Clear Sky Science · en
Energy extraction from dark Fe3+ in A2Sc2B4O11:Fe3+, Yb3+ (A = Sr, Ba) toward promoted NIR luminescence and pc-LED light source for multifunctional applications
Invisible light with everyday uses
Many technologies we rely on, from night-vision cameras to food scanners in supermarkets, depend on light that our eyes cannot see: near‑infrared (NIR) light. This study reports a new class of environmentally friendly materials that convert harmless ultraviolet light from standard LED chips into powerful NIR light. Because these materials avoid toxic elements and work efficiently even at high temperatures, they could help build safer, cheaper and more versatile infrared lamps for medicine, food safety, and industrial sensing. 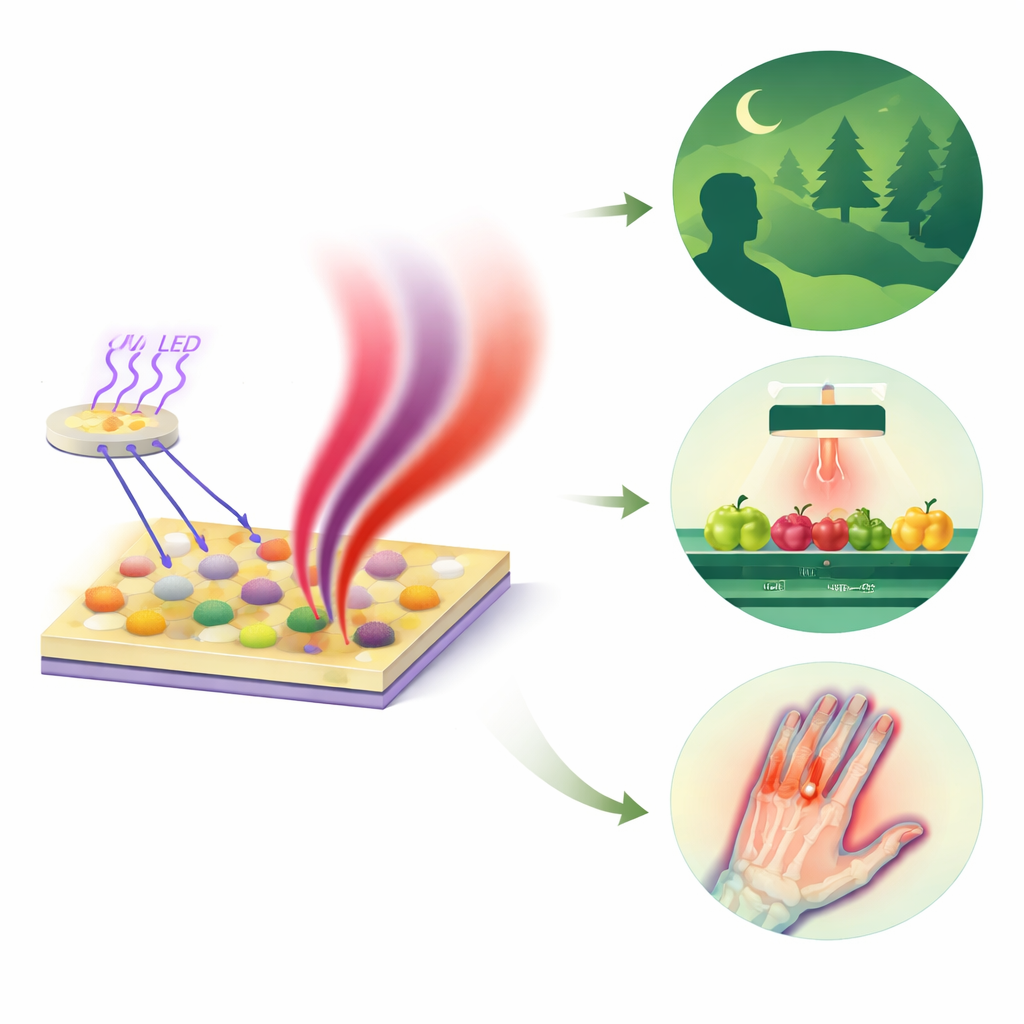
Why near‑infrared light matters
NIR light, which lies just beyond the red end of the rainbow, penetrates deeper into many materials than visible light and is strongly absorbed by specific chemical bonds in water, fats and other organic molecules. That makes it ideal for tasks such as seeing through fog, checking whether fruit is ripe without cutting it open, imaging blood vessels beneath the skin, or analyzing the composition of liquids. Today, many NIR light sources rely on hot filaments or lasers, which can be bulky, inefficient or expensive. Phosphor‑converted LEDs offer a compact alternative: a standard LED chip pumps a special powder (the phosphor), which then re‑emits light at longer wavelengths. But existing NIR phosphors often use chromium, a metal that can oxidize into a highly toxic form, or rare‑earth ions that absorb light too weakly for many real‑world devices.
Turning iron from light thief to light maker
Iron in the Fe³⁺ state is abundant and essential for life, but in many optical materials it acts as a “light quencher,” robbing nearby emitters of their glow. The team behind this work flips that role. They design a crystalline host made of strontium or barium, scandium, boron and oxygen (written as A₂Sc₂B₄O₁₁, with A = Sr or Ba) into which they deliberately introduce small amounts of Fe³⁺. In this host, iron ions sit in tightly packed octahedral pockets surrounded by oxygen. Under near‑ultraviolet light from common LED chips (around 355–370 nanometers), oxygen‑to‑iron charge‑transfer transitions allow the Fe³⁺ ions to absorb light strongly. The iron then emits very broad NIR light centered near 930–975 nanometers, with a spread wide enough to cover part of the so‑called NIR‑II window that is especially useful for deep biological imaging.
Boosting output with a helping ion
On their own, these iron‑based phosphors still waste much of the absorbed energy: many Fe³⁺ ions remain “dark,” passing energy to defects or losing it as heat rather than light. To fix this, the researchers introduce a second ingredient, ytterbium (Yb³⁺), into the same crystal sites. Yb³⁺ is a simple, highly efficient NIR emitter whose natural emission sits just around 1,000 nanometers. In the co‑doped material, ultraviolet light is first harvested by the iron network, then passed along to nearby Yb³⁺ ions. Importantly, the ytterbium ions physically interrupt chains of closely spaced iron ions that would otherwise shuttle energy into non‑radiative losses. This “energy extraction” from dark Fe³⁺ centers into bright Yb³⁺ centers increases the overall NIR brightness by up to about 160 times and shifts the main emission to around 1,000 nanometers, an especially useful region for sensing and imaging. 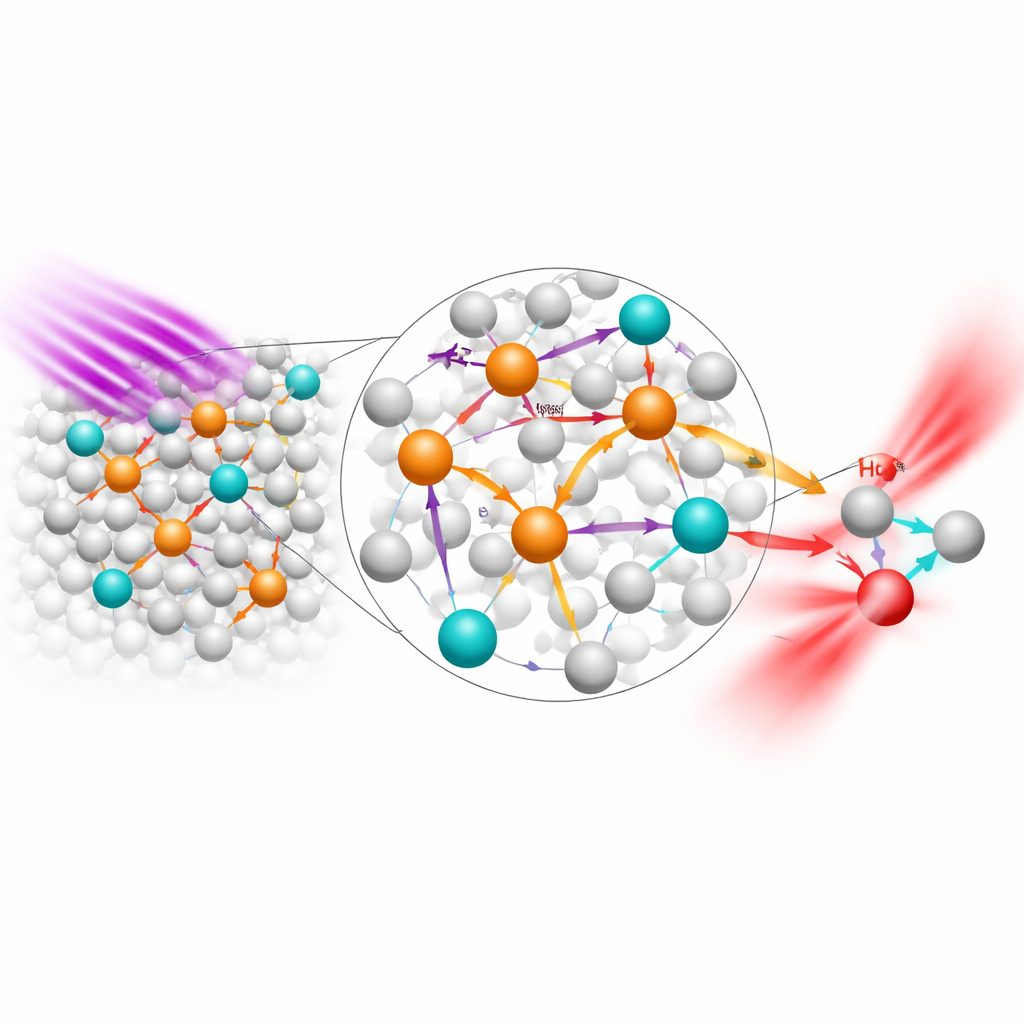
Stable light at working temperatures
For any lighting material, performance at elevated temperatures matters just as much as raw brightness, because LED packages heat up during operation. The researchers show that the iron‑only versions of their phosphor keep only about one‑third of their room‑temperature brightness at 100 °C (373 K). After adding Yb³⁺, however, the co‑doped phosphors retain more than 60 percent of their brightness at the same temperature. This improvement arises because ytterbium’s electrons are more shielded from vibrations in the crystal lattice than iron’s, making them less sensitive to heating. The way the brightness declines with temperature is smooth and predictable, allowing the same material to act as a built‑in thermometer: by measuring the intensity of NIR emission, one can infer the operating temperature of the device with a relative sensitivity of roughly 1.5 percent per kelvin near 150 °C.
From lab powder to practical devices
To demonstrate real‑world potential, the team coats their optimized Sr₂Sc₂B₄O₁₁:Fe³⁺,Yb³⁺ phosphor onto a commercial 365‑nanometer LED chip, creating a compact NIR lamp. This prototype produces a broad emission spanning roughly 850–1,150 nanometers and brightens as the drive current increases. In test scenes recorded with NIR‑sensitive cameras, the lamp reveals electronic structures hidden inside an opaque card, highlights surface defects on apples that are hard to see under visible light, and outlines blood vessels inside a human finger. It also serves as a light source for spectroscopy: by shining through mixtures of water and alcohol, the device picks up subtle changes in the NIR absorption bands associated with O–H and C–H bonds, enabling estimation of water content from changes in transmitted intensity.
What this means for future infrared lighting
In accessible terms, this work shows that a safe and abundant element—iron—can be turned from a nuisance into a powerful engine for invisible light, provided it is placed in the right crystal environment and paired cleverly with ytterbium. The resulting powders absorb light from standard ultraviolet LEDs and convert it into strong, broad NIR glow that stays bright at realistic device temperatures. With their ability to support night vision, non‑destructive food inspection, biomedical imaging and chemical analysis, these Fe³⁺/Yb³⁺ co‑doped phosphors point the way toward a new generation of compact, efficient and eco‑friendly infrared light sources.
Citation: Yu, D., Liu, H., Lv, M. et al. Energy extraction from dark Fe3+ in A2Sc2B4O11:Fe3+, Yb3+ (A = Sr, Ba) toward promoted NIR luminescence and pc-LED light source for multifunctional applications. Light Sci Appl 15, 229 (2026). https://doi.org/10.1038/s41377-026-02284-8
Keywords: near-infrared LEDs, luminescent phosphors, iron doped materials, energy transfer, spectroscopic sensing