Clear Sky Science · en
Polyethylene terephthalate degradation by Drosophila melanogaster through heterologous expression of glycosylated polyethylene terephthalate hydrolase (PETase)
Turning Bugs into Tiny Recyclers
Plastic bottles and food packaging made from PET (polyethylene terephthalate) are everywhere, but turning used PET back into raw material usually takes high heat and harsh chemicals. This study asks a surprising question with big implications for cleaner recycling: could common fruit flies be re‑engineered to help break down PET gently, using biology instead of smokestacks and furnaces?
Why Plastic Is So Hard to Get Rid Of
PET is popular because it is strong, light and long‑lasting. Those same traits make it stubborn in landfills and oceans. Today, most PET recycling relies on energy‑intensive chemical treatments at temperatures of several hundred degrees Celsius, which add to pollution and greenhouse‑gas emissions. A few years ago, researchers discovered a bacterial enzyme called PETase that can chew up PET at much lower temperatures, closer to room temperature. That raised a tantalizing possibility: if living systems could be harnessed to run such reactions in everyday conditions, plastic recycling might become cleaner, cheaper and more flexible.
Borrowing a Bacterial Trick for a Fruit Fly
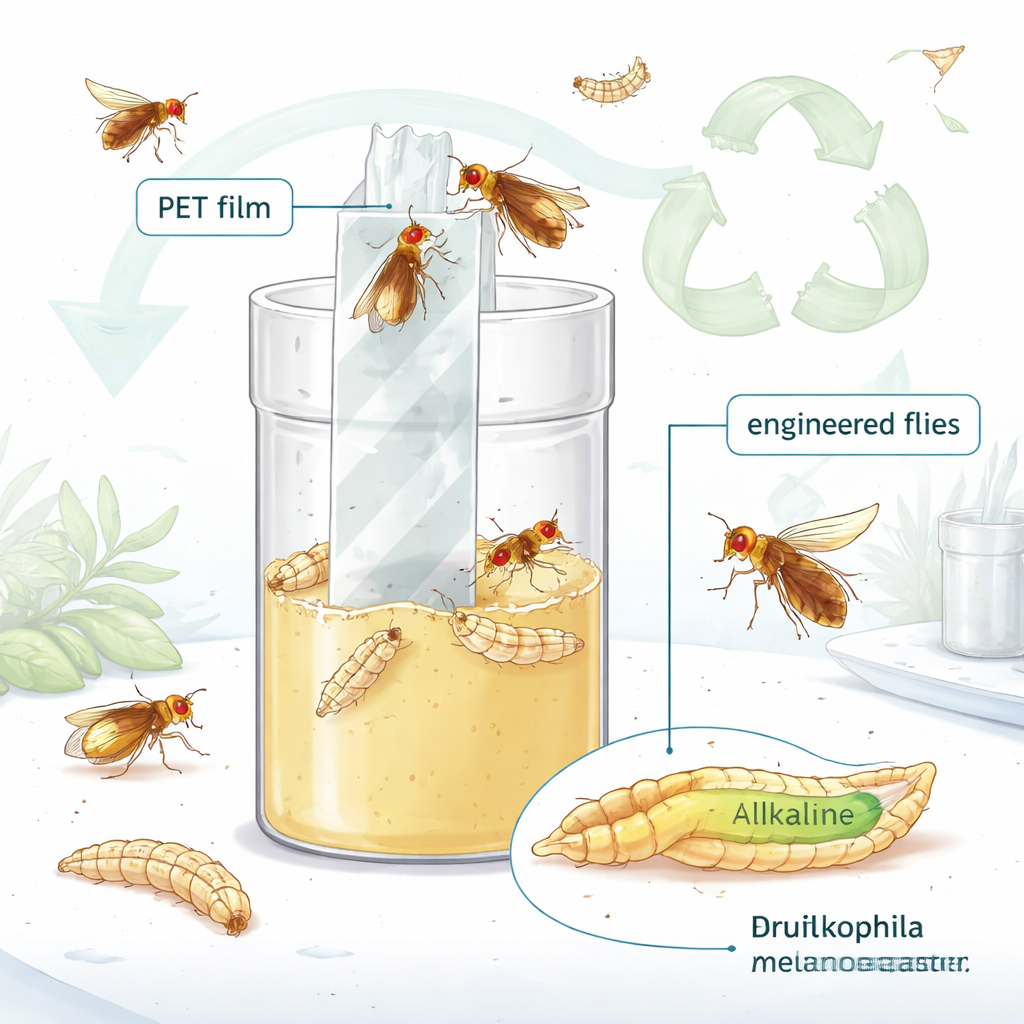
The authors of this paper took PETase from a plastic‑eating bacterium and engineered the fruit fly, Drosophila melanogaster, to produce and secrete this enzyme in parts of its gut and in its salivary glands. They chose these tissues because sections of the fly intestine are naturally neutral to alkaline—exactly the pH range in which PETase works best. First, they confirmed that the modified flies really were making the enzyme, and that it was released into the digestive tract and saliva. They then fed the larvae a specially designed water‑soluble PET‑like material and measured a key breakdown product, terephthalic acid, inside the larvae and in their food. Only the PETase‑producing flies created this product, showing that the engineered insects could indeed digest PET‑like plastic from the inside.
From Soft Plastics to Solid Films
The team next asked whether the flies could affect tougher, solid PET like that used in bottles and packaging. They stood thin PET films upright in fly food and allowed generations of the engineered flies to live, feed and crawl over them. To keep the surroundings slightly alkaline—again, favoring PETase—they mixed in different amounts of calcium carbonate, a mild base. Over weeks, films exposed to the PETase‑flies developed visible surface damage that increased with higher calcium carbonate, while films kept with control flies remained mostly unchanged. Using electron microscopes and surface‑chemistry measurements, the researchers showed that the treated films had roughened, pitted surfaces and more oxygen on their top layers, both signs of ongoing breakdown and reaction with water.
How Sugar Coats Change Enzyme Behavior
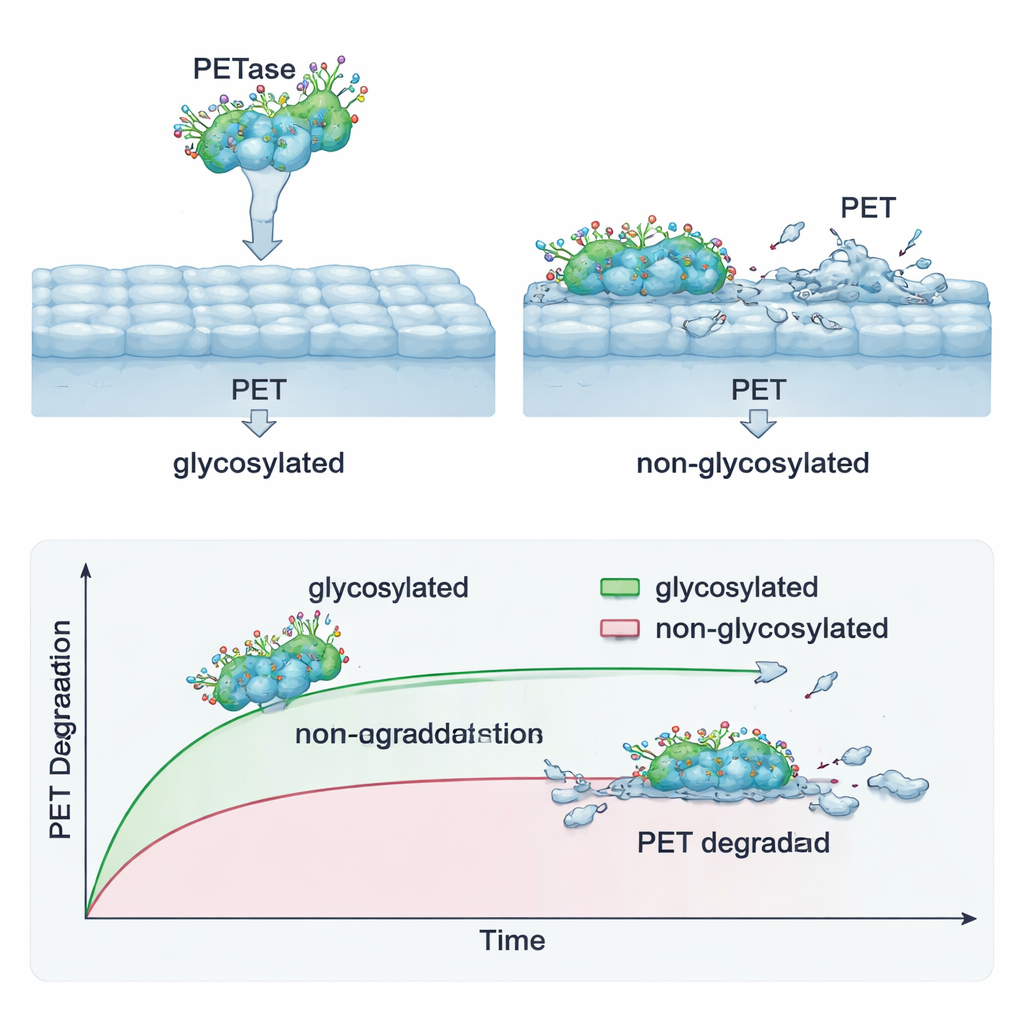
An unexpected twist came from the way animal cells process foreign proteins. When PETase was made by flies or human cells, it picked up sugar chains—chemical “coats” known as glycosylation—that made the enzyme larger. By comparing the natural bacterial version, the fly‑made version, and chemically de‑sugared versions of each, the scientists found a trade‑off. Enzymes without sugars stuck more tightly to PET and broke it down faster at first, but they lost activity more quickly over time, especially at warmer temperatures. The sugar‑coated PETase worked more slowly on solid PET but stayed active for weeks, continuing to generate breakdown products long after the faster forms had faded. Microscopy suggested that the coated enzyme nibbled in scattered pits, while the uncoated forms eroded the plastic more evenly across its surface.
From Lab Curiosity to Future Recycling Tools
Beyond flies, the study discusses how insects and other organisms might serve as moving platforms that deliver plastic‑degrading enzymes to hard‑to‑reach surfaces, including in moist but not fully wet environments. It also highlights challenges: glycosylation can keep enzymes from gripping plastic efficiently, and any real‑world use of modified insects would demand strict safety measures and public oversight. Still, the work shows that an established laboratory insect can be re‑tooled to secrete an industrially interesting enzyme and to alter actual PET items placed in its habitat.
What This Means for Everyday Life
For a layperson, the key message is that living creatures can be redesigned to help tackle one of our most stubborn waste problems. These engineered fruit flies are not ready to patrol landfills, but they provide proof that animals can host and secrete plastic‑eating enzymes that work outside the lab flask, on real pieces of plastic and at comfortable temperatures. Future advances could combine more powerful enzyme designs, safer genetic safeguards and perhaps different insect species to create biology‑based recycling systems that complement, or one day partly replace, today’s hot and dirty plastic‑processing plants.
Citation: Sanuki, R., Minami, H., Kawano, E. et al. Polyethylene terephthalate degradation by Drosophila melanogaster through heterologous expression of glycosylated polyethylene terephthalate hydrolase (PETase). Commun. Sustain. 1, 36 (2026). https://doi.org/10.1038/s44458-026-00047-5
Keywords: plastic biodegradation, PETase, engineered insects, fruit fly model, sustainable recycling