Clear Sky Science · en
Ultrasensitive Alzheimer’s disease biomarker detection with nanopillar photonic crystal biosensors
Why a Simple Blood Test for Memory Loss Matters
As new medicines emerge that can slow Alzheimer’s disease, doctors urgently need simple tests that reveal who is at risk long before symptoms become obvious. Today, confirming Alzheimer’s usually means expensive brain scans or spinal taps that few people receive in routine care. This study describes a tiny light-based sensor that can spot key Alzheimer’s-related molecules in a droplet of blood at clinically meaningful levels, potentially paving the way for fast, low-cost tests that could be used in everyday clinics or even near the bedside.
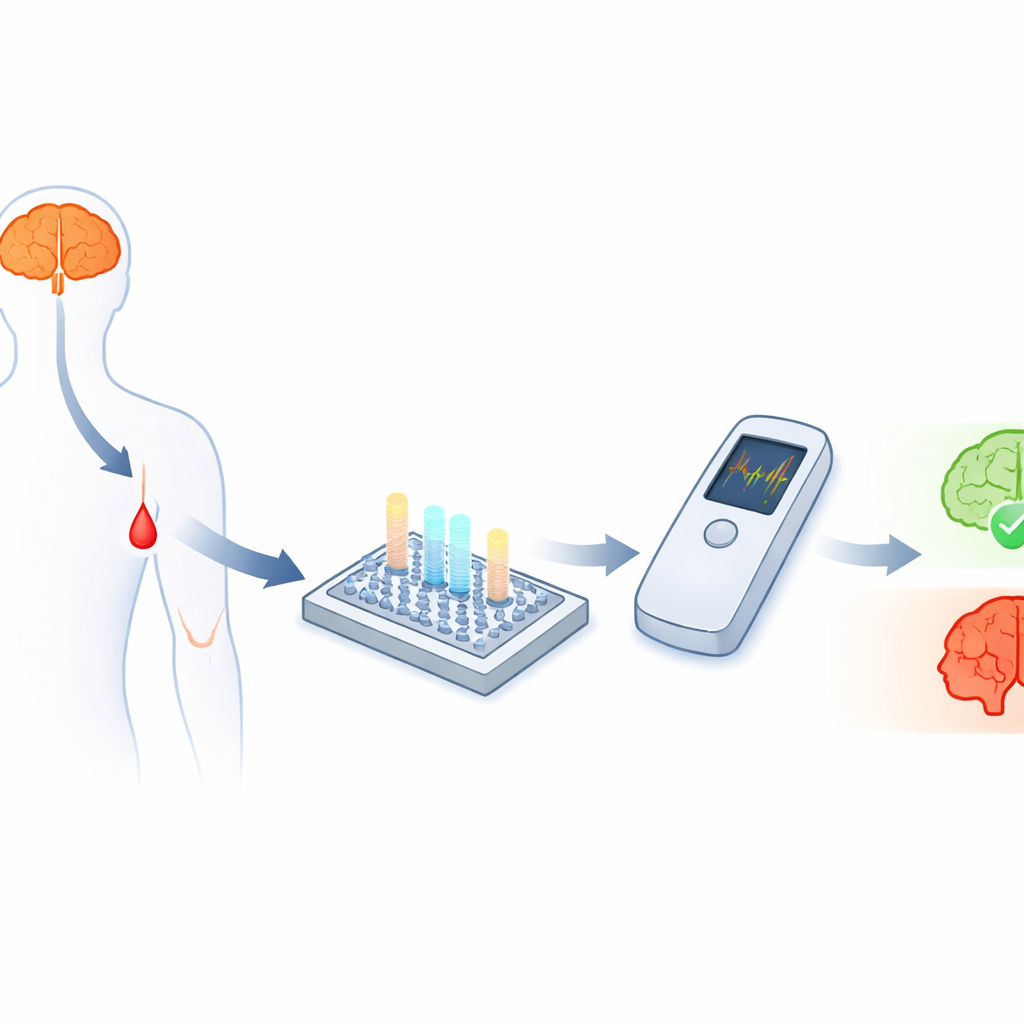
Looking for Clues in the Blood
Alzheimer’s disease is closely linked to small protein fragments called amyloid‑beta, especially two forms known as Aβ40 and Aβ42. Their amounts, and particularly their ratio, carry important clues about whether the disease is present and how it is progressing. The challenge is that these fragments are tiny and circulate in blood at extremely low levels—trillionths of a gram per milliliter—making them very hard to measure accurately outside of specialized laboratories. While electrochemical sensors can, in principle, detect such low concentrations, turning them into rugged, affordable point‑of‑care tests that can track several biomarkers at once has proven difficult.
Guiding Light Through Tiny Pillars
The team tackled this challenge using nanophotonics—structures that control light on a scale smaller than the wavelength of that light. They built a chip covered with a regular grid of paired, hair‑thin silicon pillars on glass. When light shines on this patterned surface, it becomes trapped in a special resonant mode that is extremely sensitive to changes near the pillar tops, much like how the tone of a tuning fork shifts if material is added. By carefully adjusting the distance between the pillars in each pair, the researchers balanced three crucial properties at once: how sharply defined the optical resonance is, how strongly it changes with its surroundings, and how large a signal it produces. This “sweet spot” design makes the chip especially well suited to sensing tiny amounts of material.
Making Invisible Molecules Visible
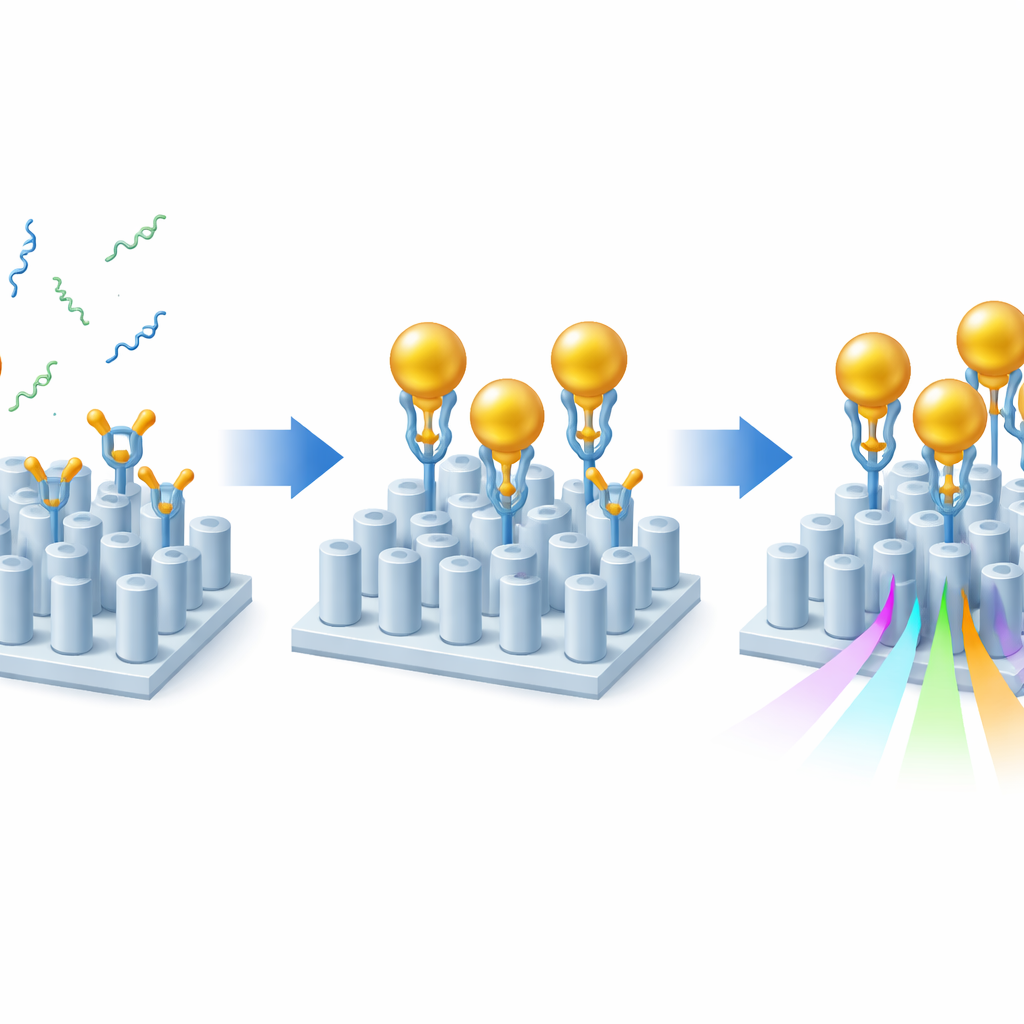
Even with this optimized design, the Alzheimer’s markers on their own would hardly disturb the light. To amplify the effect, the researchers used a sandwich‑style strategy with gold nanoparticles. First, they coated the sensor surface with layers of sticky chemistry that correctly orient specialized antibodies so that their binding sites point into the liquid. These antibodies grab either Aβ40 or Aβ42 from the sample. Next, gold nanoparticles, each decorated with a second antibody that recognizes a different end of the same amyloid fragment, are flowed over the chip. Whenever an amyloid fragment is present, it bridges between the surface and a gold particle, effectively “flagging” its presence. Because gold strongly perturbs the local optical environment, each captured fragment produces a much larger shift in the sensor’s resonance than the peptide alone, boosting sensitivity by about one to two orders of magnitude while still preserving a clean optical signal.
Testing Realistic Samples
To show that the approach works beyond simple laboratory buffers, the team tested their sensor with human blood serum. They diluted serum to make it compatible with microfluidic handling yet kept the amyloid levels representative of those found in patients. Importantly, they were able to detect both Aβ40 and Aβ42 at 0.2 picograms per milliliter in this diluted serum—equivalent to the clinically relevant level of 20 picograms per milliliter in whole blood. By spotting different capture antibodies in separate regions on the same chip, they simultaneously measured both forms of amyloid within a single tiny channel, a key step toward assessing their ratio from a single small sample. The signals clearly distinguished samples spiked with extra amyloid from those containing only the naturally occurring background levels found even in healthy people.
What This Could Mean for Future Care
Taken together, these advances show that a compact, light‑based sensor, boosted by gold nanoparticles, can reach the sensitivities needed for meaningful Alzheimer’s blood tests while handling more than one biomarker at a time. Although further work is needed to turn this into a robust commercial device and to add other markers such as forms of the tau protein, the underlying technology is built from scalable materials and can be paired with simple, handheld optics. If fully developed, it could allow clinicians to monitor brain health using only a small blood sample, making it far easier to detect Alzheimer’s early and to track how well treatments are working over time.
Citation: Guilherme S. Arruda, Katie Morris, Augusto Martins, Yue Wang, Sian Sloan-Dennison, Duncan Graham, Steven D. Quinn, Emiliano R. Martins, and Thomas F. Krauss, "Ultrasensitive Alzheimer’s disease biomarker detection with nanopillar photonic crystal biosensors," Optica 12, 1587-1596 (2025). https://doi.org/10.1364/OPTICA.566672
Keywords: Alzheimer’s blood test, nanophotonic biosensor, amyloid beta, gold nanoparticles, early diagnosis