Clear Sky Science · tr
Protein-protein interactions are a major source of epistasis in genetic interaction networks
Why this matters for understanding genes
When doctors or genetic tests tell us that a certain gene "causes" a disease, that sounds straightforward. In reality, the effect of any one gene often depends strongly on what other genes are doing at the same time. This paper digs into why combinations of gene changes can behave unpredictably, and shows that many of these surprises can be traced back to how the proteins encoded by those genes actually stick together inside cells. Understanding this link could sharpen our ability to predict disease risk and to find drug targets that exploit weak points in cancer and other illnesses.
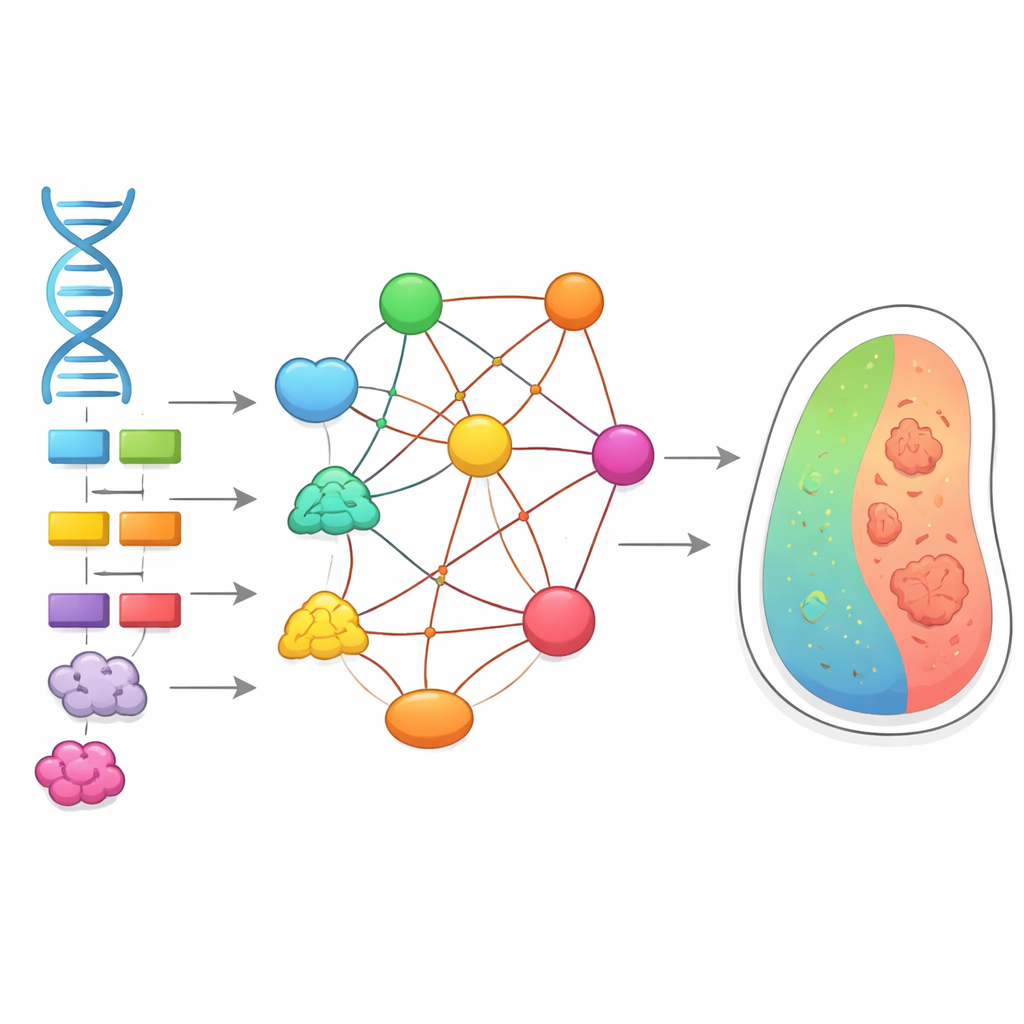
Two hidden maps inside every cell
Biologists use two powerful kinds of maps to understand how cells work. One is the genetic interaction map, which shows what happens to a cell when two genes are disrupted together: sometimes the double change is much worse than expected, sometimes milder. These non-additive effects are called epistasis and they make it hard to predict traits from DNA alone. The second map is the protein-protein interaction map, which charts which proteins physically touch one another to form complexes and pathways. Until now, these two maps were usually studied separately, and it was unclear how tightly they were connected.
How strongly proteins bind shapes gene-gene effects
The authors combined large datasets from yeast and human cells that measured both how often proteins meet and how tightly they bind. They then laid genetic interaction data on top of this physical landscape. A clear pattern emerged: gene pairs whose proteins form strong, well-balanced complexes—where both partners are present in similar amounts and bind in roughly 1-to-1 ratios—tend to show strong negative interactions when both genes are lost. In practical terms, knocking out either gene alone hurts the cell somewhat, but losing both at once is especially damaging, reflecting the central role of the shared protein complex. By contrast, weaker or more lopsided protein partnerships showed milder and more varied genetic effects.
Turning binding strengths into network-level predictions
Simply knowing which proteins touch is not enough; the key is how tight those contacts are. To capture this, the researchers used quantitative mass spectrometry to estimate binding affinities—how easily protein pairs fall apart—across thousands of interactions. They found that, as binding gets stronger, the average negative genetic interaction between the corresponding genes follows a smooth, sigmoidal curve, very similar to how a binding site fills up as concentration increases. This held for both yeast and human cells. Using this quantitative relationship, they showed that the structure of a genetic interaction network can be partly reconstructed from protein binding data alone, with clusters of related genes reappearing from the protein measurements.
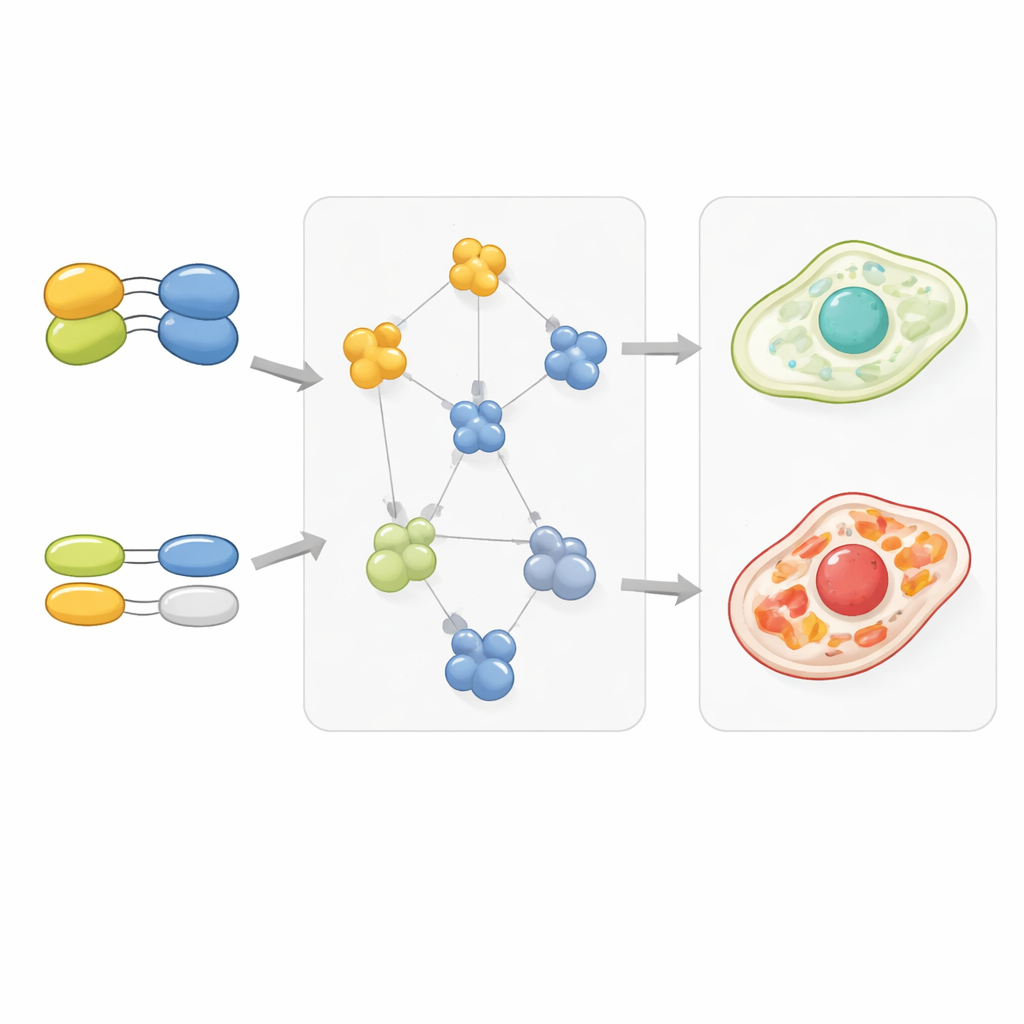
Gene duplicates and cellular wiring patterns
The study also looked at genes that have duplicates, known as paralogs, which often provide backup for one another. Gene pairs where one or both partners had duplicates tended to show weaker genetic interactions with other genes, suggesting that redundancy softens the impact of losing any single copy. Strikingly, proteins encoded by duplicated genes also tended to bind their partners more weakly, as if evolution had tuned down the stickiness to keep complex numbers balanced despite extra copies. When duplicates diverged more in sequence and function, one copy often retained strong, focused interactions while the other lost or weakened many of its contacts, a pattern that matched changes in how those genes interacted genetically with the rest of the cell.
Shared hubs that tie cellular systems together
Beyond individual pairs, the authors asked whether the large-scale shapes of the genetic and protein maps match. They found that groups of genes that cluster together in genetic interaction data often correspond closely to known protein complexes. Just as important, the same kinds of "connector" proteins frequently bridge these complexes in both maps, forming recurring module-connector-module patterns. For example, transport factors that move cargo through the nuclear pore and signaling proteins like Ras appear as shared connectors, linking distant cellular processes in both the physical and genetic networks.
What this means for genes, disease, and therapy
The central message for non-specialists is that many puzzling gene-gene effects are not mysterious after all: they arise because the proteins encoded by those genes physically depend on one another, and the strength of that dependence can be measured. By tying genetic epistasis to the chemistry of protein binding, this work moves us closer to predicting how combinations of mutations will affect cells, including in human disease. In the long run, such integrated maps may help identify combinations of genes that, when targeted together, selectively cripple cancer cells or reveal new ways to restore balance in disrupted protein networks.
Atıf: Castellanos-Girouard, X., Serohijos, A.W.R. & Michnick, S.W. Protein-protein interactions are a major source of epistasis in genetic interaction networks. Nat Commun 17, 2398 (2026). https://doi.org/10.1038/s41467-026-69152-2
Anahtar kelimeler: genetic interactions, protein networks, epistasis, protein complexes, systems biology