Clear Sky Science · en
TANK-binding kinase 1 protects against MASH progression via mitochondrial quality control
Why your liver’s tiny power plants matter
Millions of people with obesity develop a fatty liver condition now called metabolic dysfunction–associated steatotic liver disease (MASLD). In some, this seemingly silent problem progresses to a dangerous, scarred form known as steatohepatitis (MASH), which can lead to cirrhosis and liver cancer. This study explores how liver cells normally keep their energy factories—mitochondria—clean and functional, and how failure of this clean‑up system helps push a fatty liver toward serious disease.
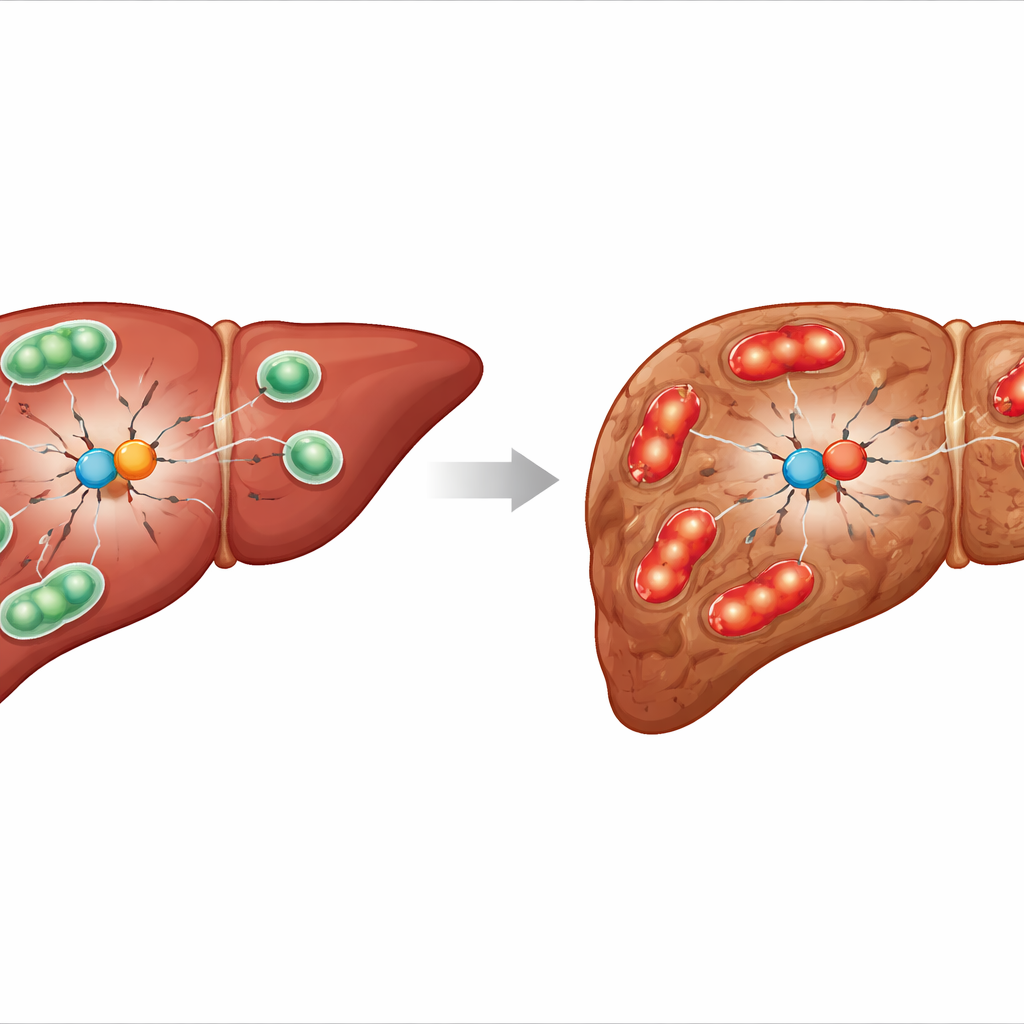
From simple fat buildup to serious liver damage
MASLD begins when liver cells are overloaded with fat. Mitochondria, which burn fat to produce energy, become overworked and start to malfunction. They swell, fragment and leak harmful reactive oxygen species, damaging the cell from within. The body tries to cope using “mitochondrial quality control,” a set of processes that replace worn‑out mitochondria with new, healthy ones. When this balance is lost, defective mitochondria accumulate, driving inflammation, cell death and ultimately the progression from simple fat accumulation to MASH. Understanding who polices this quality‑control system is crucial for finding new treatments.
A cellular traffic cop called TBK1
The researchers focused on a protein kinase named TANK‑binding kinase 1 (TBK1), already known to participate in immunity, energy use and a recycling process called autophagy. Using human liver cell lines and mice engineered to lack TBK1 only in liver cells, they found that TBK1 serves as a guardian of mitochondrial quality. When TBK1 was missing, liver cells contained more mitochondria, but these were often depolarized, produced excess reactive oxygen species and were less capable of burning fat. Under stress—such as exposure to fatty acids or chemicals that injure mitochondria—TBK1‑deficient cells were more prone to die and to switch on inflammatory genes.
How the clean‑up machinery breaks down
Closer inspection showed that the problem was not simply too many damaged mitochondria being formed, but that the cell’s clean‑up system, mitophagy, was jammed. Normally, damaged mitochondria are tagged and delivered to lysosomes, the cell’s digestive sacs, for breakdown. TBK1 helps by modifying a helper protein called p62 and by supporting lysosome activity. In cells lacking TBK1, damaged mitochondria were not efficiently delivered to lysosomes, lysosomal enzymes were less active, and telltale markers of stalled recycling piled up. At the same time, signaling through another nutrient‑sensing pathway, mTOR, was abnormally high, a state known to suppress lysosome function. TBK1 physically interacted with mTOR in liver cells, suggesting it usually helps keep this brake on digestion in check.
What goes wrong in diseased livers
The team next asked whether TBK1 fails during real‑world liver disease. In mouse models that progress from simple fatty liver to MASH‑like scarring, TBK1 activity in liver mitochondria rose early but then fell sharply as disease advanced. In a separate genetic model that develops MASH on a special diet, TBK1 activity again dropped, while mitochondrial proteins declined and markers of inflammation and fibrosis rose. Human liver samples told a similar story: people with MASH had lower TBK1 levels and activity than healthy controls, and higher liver fat was associated with less TBK1. Genes involved in lysosome function tended to track positively with TBK1, linking this protein to the health of the cell’s recycling system in patients.

Restoring protection and easing liver scarring
To test whether boosting TBK1 could help, the researchers used a viral delivery system to increase TBK1 in the livers of MASH‑prone mice. Animals receiving normal TBK1 showed more active mitophagy, fewer damaged mitochondria, and less expression of genes tied to scarring and cell death, even though overall body weight and fat mass did not change. A mutated form of TBK1 lacking kinase activity only partially improved metabolism and did not reduce fibrosis to the same extent, underscoring the importance of TBK1’s enzymatic function for protecting mitochondria and lysosomes. Electron microscopy confirmed more mitochondria being safely enclosed and degraded in treated livers, along with healthier mitochondrial shapes.
What this means for future therapies
Together, the findings paint TBK1 as a central switch that helps liver cells recognize and remove faulty mitochondria while keeping lysosomes working and harmful signaling in check. In early fatty liver, this system ramps up to cope with stress, but as disease deepens, TBK1 activity fades, mitophagy stalls and damaged mitochondria accumulate, fueling inflammation and fibrosis. Therapies that restore or mimic TBK1’s protective actions—particularly its support of mitochondrial clean‑up and lysosomal health—could offer a new way to slow or prevent the progression from simple fatty liver to life‑threatening MASH.
Citation: An, SM., Jang, J.H., Sung, J.H. et al. TANK-binding kinase 1 protects against MASH progression via mitochondrial quality control. Exp Mol Med 58, 917–931 (2026). https://doi.org/10.1038/s12276-026-01672-9
Keywords: fatty liver disease, mitochondria, autophagy, lysosomes, TBK1