Clear Sky Science · en
In vitro modelling of extracellular matrix changes during skin aging: from static 2D to 3D dynamic microphysiological systems
Why Studying Skin Aging in a Dish Matters
Wrinkles, sagging, and age spots are more than cosmetic annoyances—they reflect deep changes in the architecture of our skin. Because animal tests are now heavily restricted, scientists are racing to build human-like skin models in the lab to uncover how and why skin ages, and to test safer, more effective anti-aging treatments. This article explains how researchers are moving from flat cell layers to complex 3D “mini-skins on chips” that mimic real-life aging, offering a glimpse of the future of skincare, medicine, and safety testing.
The Hidden Scaffold Beneath Our Wrinkles
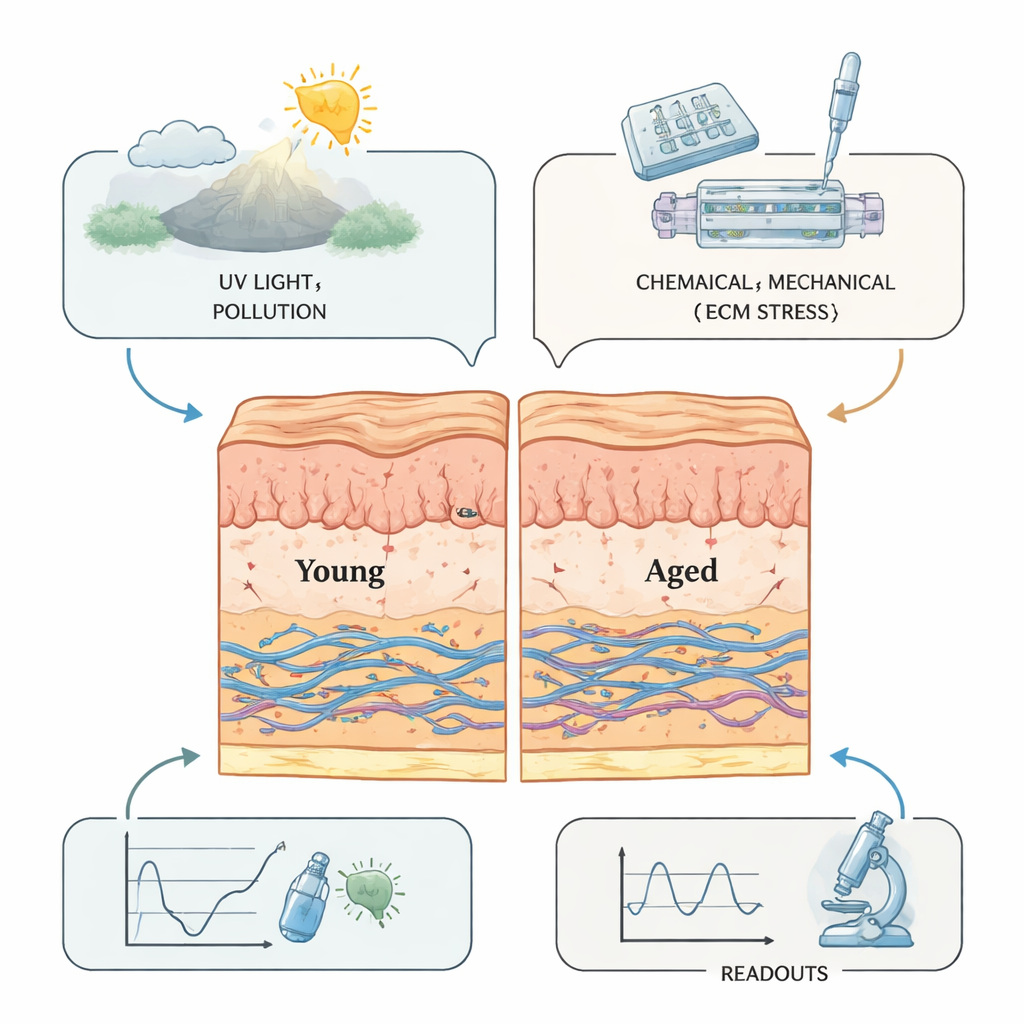
Skin’s youthful look and resilience come from the extracellular matrix, a microscopic scaffold of proteins and sugars that supports cells and connects the outer epidermis to the deeper dermis. With age and sun exposure, this framework is constantly remodeled: collagen and elastic fibers break down, sugar-related crosslinks stiffen the tissue, and the junction between the top and bottom layers flattens. These changes thin the epidermis, reduce elasticity, and encourage wrinkles and sagging. Environmental stressors like ultraviolet light, pollution, and cigarette smoke add to the damage, creating chronic low-grade inflammation—sometimes called “inflammaging.” Because this remodeling is dynamic rather than static, any convincing lab model of aging skin must capture not just which molecules are present, but how they change over time and respond to stress.
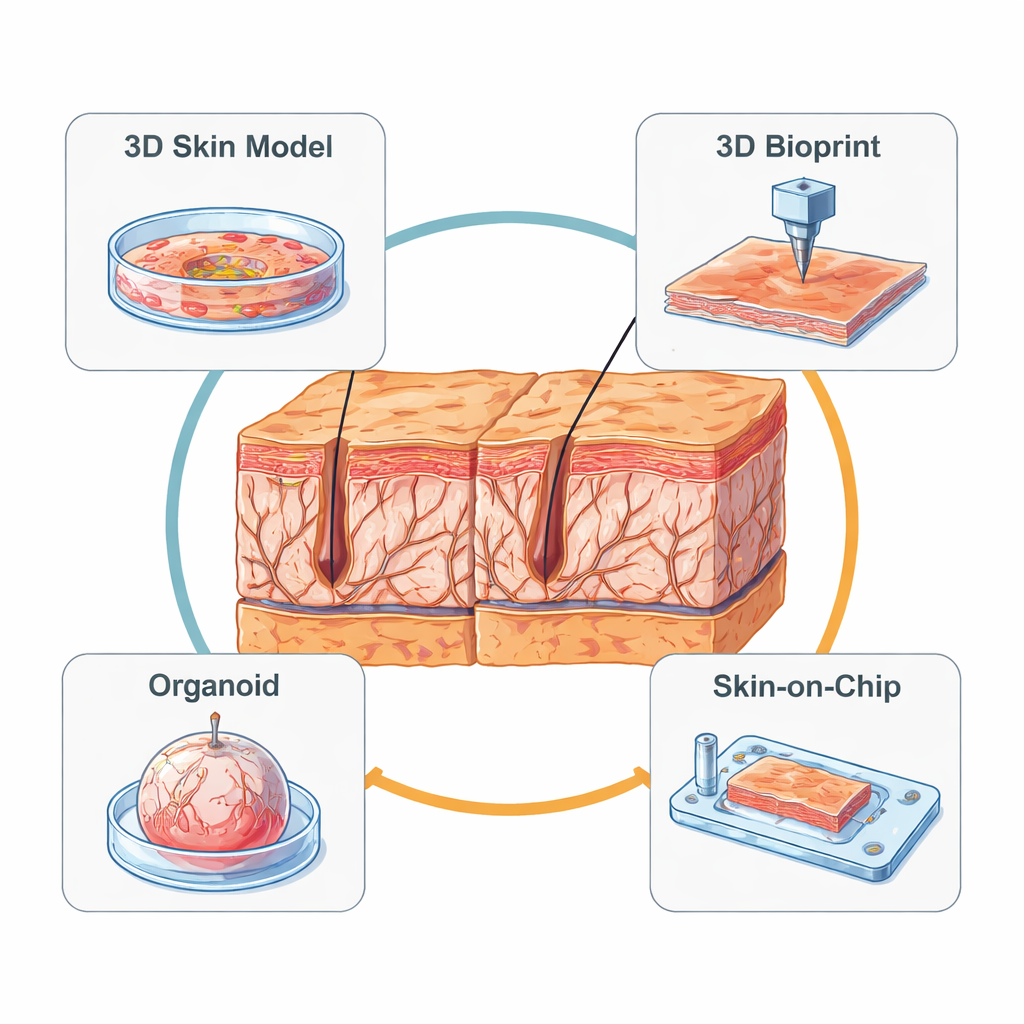
From Flat Cell Layers to 3D Mini-Skin
Early lab models relied on simple two-dimensional sheets of skin cells. These flat cultures are easy to handle and useful for measuring single markers such as collagen, elastin, or enzymes that break down the matrix. However, they miss the layered structure of real skin and cannot reproduce how cells sense and pull on a 3D scaffold. To move closer to reality, scientists developed reconstructed human skin: a 3D gel containing fibroblasts (the main matrix-producing cells) topped with a stratified epidermis grown at an air–liquid interface. These models can tan, form a barrier, and show age-like traits when researchers introduce “old” fibroblasts, expose them to UV, or chemically stiffen the matrix. Still, they lack blood vessels, immune cells, and realistic mechanical forces, and they are difficult to maintain for long enough to follow slow aging processes.
Printing, Growing, and Self-Assembling Mini-Skins
Newer approaches add engineering precision to biology. Three-dimensional bioprinting uses nozzles or light-based printers to place cells and soft “bioinks” in defined patterns, layer by layer. This lets researchers design artificial skin with controlled surface texture, including in-vitro wrinkles whose depth and spacing can be tuned and measured. Bioprinted models can also include early blood vessel structures and immune cells, making them powerful testbeds for anti-aging products and wound therapies, though the printers and materials remain expensive and technically demanding. In parallel, organoid technology starts from stem cells that self-organize into tiny, spherical skin-like structures. Remarkably, these mini-organs can form hair follicles and other appendages, and they show realistic responses to solar-like UV, including collagen loss, inflammation, and even thinning of hair shafts—effects that were hard to see in previous models.
Skin-on-a-Chip: Bringing Motion and Flow to Aging
Perhaps the most futuristic systems are “skin-on-a-chip” devices, which embed skin tissue in a transparent microfluidic cartridge. Tiny channels bring nutrients and remove waste, while built-in mechanisms gently stretch or compress the tissue to mimic facial expressions or day–night pressure cycles. By carefully tuning how strong and how frequent these forces are, scientists can make skin models develop deeper wrinkles, increased inflammatory signals, and reduced collagen—much like aging in real life. These chips can also host mini blood vessels and immune cells, allowing studies of how circulating cells enter the skin and influence aging. National and international standards are now emerging to harmonize how these devices are built and tested, paving the way for wider use in industry and regulation.
What This Means for Future Anti-Aging Solutions
Taken together, these advances point toward next-generation skin models that combine 3D structure, controlled mechanics, living microvessels, immune cells, and even microbes that normally live on our skin. Such systems can be tuned to represent “young” or “old” microenvironments and used to track how the skin’s scaffold softens, stiffens, or fragments over time under realistic sunlight, pollution, or cosmetic use. For everyday consumers, this means future anti-aging creams and treatments are more likely to be tested in human-relevant, animal-free systems that capture the true biology of aging skin, improving both safety and the chances that promised benefits will hold up in real life.
Citation: Yao, Y., Zhang, Z., Zhang, J. et al. In vitro modelling of extracellular matrix changes during skin aging: from static 2D to 3D dynamic microphysiological systems. Microsyst Nanoeng 12, 70 (2026). https://doi.org/10.1038/s41378-026-01170-y
Keywords: skin aging, extracellular matrix, 3D skin models, organoids, skin-on-a-chip