Clear Sky Science · en
Biostable wireless sensor-integrated bioresorbable stent for real-time monitoring of vascular pressure and fractional flow reserve
Why keeping heart vessels open matters
Blocked heart arteries are a leading cause of heart attacks and death worldwide. Doctors often use tiny metal or polymer tubes called stents to prop these vessels open, but even after a stent is placed, the artery can slowly narrow again, a problem known as in-stent restenosis. Today, checking whether a stent is still working well usually means going back into the artery with catheters, dyes, and X-rays in a hospital procedure. This study explores a different idea: turning the stent itself into a long-term, dissolving "smart" device that can quietly and wirelessly track how well blood is flowing, day after day, without repeated invasive tests.
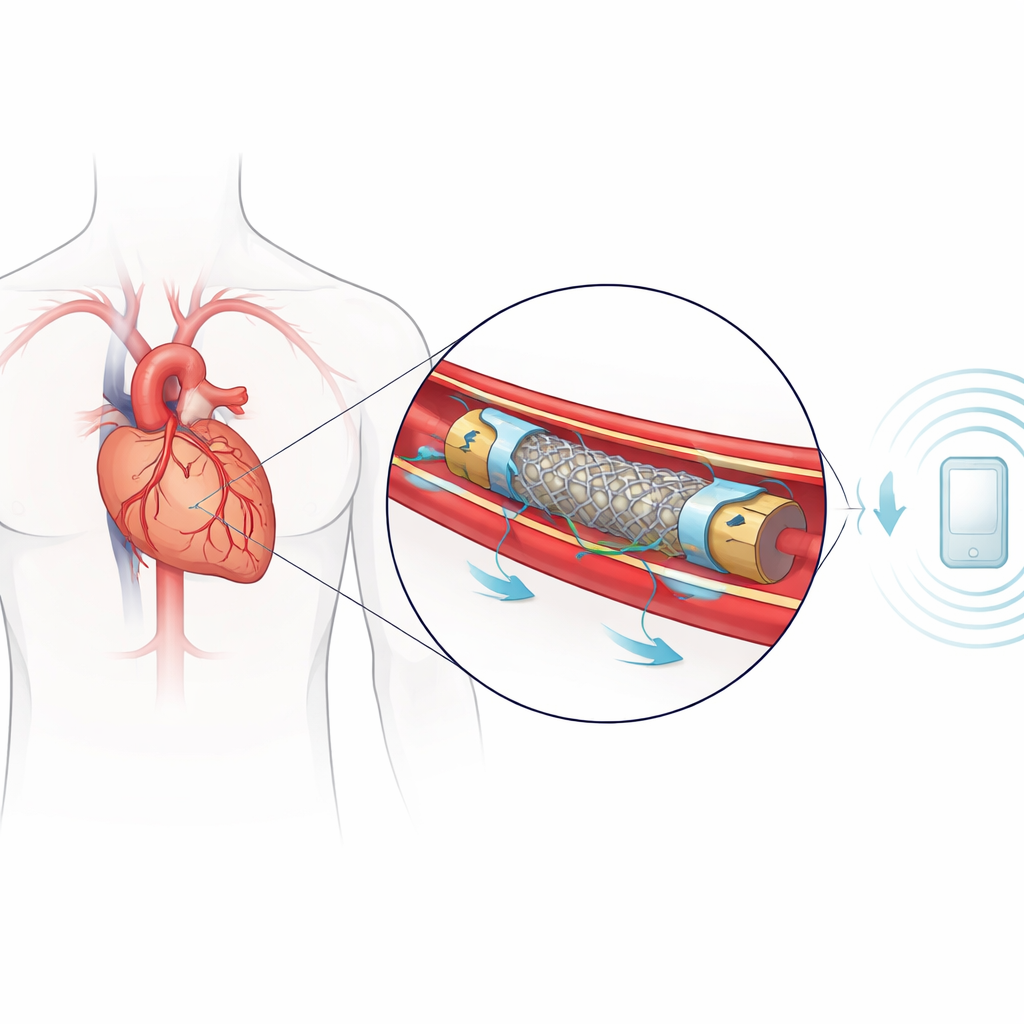
A stent that senses from the inside
The researchers designed a bioresorbable vascular stent that doubles as a pressure-sensing platform. The stent’s scaffolding is 3D-printed from two medical plastics, PLA and PCL, chosen to provide enough strength to hold the artery open while gradually breaking down in the body after healing. On this framework they mount a pair of tiny pressure sensors—one just before and one just after a narrowed segment inside the stent. These sensors work without batteries or wires: they respond to pressure changes by shifting their natural radiofrequency “ring,” which can be detected from outside the body by a small antenna. By reading these frequencies, clinicians can reconstruct the local blood pressure at both locations inside the artery.
Measuring a key sign of vessel health
The main quantity the team wants to track is called fractional flow reserve, or FFR, which compares blood pressure after a narrowing to the pressure before it. In current practice, FFR is measured by pushing a special pressure wire through the artery during a catheterization procedure. Here, the dual-sensor stent provides the same information continuously: the front sensor senses the upstream pressure, the rear sensor senses the downstream pressure, and their ratio reveals how much the narrowing is restricting flow. If restenosis develops and the downstream pressure starts to fall, the FFR value drops, signaling trouble early. Because the sensing is wireless and fully implanted, this monitoring could happen repeatedly over time without bringing the patient back for invasive tests.
Engineering a tiny stable pressure gauge
Creating such a dependable pressure sensor at millimeter scale is not trivial. Earlier versions of similar devices were prone to subtle warping during high-temperature bonding steps, which changed the spacing inside their tiny electrical components and caused their baseline frequency to drift. In the new design, the team reshaped the sensor’s internal plates, added a small air channel, and created an open window above the most delicate region. These changes allowed air to escape during bonding and reduced mechanical stress, keeping the diaphragm and coil nearly flat. Across 100 sensors, the devices showed very similar starting frequencies and a consistent, linear change in response to pressure, with fast reaction times and stable behavior over many hours in both air and saline at body temperature.
From lab bench to realistic blood flow
The researchers also had to make sure the smart stent could be delivered like a conventional one. They printed a hybrid PLA/PCL stent strong enough to support an artery, then crimped it to catheter size before attaching the sensors on the outside with a low-melting PCL connector and a temporary water-soluble film that holds everything in place. In transparent vessel models and a heart-shaped phantom, the stent expanded smoothly using a balloon catheter and hugged the vessel wall, while the sensors continued to work. In a closed-loop setup that mimicked pulsing blood, the wireless frequency signals closely matched readings from a commercial pressure transducer; pressure waves reconstructed from the stent data agreed with the reference with a correlation above 0.97 and could detect changes as small as 1 mmHg.
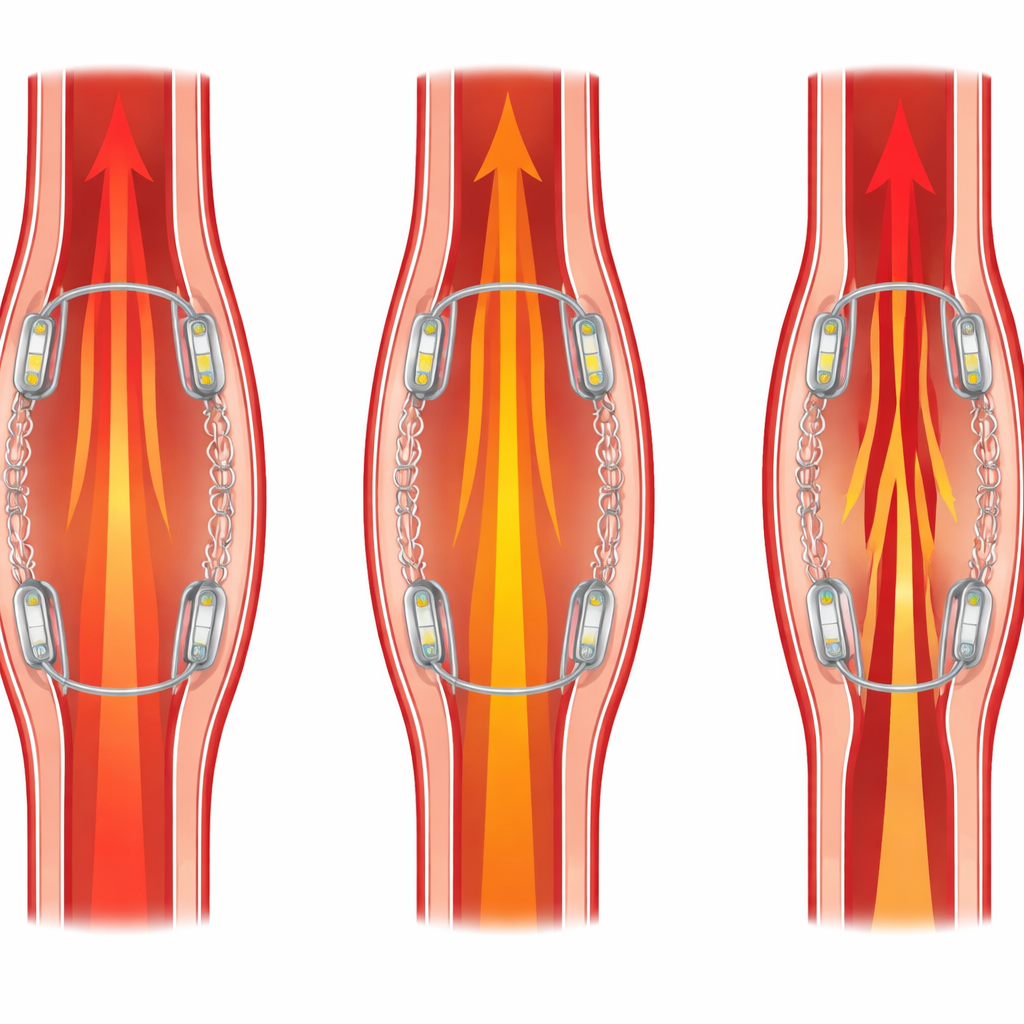
Reading narrowing severity like a clinical system
To test whether the device could truly stand in for current FFR tools, the team built a model artery with adjustable degrees of narrowing—none, mild, moderate, and severe—and placed the dual-sensor stent inside. For each condition, they recorded the wireless sensor signals, converted them to pressure, and calculated FFR, while a commercial FFR wire measured the same quantities. As the narrowing worsened, the stent sensed rising pressure before the blockage and falling pressure after it, producing FFR values that dropped in step with the commercial system and stayed within clinically acceptable differences. Even in the most severe case, where flow was strongly restricted, the smart stent’s FFR values closely tracked the reference, demonstrating that a fully implanted, dissolving scaffold can provide quantitative information usually available only during invasive procedures.
What this could mean for patients
In plain terms, this work shows that a temporary stent can be turned into a sensitive, wireless pressure gauge that lives inside the artery long enough to watch for trouble and then largely disappears. By continuously watching how much pressure is lost across a stented region, the device can warn of re-narrowing earlier and with far less inconvenience than repeated catheter tests. Although further studies in animal arteries and, eventually, humans are still needed, the concept points toward a future in which heart patients receive implants that do double duty: first mechanically propping vessels open, then silently reporting on vessel health to guide timely, less invasive care.
Citation: Wei, J., Shanmugasundaram, A., Oyunbaatar, NE. et al. Biostable wireless sensor-integrated bioresorbable stent for real-time monitoring of vascular pressure and fractional flow reserve. Microsyst Nanoeng 12, 115 (2026). https://doi.org/10.1038/s41378-026-01182-8
Keywords: smart stent, fractional flow reserve, wireless pressure sensor, bioresorbable scaffold, coronary artery disease