Clear Sky Science · en
The quaternary structure origin of the fibrillation of sickle hemoglobin: a molecular dynamics study
Why this blood story matters
Sickle cell disease begins with a tiny change in one blood protein, yet it can reshape red blood cells, block vessels, and cause lifelong pain. This study takes a close, physics-based look at how that single change makes the protein hemoglobin stack into stiff fibers inside the cell. By using advanced computer simulations, the authors connect atomic details to whole-cell problems, offering fresh clues for how future drugs might keep hemoglobin from jamming our bloodstream.
A tiny swap with big consequences
Hemoglobin is the oxygen-carrying protein that packs our red blood cells. Each hemoglobin unit is built from four chains that can arrange themselves in two main overall shapes, known as the relaxed (R) state and the tense (T) state. In sickle cell disease, just one building block in one of these chains is swapped: a charged glutamate is replaced by a greasy valine at position six of the beta chain. Under low-oxygen conditions, this altered version, called sickle hemoglobin, links together into long fibers that distort normally round red blood cells into stiff, sickle-like shapes, which can clog small vessels and starve tissues of oxygen.
Following the shifting shape of hemoglobin
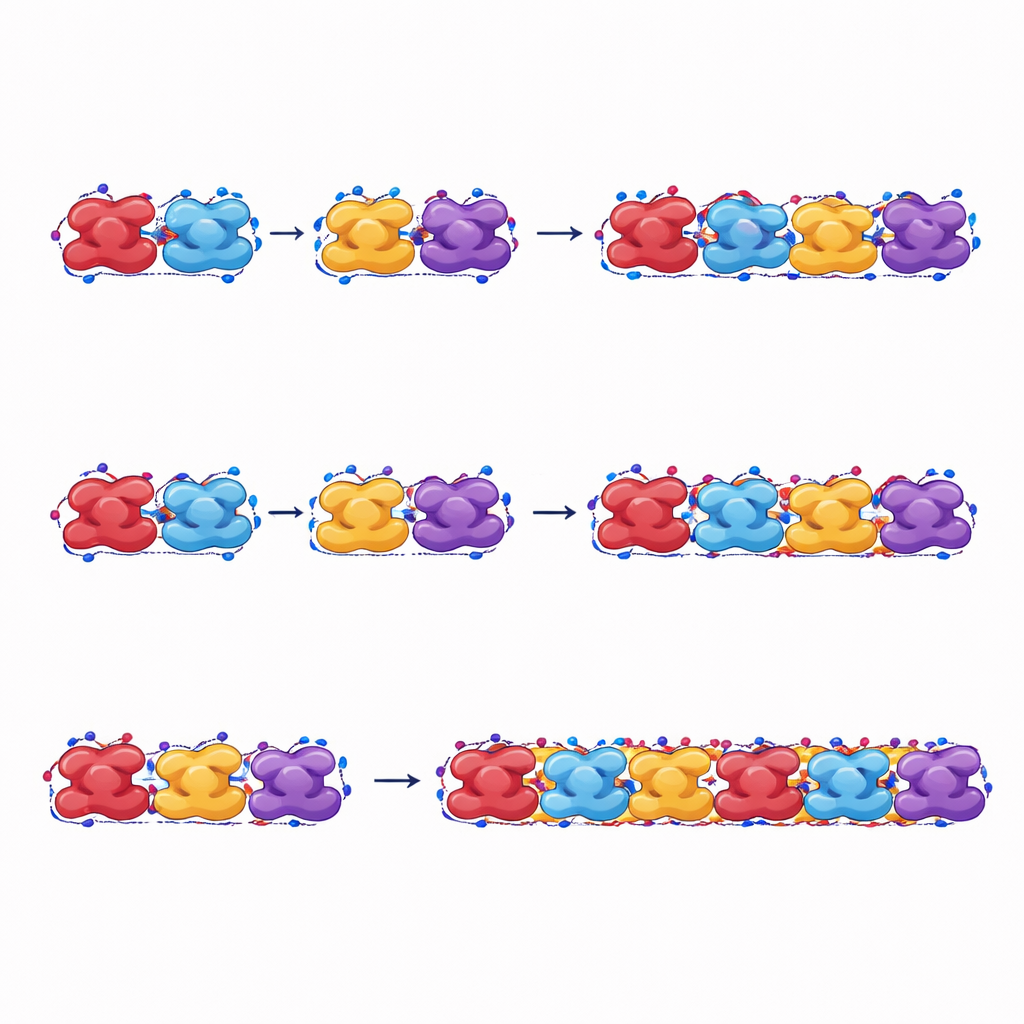
The authors focus on how whole hemoglobin units rotate and pack together in a fiber. They track this large-scale shape change with a single angle that measures how two halves of the protein twist relative to each other. Using molecular dynamics simulations—virtual experiments that compute how atoms move over time in water and salt—they gently push this angle across a wide range and calculate how much energy each shape costs. They find that both normal and sickle hemoglobin can explore many such shapes at body temperature. Sickle hemoglobin shows a slightly preferred angle, but these shape preferences alone are not enough to explain why only the sickle form forms stable fibers.
Why sickle fibers stick and normal ones fall apart
To get at the heart of fiber formation, the study measures how strongly neighboring hemoglobin units attract each other along and across the fiber. The team computes the cohesive energy—the net pull that holds units together—for many different overall shapes. For sickle hemoglobin, this cohesive energy stays negative (meaning attractive) over the full range of shapes they tested, so fibers remain stable and do not readily fall apart. For normal hemoglobin, in contrast, some shapes actually have positive cohesive energy, making fibers at those angles unstable and prone to spontaneous breakup. A key difference is how the mutated valine side groups tuck into neighboring proteins: in sickle hemoglobin, these greasy patches form robust contacts both sideways and along the fiber axis, directly promoting fiber growth.
How these fibers bear load and break
The authors also stretch their simulated fibers to mimic mechanical stress. When pulled along their length, fibers made from sickle hemoglobin endure higher forces and strains and dissipate more energy before breaking than those made from normal hemoglobin. Even the relaxed form of sickle fibers, which is not the one where fibers first appear, can still maintain the chain through plastic changes in shape, helping keep the fiber intact once it has formed. Fibers of normal hemoglobin, by contrast, separate more easily between neighboring units and absorb less energy before failure, confirming that they are less stable under stress.
Clues for future treatments and new materials
Taken together, the simulations show that a single amino-acid change reshapes the way hemoglobin units meet, twisting the overall assembly into angles that favor strong attraction, especially in the tense state. This combination of geometry and stickiness lets sickle hemoglobin form long, stiff fibers that resist both thermal motion and mechanical pulling, which ultimately deform red blood cells and block blood flow. By pointing to specific features of the overall protein arrangement that control fiber stability, the work suggests that effective antisickling drugs could work by nudging hemoglobin toward shapes where fibers become energetically unfavorable and mechanically fragile—turning a deadly molecular jam back into a free-flowing blood stream.
Citation: Jiang, M., Qin, Z. The quaternary structure origin of the fibrillation of sickle hemoglobin: a molecular dynamics study. npj Soft Matter 2, 7 (2026). https://doi.org/10.1038/s44431-026-00019-8
Keywords: sickle cell disease, hemoglobin fibers, protein aggregation, molecular dynamics, blood disorders