changes in the brain. Yet most of what we know about these changes comes either from people with long histories of heavy drinking or from laboratory animals. This study asks a crucial question with real-world impact: do the brain changes seen in animal models of heavy and binge drinking actually resemble those in humans with alcohol use disorder? By comparing brains across species at the level of gene activity and cell types, the researchers aim to identify shared biological weak spots that might one day guide better treatments.
Looking inside brains across species
To explore this, the team analyzed brain tissue from humans who had alcohol use disorder, alongside data from monkeys and mice exposed to different drinking patterns. They focused on three brain areas deeply involved in motivation, reward, and stress: the prefrontal cortex, which supports decision-making and self-control; the nucleus accumbens, a key reward hub; and the central amygdala, which helps process fear and negative feelings. In each species, they measured which genes were turned up or down and estimated the mix of cell types present, allowing a detailed comparison of brain biology across humans and laboratory models.
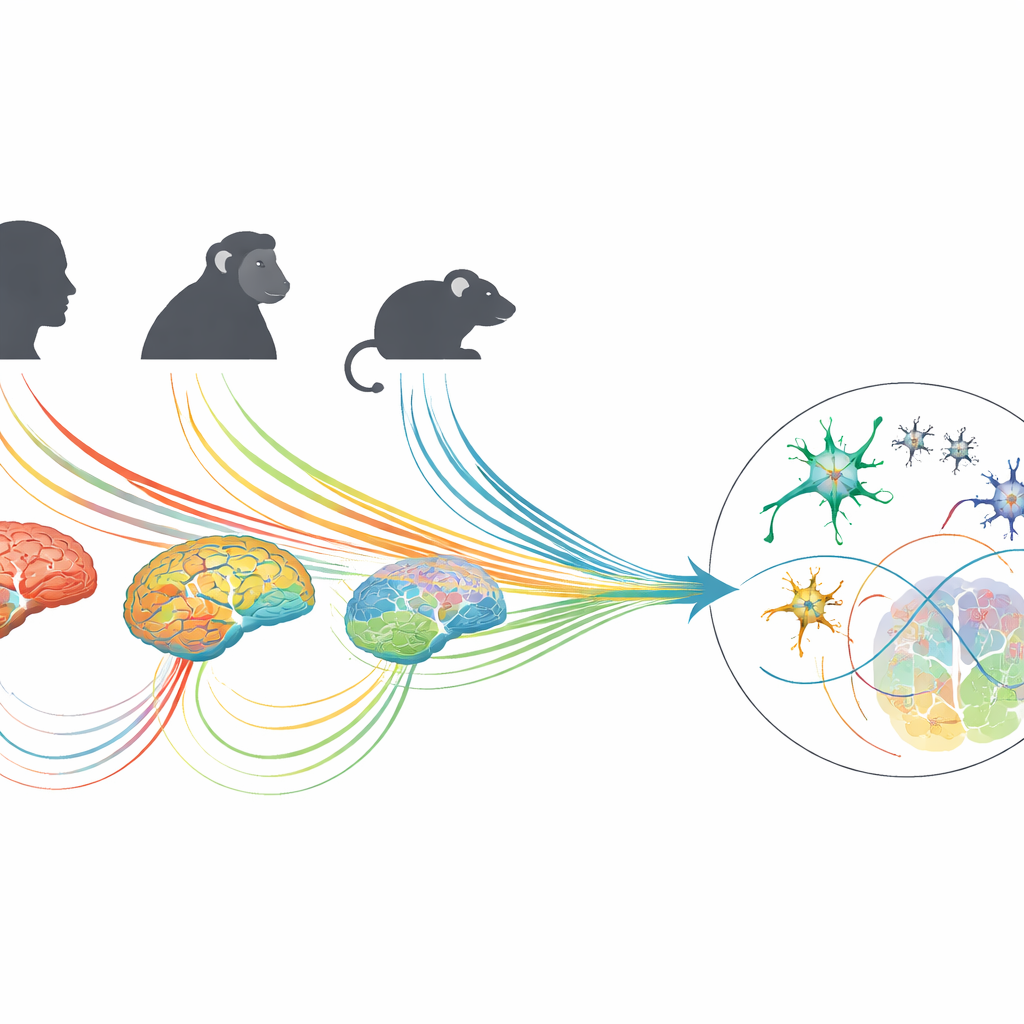
Finding shared brain signatures of heavy drinking
The researchers discovered that patterns of gene activity in human alcohol use disorder significantly tracked with those from animal models, but the strength of the match depended on species, brain region, and how alcohol was given. Overall, monkeys showed the closest resemblance to humans, especially in models where the animals could voluntarily drink large amounts of alcohol, including binge-like drinking. Certain monkey binge-drinking patterns produced brain signatures that were roughly 40% as similar to human alcohol use disorder as two independent human datasets were to each other. Mouse models of binge drinking also showed notable overlap, while some chronic exposure models, particularly those relying on forced alcohol vapor, sometimes showed opposite patterns in specific brain regions such as the prefrontal cortex.
Changes in key brain cells
Beyond genes, the study examined which brain cell types were more or less common in people with alcohol use disorder and in heavy-drinking animals. A consistent finding across species and brain regions was a drop in oligodendrocytes and their immature precursors—cells that build and maintain the insulating myelin sheaths that help nerve signals travel quickly and efficiently. In the human prefrontal cortex and nucleus accumbens, individuals with alcohol use disorder had fewer of these cells than people without the disorder, and similar decreases were seen in binge-drinking mice and alcohol-exposed monkeys. The team also observed signs that certain reward-related neurons in the nucleus accumbens, particularly those that respond to one form of dopamine signaling, tended to be reduced in humans and heavy-drinking mice, hinting at a common disturbance in circuits that govern motivation and control.
Networks that link biology to behavior
Gene activity does not change in isolation, so the researchers grouped genes into “networks” that tend to rise and fall together. They then asked which of these networks were preserved across humans, monkeys, and mice and were tied to alcohol use. They identified several such shared networks in all three brain regions. These clusters of genes were strongly tied to processes like immune signaling in the brain, myelin formation, and the ability of synapses to strengthen or weaken with experience. When the team looked at large genetic studies in humans, they found that variants near these network genes together explained about one-fifth of the inherited risk for drinking amounts and problematic alcohol use—far more than would be expected by chance, and not seen for unrelated traits such as wearing glasses. Many “hub” genes sitting at the center of these networks were also linked, in humans or mice, to traits like impulsivity, motivation, and the tendency to use alcohol or other drugs.
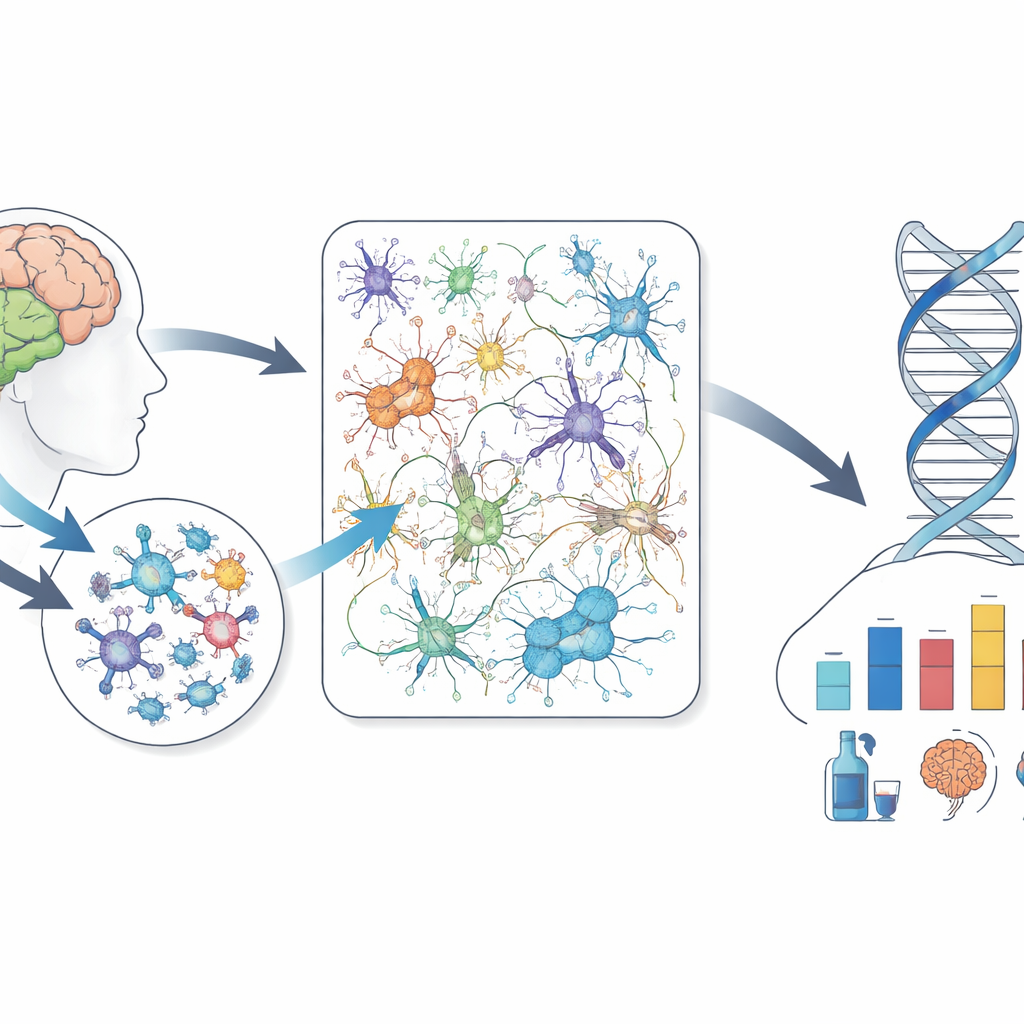
What this means for understanding addiction
For non-specialists, the takeaway is that long-term and binge drinking leave a partly shared biological footprint in the brains of people, monkeys, and mice. This footprint includes damage or loss of myelin-forming cells, shifts in reward-related neurons, and coordinated changes in gene networks that handle inflammation and brain wiring. While the details vary by species and drinking pattern, and the data have limits in sample size and diversity, the cross-species similarities lend confidence that certain animal models do capture key aspects of human alcohol use disorder. Mapping these conserved brain changes brings scientists closer to understanding why some people are more vulnerable to problem drinking and may help focus future prevention and treatment efforts on the most biologically relevant pathways.
Citation: Huggett, S.B., Selveraj, S., McGeary, J.E. et al. Bulk and single-cell transcriptomic brain data identify overlapping processes and cell-types with human AUD and mammalian models of alcohol use. Transl Psychiatry 16, 212 (2026). https://doi.org/10.1038/s41398-026-03919-5
Keywords: alcohol use disorder, addiction neuroscience, brain gene expression, animal models, oligodendrocytes