Clear Sky Science · en
A synergistic CoO/MXene heterostructure anode with facilitated interfacial charge transfer for high-rate micro lithium-ion batteries
Powering Tiny Gadgets
From smart contact lenses to dust sized sensors, our smallest gadgets all face the same big problem: how to squeeze a strong, long lasting battery into a space no wider than a grain of rice. This study explores a new way to build the heart of such micro batteries so they can store more energy, charge faster, and survive thousands of charge discharge cycles without falling apart.
Why Shrinking Batteries Is Hard
Micro lithium ion batteries are shrunken versions of the batteries in phones and electric cars. When they are squeezed onto a chip or a thin flexible strip, there is little room left for the materials that actually store energy. At the same time, fast charging sends ions and electrons rushing through the battery, which can crack and crumble fragile materials. The result is a frustrating trade off between size, capacity, and lifetime that limits what tiny devices can do.

Building a Better Battery Skeleton
The team focused on the battery anode, the side that soaks up lithium during charging. They chose cobalt oxide, a material that can hold a lot of lithium but usually conducts electricity poorly and breaks down with use. To fix this, they anchored cobalt oxide nanoparticles like beads onto flat sheets of a two dimensional material called MXene. These sheets act as a strong, conductive scaffold, spreading out the tiny particles, shortening the path for ions, and giving the whole structure room to breathe as it swells and shrinks.
How the New Anode Behaves Inside
Careful imaging confirmed that the cobalt oxide forms a dense but even layer of nanosized particles on the MXene sheets, creating what the authors call a 0D 2D structure. Tests of the surface area and pore structure showed plenty of open channels where lithium ions can slip in and out. Chemical measurements revealed that the two materials touch each other closely without forming new stiff bonds, so their interface is held together mainly by gentle van der Waals forces. This soft contact helps the layers slide slightly and relieves mechanical stress during charging.
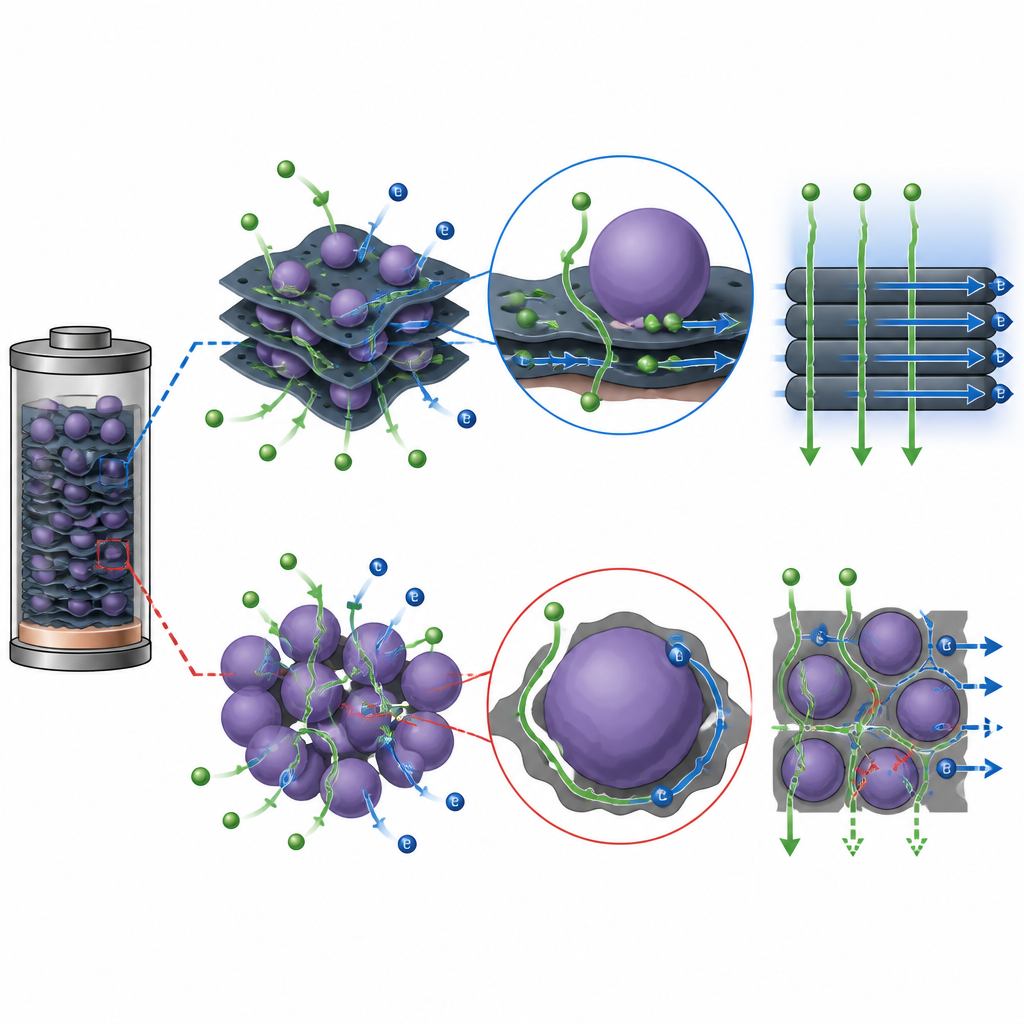
Fast Charging and Long Life Performance
In lab coin cells, the new anode stored more charge and held it for far more cycles than cobalt oxide alone or MXene alone. Even when charged and discharged quickly, it kept a high capacity and recovered strongly when the current was reduced, showing that its structure remained intact. Electrical tests showed lower resistance at the interface, meaning faster charge transfer. Computer simulations backed up these findings: they indicated more available electronic states for conduction, stronger attraction between lithium and the surface, and a lower energy barrier for lithium to diffuse, all of which point to quicker and more efficient ion motion.
From Lab Cell to Flexible Power Source
To show that the material can work in real devices, the researchers printed a flexible full battery using their anode paired with a common lithium iron phosphate cathode. Shaped like interlocking fingers, the electrodes shorten ion travel routes while fitting into a small footprint. The printed battery delivered strong areal capacity at high currents and retained most of its capacity after many cycles. In a simple demonstration, the thin flexible cell powered a digital clock, hinting at its use in wearables and other bendable electronics.
What This Means for Everyday Tech
For non specialists, the takeaway is that the authors found a clever way to stack tiny oxide particles on a flat, conductive skeleton so they can store more energy without tearing themselves apart. By improving how charges move at the interface and how the material copes with repeated swelling, they created a micro battery anode that is both powerful and durable. While more work is needed to scale up fabrication and pair it with solid electrolytes, this design offers a realistic route toward longer lasting, faster charging power sources for the next generation of miniature and wearable devices.
Citation: Hu, B., Wei, H., Zhou, H. et al. A synergistic CoO/MXene heterostructure anode with facilitated interfacial charge transfer for high-rate micro lithium-ion batteries. Microsyst Nanoeng 12, 172 (2026). https://doi.org/10.1038/s41378-026-01246-9
Keywords: micro lithium ion batteries, MXene anode, cobalt oxide nanoparticles, high rate energy storage, flexible microbattery