Clear Sky Science · en
Human papillomavirus16 E7 enhances cell stemness by regulating the APC2/SPIN4/β-catenin axis in cervical cancer
Why this research matters
Cervical cancer remains a major cause of death for women worldwide, especially where screening and vaccination are limited. Scientists know that certain strains of human papillomavirus (HPV), particularly HPV16, are key drivers of this disease, but the detailed steps by which the virus keeps tumors aggressive and prone to relapse are still being uncovered. This study digs into how one HPV protein, called E7, helps maintain a small but powerful population of "cancer stem" cells that can fuel tumor growth, spread, and treatment resistance—offering clues to new, more precise therapies.
From common virus to dangerous cancer
Most HPV infections clear on their own, but persistent infection with high-risk types can transform healthy cells of the cervix into cancer. The viral E7 protein is central to this process: it disables key safeguards that normally keep cell division under control. Previous work from this research group showed that HPV16 E7 boosts stem-like properties in cervical cancer cells, making them harder to eradicate. In the current study, the team set out to map the gene networks controlled by E7 in order to understand exactly how it keeps these dangerous cells alive and thriving.
Tracking E7’s impact inside tumor cells
To follow E7’s influence, the researchers used cervical cancer cell lines grown in the lab and reduced E7 levels with small interfering RNA, a tool that temporarily silences specific genes. They then measured which genes changed their activity across the entire genome. They combined this with a battery of functional tests: how fast the cells grew, how well they migrated and invaded through artificial membranes, and how readily they formed spherical clusters in culture—a hallmark of cancer stem cells. They also examined tumors grown in mice, and analyzed large public cancer datasets to see how their findings matched patterns in patient samples.
A surprising role for a known pathway
The gene-mapping experiments pointed strongly to the Wnt/β-catenin pathway, a well-known controller of normal stem cells and many cancers. When E7 was silenced, levels of β-catenin fell, suggesting this pathway was being switched off. At the same time, the team noticed big changes in a gene called APC2. In many other cancers, APC-related proteins act as brakes on Wnt signaling, helping to keep cell growth in check. Here, however, cervical cancer cells seemed to be using APC2 in an unexpected way: when APC2 was reduced, the cancer cells grew more slowly, were less able to move and invade, and lost stem-like features. In mice, tumors with long-term APC2 silencing were much smaller, yet the animals stayed healthy, hinting at a potentially safe therapeutic window.
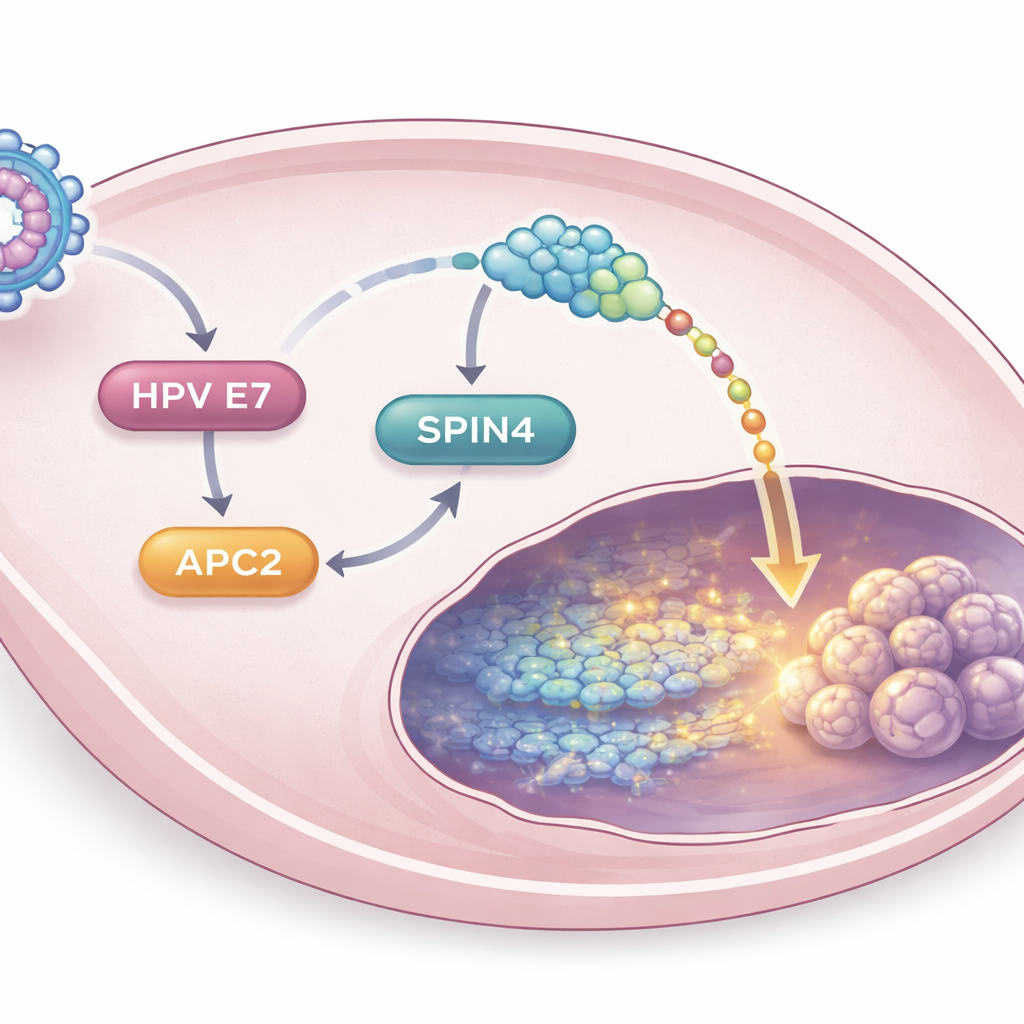
Uncovering a new cancer-promoting chain
Diving deeper, the scientists showed that HPV16 E7 increases APC2 levels indirectly through another protein called E2F1, which helps switch on sets of growth-related genes. Once elevated, APC2 in turn boosts the activity of a lesser-known protein called SPIN4. Analysis of human tumor data revealed that SPIN4 is more abundant in cervical cancers than in normal tissue, and higher levels are linked to poorer survival. In the lab, turning off SPIN4 blunted the ability of APC2 to drive cell growth, invasion, Wnt/β-catenin activity, and cancer stem-like behavior. In mouse experiments, tumors lacking SPIN4 were smaller and showed reduced markers associated with stemness. Together, these findings outline a stepwise chain—HPV16 E7 to APC2 to SPIN4 to Wnt/β-catenin—that collectively keeps cervical cancer cells aggressive and stem-like.
Looking ahead to smarter treatments
For non-specialists, the key message is that this study identifies a previously unrecognized wiring diagram inside HPV-driven cervical cancers. Rather than acting as a simple on–off switch, the viral E7 protein hijacks a specific series of host proteins—APC2 and SPIN4—to keep a dangerous pool of cancer stem cells alive through the Wnt/β-catenin pathway. This axis not only helps explain why some tumors are so persistent but also highlights several new targets that drugs or combination therapies could aim at, especially in patients whose cancers resist standard treatment. By focusing on the virus-shaped circuitry that sustains tumor "seed" cells, future therapies may become more effective at preventing regrowth and improving long-term outcomes.
Citation: Shen, T., Ma, Y., Wu, T. et al. Human papillomavirus16 E7 enhances cell stemness by regulating the APC2/SPIN4/β-catenin axis in cervical cancer. Oncogenesis 15, 10 (2026). https://doi.org/10.1038/s41389-026-00602-8
Keywords: cervical cancer, human papillomavirus, cancer stem cells, Wnt beta-catenin pathway, oncogenic signaling