Clear Sky Science · en
Selective tumor ablation via femtosecond laser resonant with collagen
Turning Light into a Targeted Cancer Tool
Pancreatic cancer is one of the deadliest cancers in medicine, in part because it is hard to remove without harming the fragile organ that surrounds it. This study explores a new way to use ultrafast pulses of invisible infrared light to burn away pancreatic tumors more precisely, by homing in on a structural ingredient that tumors carry in excess. The work hints at future treatments that could spare more healthy tissue while still striking hard at cancer.
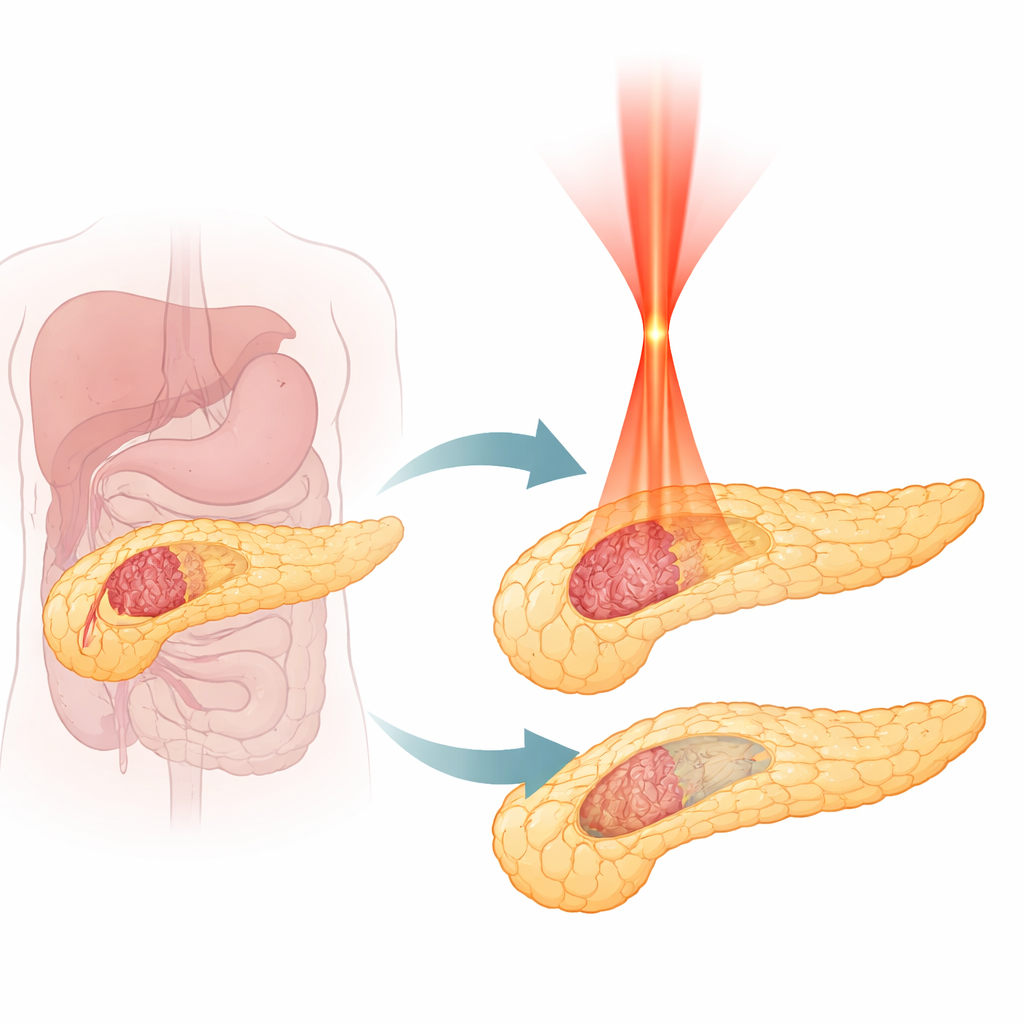
Why Pancreatic Tumors Are So Hard to Treat
Pancreatic ductal adenocarcinoma, the most common form of pancreatic cancer, grows aggressively and is often detected late. Only a small fraction of patients are candidates for surgery, and even modern chemotherapy and radiation offer limited benefit. Heat-based treatments that destroy tissue, such as radiofrequency, microwave, or standard laser ablation, can shrink tumors, but they tend to burn everything in their path. Because the pancreas lies close to vital blood vessels and delicate digestive structures, expanding the treatment zone risks serious complications. Doctors therefore need a method that can distinguish tumor from normal pancreas during destruction, rather than simply cooking a broad area.
Finding a Hidden Weak Spot in Tumor Tissue
The authors focused on a key physical difference between pancreatic tumors and healthy pancreas: tumors are packed with stiff, fibrous material rich in collagen, while normal tissue is softer and looser. Using standard tissue stains and electron microscopy on samples from surgery, they showed that cancer tissue contains dense bundles of collagen fibers, whereas nearby healthy pancreas contains far fewer. They then used infrared spectroscopy to measure how strongly these tissues absorb different colors of mid-infrared light. Both tissues absorb at similar wavelengths, but tumors show a much stronger peak near a wavelength of 6.1 micrometers, which corresponds to vibrations of collagen. This suggested that a laser tuned precisely to that wavelength might heat and break down tumor tissue more efficiently than normal pancreas.
Building a Laser That Listens to Collagen
To test this idea, the team built a powerful mid-infrared laser that emits ultra-short pulses—lasting only a few hundred quadrillionths of a second—centered at 6.1 micrometers. These “femtosecond” pulses limit unwanted heat spread, much like a series of tiny, controlled lightning strikes. The system converts light from a high-power industrial laser into the desired wavelength using special crystals and can deliver over one watt of average power; this is sufficient for practical tissue ablation. The researchers also developed a hollow fiber made of glass that can guide this light along a flexible path, an important step toward threading the laser through a thin needle into the body for minimally invasive procedures.
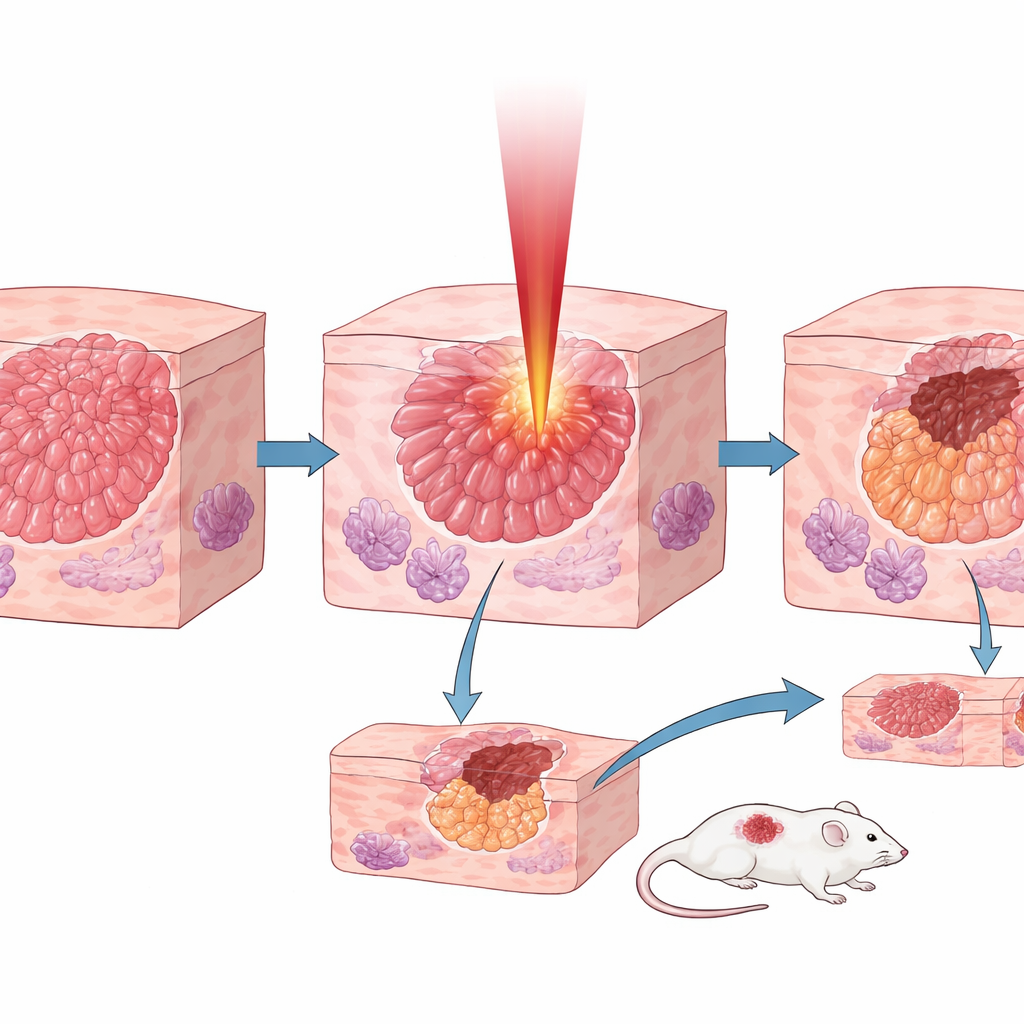
Testing Selectivity in Cells, Mice, and Human Tissue
In cell cultures of two pancreatic cancer lines, the 6.1-micrometer laser was far more lethal than lasers at 1 or 3 micrometers, sharply reducing cell survival within seconds to minutes of exposure. In mice bearing pancreatic tumors under the skin, three different wavelengths were compared. Even though the 1-micrometer beam carried much higher power, it removed only shallow layers of tumor. The collagen-tuned 6.1-micrometer laser achieved ablation depths five to ten times greater and slowed tumor growth so dramatically that treated tumors ended up only about one-eighth the size of those in untreated animals. Most importantly, when the team applied the 6.1-micrometer laser to human tumor samples and adjacent normal pancreas, the cuts in tumor were two to three times deeper than in healthy tissue under the same conditions, revealing true selectivity. In contrast, when they tested another liver tumor type that does not accumulate extra collagen, the advantage largely disappeared, reinforcing collagen’s central role. Finally, they showed that the hollow fiber could deliver the same selective effect, supporting future needle-based treatments.
What This Could Mean for Future Cancer Care
The study demonstrates that carefully tuned bursts of mid-infrared light can exploit a material difference between tumors and healthy tissue to achieve more selective destruction. By targeting collagen-rich regions, the 6.1-micrometer femtosecond laser cuts deeper into pancreatic cancer while sparing more of the surrounding organ. Although the work is still at the experimental stage, and more testing in realistic models and clinical settings is needed, it points toward a new class of image-guided, fiber-delivered procedures that could treat not only pancreatic tumors but also other collagen-rich cancers with greater precision and fewer side effects.
Citation: Dunxiang Zhang, Xing Huang, Xuemei Yang, Ning Xia, Kan Tian, Jinmiao Guo, Maoxing Xiang, Linzhen He, Zhizhuo Fu, Ang Deng, Han Wu, Yuxi Wang, Wonkeun Chang, Bole Tian, Junjie Xiong, Qi Jie Wang, Anderson S. L. Gomes, and Houkun Liang, "Selective tumor ablation via femtosecond laser resonant with collagen," Optica 12, 1578-1586 (2025). https://doi.org/10.1364/OPTICA.561337
Keywords: pancreatic cancer, laser ablation, collagen, mid-infrared, minimally invasive surgery