Clear Sky Science · en
Dynamin optimizes protein-membrane interactions for fission
How cells neatly pinch off their own membranes
Every second, your cells are busy pinching off tiny bubbles of membrane to move cargo, remodel internal compartments, and fight viruses. This seemingly simple act of cutting a narrow membrane tube into two pieces turns out to be surprisingly hard from a physics standpoint. This paper asks a deceptively basic question: how does a protein called dynamin make membrane cutting both possible and reliable, and what features of the protein are truly essential for the job?
The cellular pinchers at work
Dynamin is a molecular machine that forms a collar around thin membrane tubes and tightens like a noose to help them break. Such fission events underlie processes like endocytosis, where cells draw material inward, as well as the division of internal structures such as mitochondria. Left alone, a membrane tube strongly resists breaking, because it must first be squeezed down to nearly the thickness of the membrane itself and pass through an unstable intermediate state. This energetic hurdle is far higher than the random jostling energy available inside a cell, so specialized fission proteins like dynamin are essential.
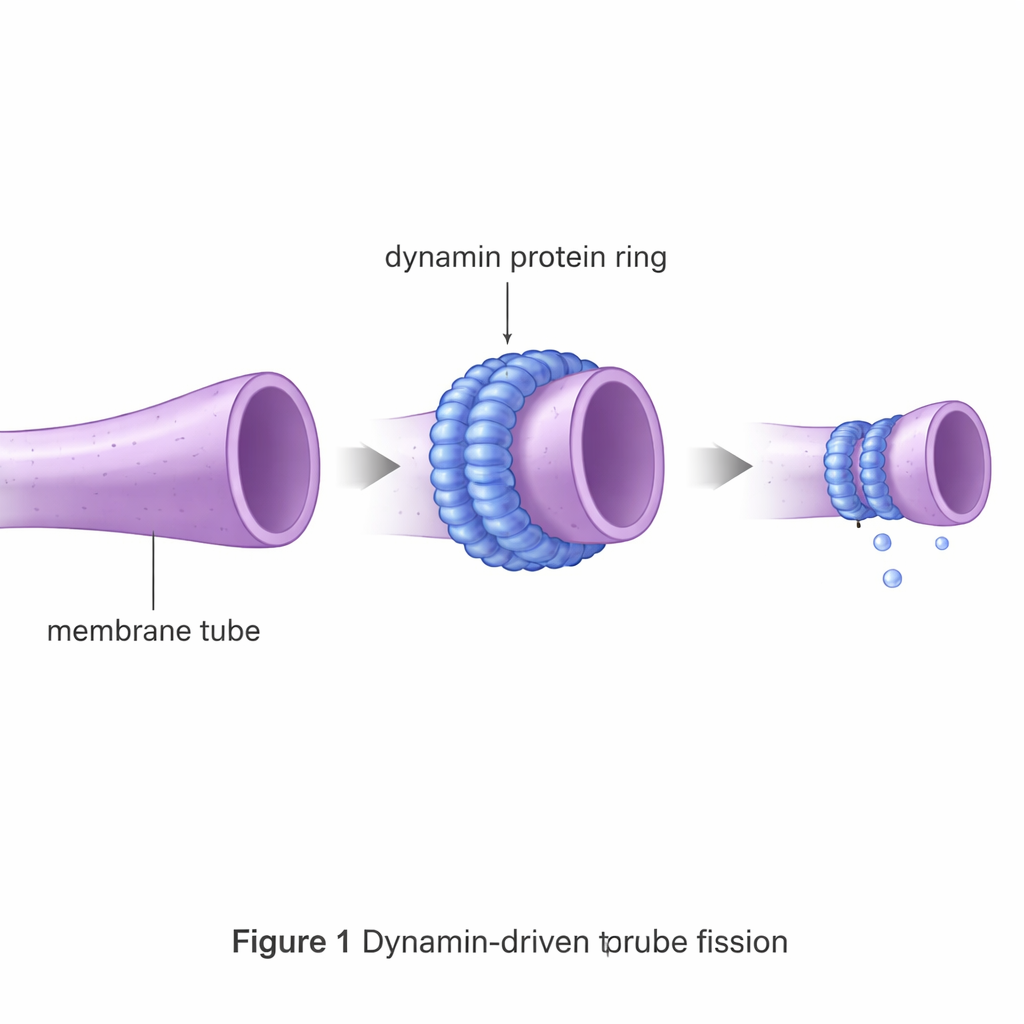
Using virtual membranes to test protein designs
Directly watching the critical instant when a tube snaps is extremely difficult in experiments: fast imaging lacks molecular detail, and high-resolution methods freeze the system in time. To bridge this gap, the authors use a powerful theoretical tool called self-consistent field theory. Instead of tracking every atom, they describe lipids and solvent as flexible chains that feel smoothly varying fields. Proteins are introduced not as full atomic structures but as torus-shaped external potentials that either simply exclude space, stick to the membrane surface, or insert into the outer layer. With this framework, they can calculate both the resulting membrane shapes and the full free-energy cost of going from an intact tube to the fission intermediate.
What kinds of grip help a tube break?
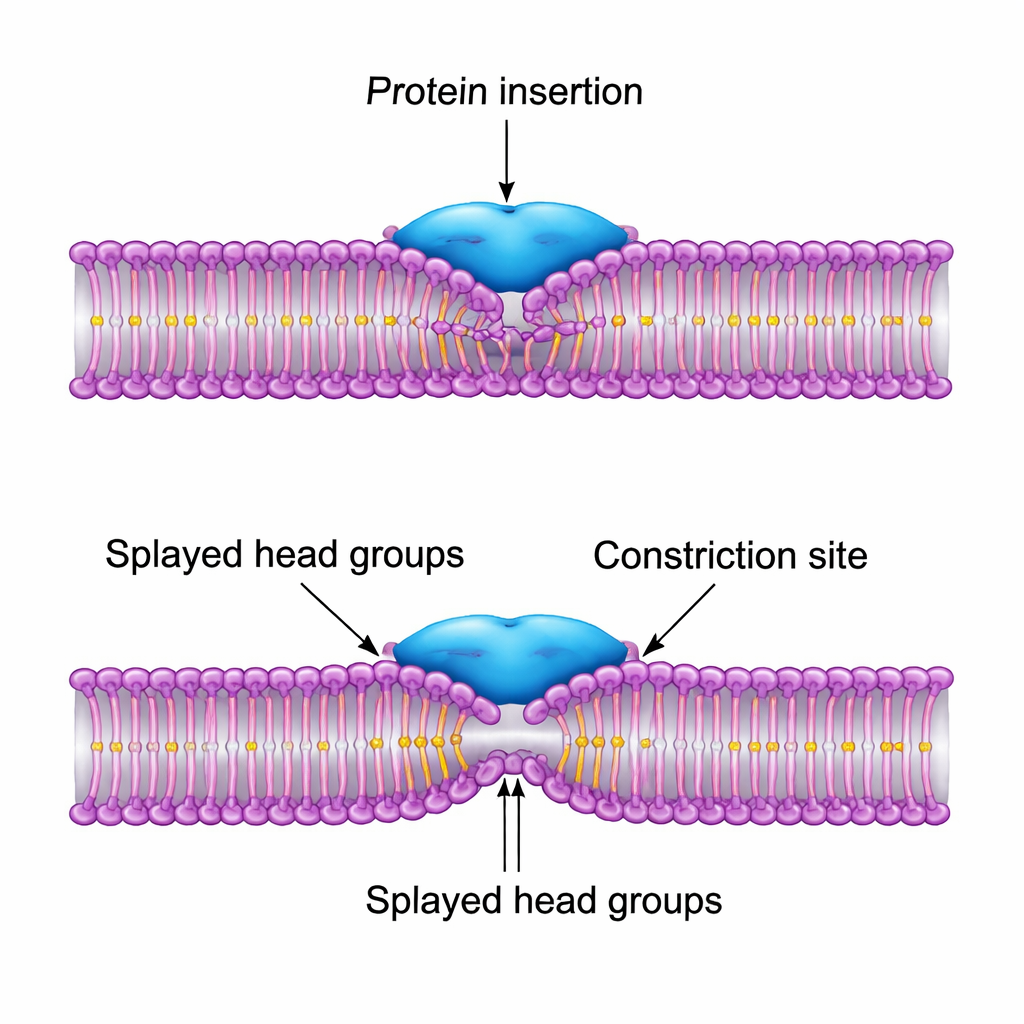
The team systematically varies how a dynamin-like protein interacts with the membrane. Some model proteins only crowd the surface, acting as a rigid cuff; others are mildly or strongly sticky to the outer head groups; still others imitate the real dynamin PH domain by inserting hydrophobic parts into the outer leaflet and “splaying” the head groups aside. For each case they examine three linked quantities: how strongly the protein adheres to an unconstricted tube, how much curvature and constriction it induces while bound, and how high the energy barrier remains for the tube to pass into a hemifission state where the inner layers have merged. They find that simple squeezing lowers the barrier but not enough to explain efficient fission, and strong surface adhesion can actually hinder breaking because the membrane must first peel away from the protein to complete collapse.
Why shallow insertion beats simple squeezing
The most effective design is one in which the protein partially inserts between lipid head groups and pulls their tails upward, creating a local, chevron-shaped bend in the membrane. This “splay” shifts the narrowest point of the tube slightly to the side of the protein ring rather than directly beneath it. As a result, the membrane can complete the critical collapse into the hemifission intermediate without detaching from the protein, and the overall energy barrier drops by more than an order of magnitude compared with a bare tube. When the authors let the protein parameters adjust automatically to minimize this barrier, the optimal solution closely matches the size and action of dynamin’s PH domain: a moderately wide hydrophobic patch that inserts shallowly, not a broad, overly sticky surface that would strip lipids from solution.
Design rules for nature and for nanotechnology
To a non-specialist, the key message is that dynamin works not just by tightening a belt around the membrane, but by carefully tuning how and where it grips the outer layer. A shallow wedge that splays the outer lipids and focuses curvature just beside the protein proves far more effective than brute-force constriction or strong sticking. These results help explain why evolution has favored dynamin’s particular architecture and suggest design principles for synthetic proteins or drugs that need to controllably cut or reshape membranes in medicine and nanotechnology.
Citation: Spencer, R.K.W., Müller, M. Dynamin optimizes protein-membrane interactions for fission. npj Soft Matter 2, 6 (2026). https://doi.org/10.1038/s44431-026-00018-9
Keywords: membrane fission, dynamin, protein–membrane interactions, membrane curvature, self-consistent field theory