Clear Sky Science · en
Light-induced in situ reconstruction of CoOOH-modified TiO2/CoNi-LDH heterojunction photoanode: achieving excellent photoelectrochemical cathodic protection and bacterial inactivation
Why protecting metal in the ocean matters
Metal structures in the ocean, from ships to offshore platforms, constantly battle salty water and bacteria that eat away at their surfaces. This slow damage costs industry vast sums and risks serious failures. The study described here explores a sunlight powered coating that can both slow rust and kill harmful bacteria in seawater, aiming for cleaner, longer lasting marine infrastructure with lower energy use.
A smart coating that wakes up under light
The researchers built a special coating on transparent conducting glass, designed to be attached electrically to stainless steel. The base of the coating is titanium dioxide, a well known material that can use light to move electrons but normally responds mostly to ultraviolet rays. On top, they grew a thin, layered film made from cobalt and nickel compounds. Under light, this upper film does not stay fixed; instead, it reshapes itself into a closely related material that turns out to be the true workhorse of the system.

How light driven rebuilding boosts protection
When the coating is first prepared, the upper layer is a cobalt nickel layered double hydroxide. During testing in salty water under simulated sunlight, the color of the electrode changes from bluish green to brownish yellow. Detailed measurements with X ray and vibrational methods show that part of the cobalt compound converts into cobalt oxyhydroxide. This change happens in place, driven by the positive charges created when light hits the material. The new phase acts like a helper that makes it easier for these charges to move and to drive chemical reactions at the surface.
Fighting both rust and bacteria at once
The most practical question is whether this light activated surface can actually protect steel in seawater. The team connected the coated glass to 304 stainless steel in a salt solution and monitored its electrical potential under flickering light. The best version of the coating, prepared at a specific voltage, pushed the steel to a much safer, more negative potential by about 380 millivolts, an amount that compares favorably with other systems in the literature. Microscopy images show that protected steel stays smooth after a day in salty water, while unprotected steel shows clear signs of corrosion. At the same time, tests with the common marine bacterium Pseudomonas aeruginosa show that the coating can inactivate all detectable cells within two hours of light exposure, far outperforming titanium dioxide alone.
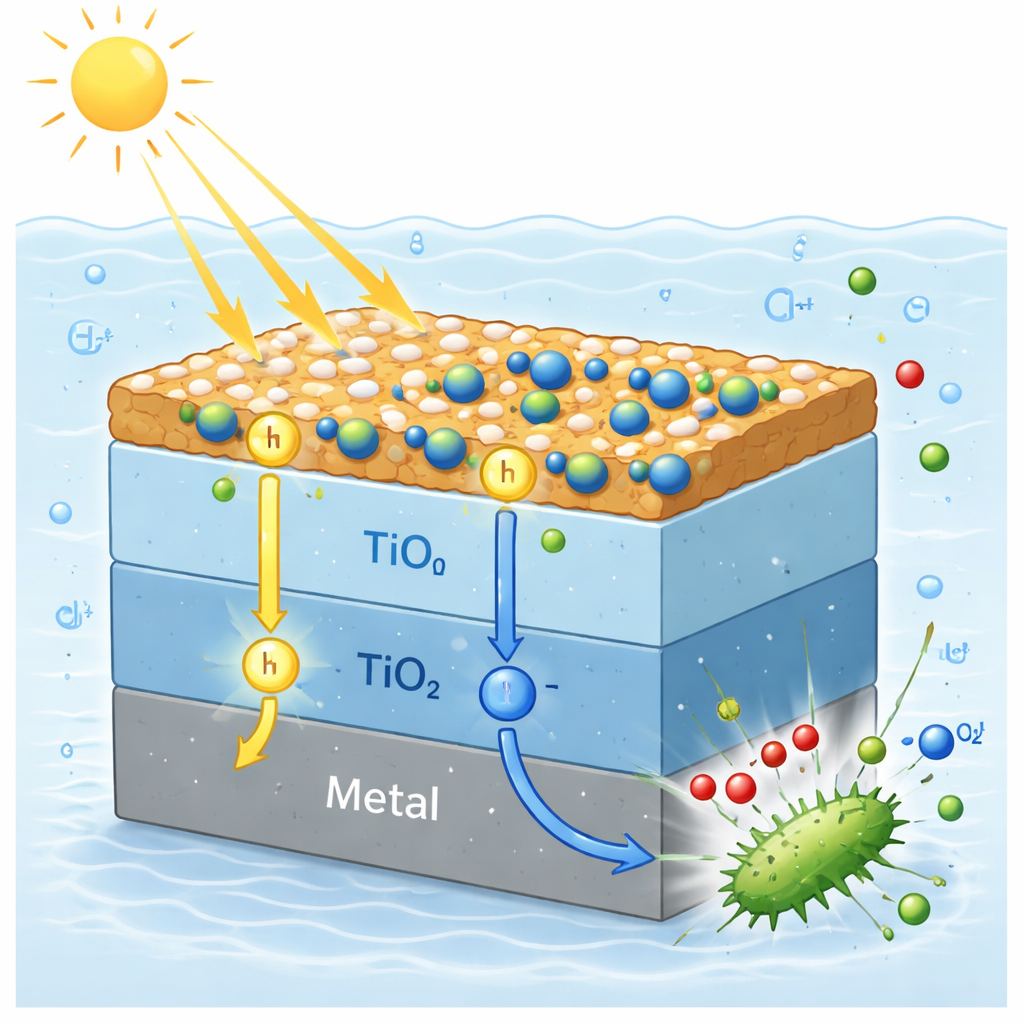
Peeking inside the charge traffic
To understand why performance improves so much, the authors probed how charges move in the coating and used computer calculations to map energy levels. They found that when the different layers touch, electrons naturally shift from titanium dioxide to the cobalt nickel layer, setting up internal electric fields. Under light, these fields guide electrons toward the titanium dioxide and on to the steel, while positive charges flow in the opposite direction toward the rebuilt cobalt oxyhydroxide. This separation keeps charges apart longer so they can either travel to the metal to prevent rust or react with water and oxygen to form highly reactive oxygen species that damage bacterial membranes and DNA.
What this means for cleaner, longer lasting structures
In simple terms, this study shows that a carefully stacked, light responsive coating can shield steel in salty water while also sterilizing its surface. The key is that the upper layer quietly rebuilds itself under light into a more active form, which helps move charges efficiently and generate reactive molecules. Together these effects slow down corrosion and wipe out harmful bacteria, hinting at future marine materials that use sunlight, rather than extra electricity or toxic chemicals, to stay strong and clean over long periods.
Citation: Wang, M., Tang, Y., Liu, J. et al. Light-induced in situ reconstruction of CoOOH-modified TiO2/CoNi-LDH heterojunction photoanode: achieving excellent photoelectrochemical cathodic protection and bacterial inactivation. Light Sci Appl 15, 230 (2026). https://doi.org/10.1038/s41377-026-02328-z
Keywords: marine corrosion, photoelectrochemical protection, titanium dioxide coating, reactive oxygen species, antibacterial surfaces