Clear Sky Science · en
Microfluidic device to study spatial and temporal response of astrocyte networks in response to changes in the biochemical milieu
Why brain support cells matter
When we think about the brain, we usually picture neurons firing electrical signals. But another cell type, astrocytes, quietly keeps those neurons alive and working. This study shows how brief bursts of chemical stress—like the kind caused by oxidative molecules or alcohol—can leave long-lasting scars on networks of astrocytes. Using a custom miniature device, the researchers watched how three-dimensional webs of these cells grow, break down, and struggle to recover after short insults that do not kill most of the cells outright.
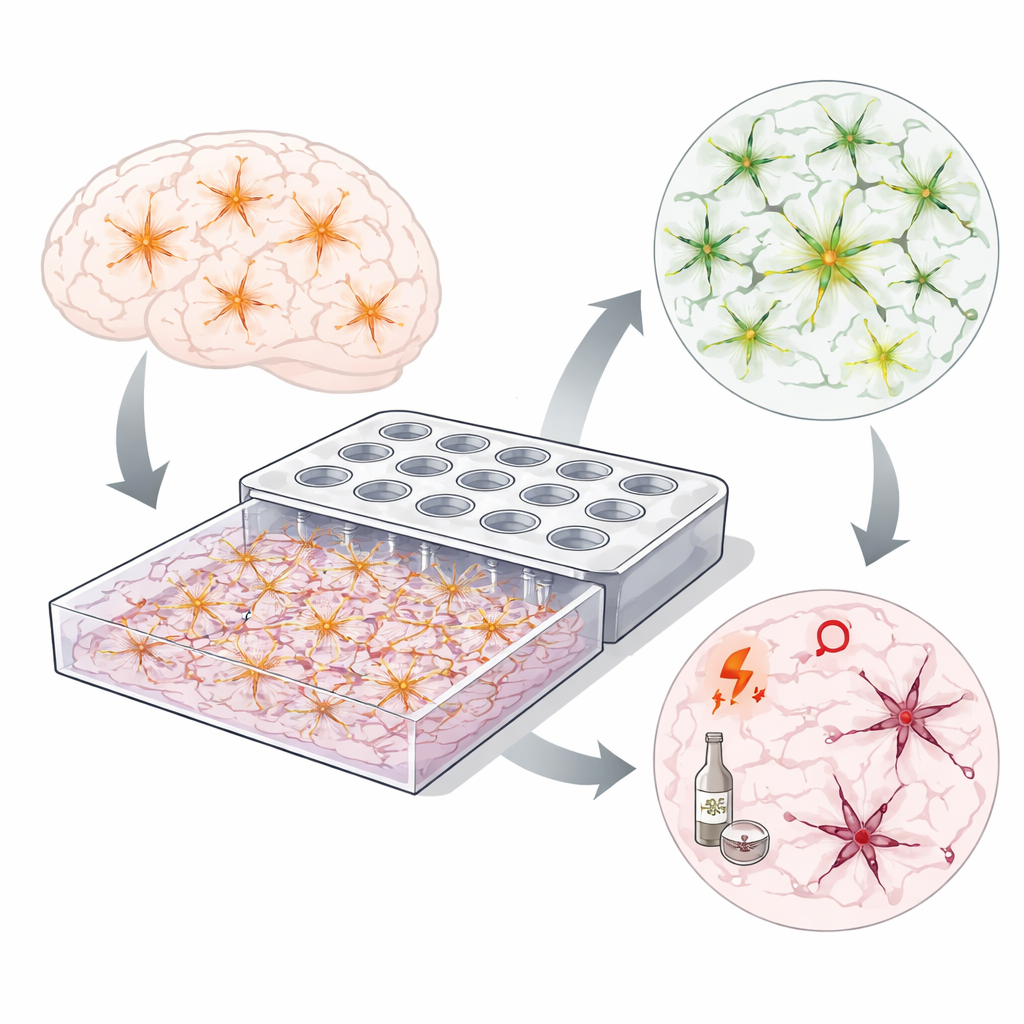
Building tiny brain-like neighborhoods
To move beyond flat petri dishes, the team engineered a microfluidic device carved into the bottom of a standard 384-well plate. Beneath the wells lies a shallow chamber that can be filled with a soft gel and living cells. Liquid added to the wells seeps through small openings, creating controlled chemical gradients across the gel. This setup lets scientists expose only part of the cell network to a substance or bathe the entire chamber, while still using familiar lab equipment such as microscopes and plate readers.
Letting astrocytes weave their web
Astrocytes in the brain form a three-dimensional mesh, reaching out long, branching processes to contact neighbors. The researchers found that the choice of surrounding gel strongly influenced whether such a mesh formed in their device. In a simple collagen gel, astrocytes stayed more rounded and less connected. In a richer basement membrane gel mimicking brain tissue, they rapidly extended numerous processes and, within about two days, built an intricate, highly interconnected network. Image analysis confirmed many more branches, junctions, and closed loops in this environment—hallmarks of a robust cellular web.
Short insults, long-lasting damage
Next, the team asked what happens when this budding or established network experiences a brief chemical shock. They exposed astrocytes to hydrogen peroxide (a source of reactive oxygen species) for just two hours, or to ethanol for 24 hours, either across the whole chamber or only under one well to create a local “hot spot” of stress. Surprisingly, most cells survived: viability generally remained above 80 percent even days later. Yet the networks themselves fared poorly. After exposure, astrocytes failed to form or maintain long branches, leaving many cells isolated instead of linked. Local hydrogen peroxide exposure mainly damaged the nearby region, while ethanol’s volatility spread its effects more broadly.
Inside the cells: power plants and signals falter
To understand why the networks collapsed despite limited cell death, the researchers zoomed in on mitochondria, the cell’s energy producers, and on calcium signals, a key way astrocytes “talk.” In healthy networks, mitochondria formed long, filament-like structures that reached into astrocyte processes, and calcium levels rose and fell in frequent pulses throughout cells. After oxidative stress or alcohol exposure, mitochondria rapidly fragmented into small, round units that retreated into the cell body, and overall mitochondrial content in the branches shrank. At the same time, calcium pulses became rarer, weaker, and slower—or disappeared almost entirely with ethanol. These changes persisted long after the chemicals were washed away, indicating a lasting disruption of cell metabolism and communication rather than a brief, reversible shock.
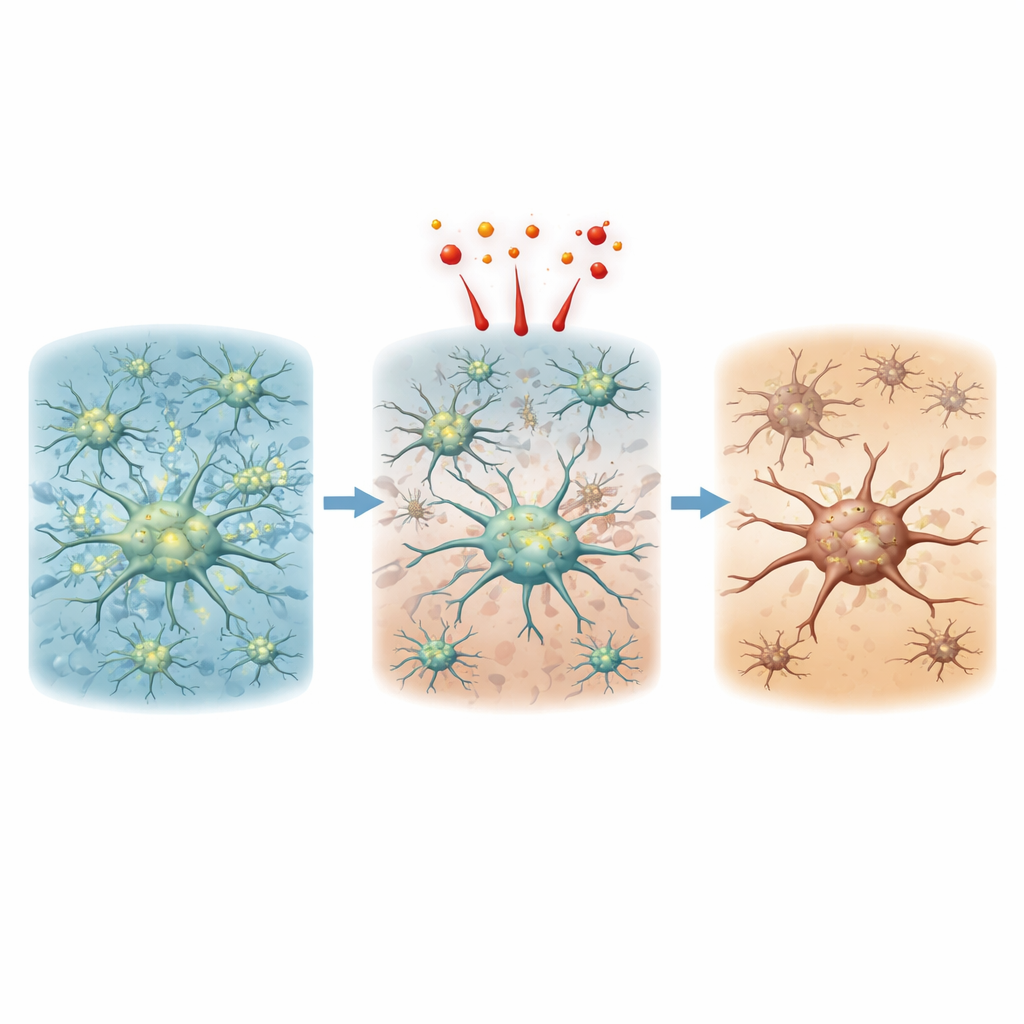
What this means for brain health
This work reveals that astrocyte networks can look deceptively intact if we only count living cells. Short, sublethal episodes of oxidative stress or alcohol exposure can quietly dismantle the fine-grained web of connections, fragment mitochondria, and silence calcium-based communication for days. Because astrocytes help control blood flow, protect neurons, and maintain the brain’s chemical balance, such hidden damage may contribute to long-term problems seen in aging, stroke, neurodegenerative disease, and heavy drinking. The new device offers a powerful way to study how brief insults ripple through brain-like tissue over space and time, and may help researchers test therapies aimed at preserving or restoring the brain’s support network before irreversible harm occurs.
Citation: Reed-McBain, C.A., Anchan, A.S., Patel, J.D. et al. Microfluidic device to study spatial and temporal response of astrocyte networks in response to changes in the biochemical milieu. Microsyst Nanoeng 12, 150 (2026). https://doi.org/10.1038/s41378-026-01213-4
Keywords: astrocyte networks, oxidative stress, microfluidic models, mitochondrial dysfunction, ethanol and brain