Clear Sky Science · en
White matter aging across the lifespan in Down syndrome: developmental origins, Alzheimer's disease progression, and therapeutic implications
Why this matters for families and caregivers
People with Down syndrome are now living longer than ever, but this welcome progress has brought a sharp rise in Alzheimer’s disease at relatively young ages. This review explains how the brain’s “wiring” system—called white matter—changes from before birth through late adulthood in Down syndrome, and how those changes help drive memory loss and thinking problems. Understanding this wiring story opens new paths for earlier diagnosis and more targeted treatments.
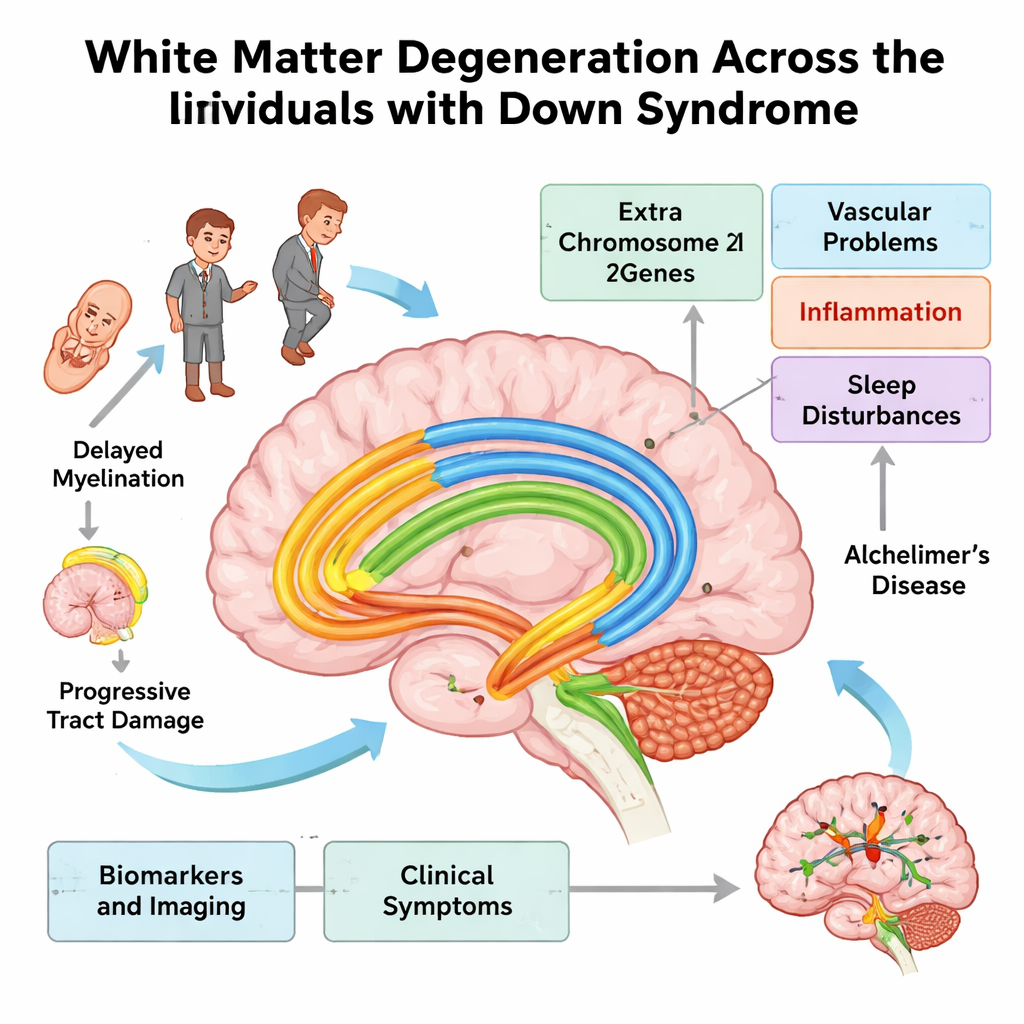
The brain’s wiring and how it shapes thinking
Nearly half of the human brain is made up of white matter, the bundled, insulated nerve fibers that connect distant regions—much like high-speed internet cables. These fibers are wrapped in a fatty coating called myelin, made by support cells called oligodendrocytes, which allows signals to travel quickly and efficiently. When myelin or the underlying fibers are damaged, signals slow down, networks fall out of sync, and the brain must work harder. The authors argue that in Down syndrome, this wiring is vulnerable across the entire lifespan, contributing to slower processing, learning challenges, and, later on, dementia.
Early-life wiring differences that never fully catch up
Evidence from brain tissue and child brain scans shows that white matter in Down syndrome develops differently from the very beginning. Studies of fetuses, infants, and children reveal delayed and reduced myelination, especially in memory-related areas such as the hippocampus and in long-range connections between the front, side, and back of the brain. Advanced MRI techniques confirm that school-age children with Down syndrome already have signs of weaker wiring in these long association pathways, even as some crossing pathways between the two brain hemispheres look unusually compact and immature. These early wiring differences do not simply reflect later damage; they represent a built-in developmental vulnerability that the brain carries forward into adulthood.
Accelerated wear and tear with aging and Alzheimer’s disease
As adults with Down syndrome age, almost all develop Alzheimer’s brain changes—amyloid plaques and tau tangles—by midlife. The review shows that white matter does not degenerate evenly; instead, specific tracts are hit hardest, especially those that mature late in life. These include long association fibers that support memory and language, the corpus callosum that links the two hemispheres, and projection and cerebellar pathways that coordinate movement. MRI measures reveal declining fiber organization and increasing water content in these tracts, while bright spots on scans called white matter hyperintensities, often linked to small vessel disease and low blood flow, expand rapidly starting in the mid-30s. These imaging changes track closely with amyloid build-up, blood markers of nerve injury and inflammation, and stepwise worsening from normal cognition to mild impairment and dementia.
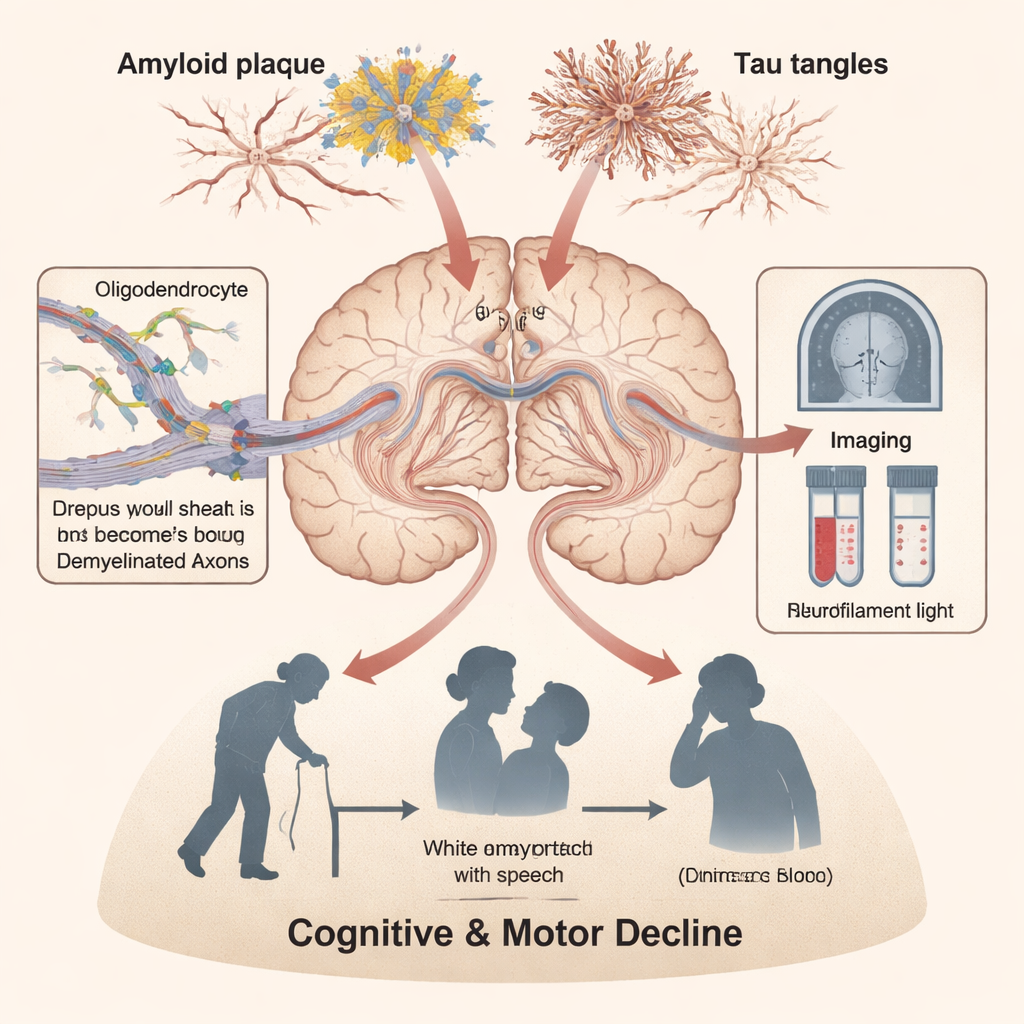
Cellular, blood, and lifestyle clues to white matter damage
Zooming in to the cellular level, the authors describe how the extra copy of chromosome 21 alters key genes that guide oligodendrocyte development and myelin formation. In brain tissue and mouse models, myelin sheaths are thinner, fewer axons are properly wrapped, and progenitor cells that should renew the myelin system show signs of premature aging and inflammatory stress. Blood and spinal fluid tests reveal that higher levels of neurofilament light, a protein released when long nerve fibers are injured, strongly mirror white matter damage on MRI. Other markers of amyloid, tau, and glial activation also rise as white matter hyperintensities grow. Common problems like sleep apnea appear to worsen this wiring damage, particularly in the same long tracts that support thinking and attention.
What this means for treatment and early detection
The authors conclude that white matter degeneration is both a sensitive warning sign and a promising treatment target in Down syndrome–related Alzheimer’s disease. Because wiring changes emerge before obvious memory loss, combining brain scans with blood tests such as neurofilament light may help identify people at highest risk and monitor how quickly disease is progressing. At the same time, the biology of oligodendrocytes, inflammation, blood vessel health, and sleep offers multiple levers for intervention—from drugs that promote myelin repair or clear senescent cells to aggressive treatment of sleep-disordered breathing. By protecting the brain’s wiring, it may be possible to delay or lessen dementia in people with Down syndrome and to gain broader insights relevant to Alzheimer’s disease in the general population.
Citation: Silva, J.A., Liou, JJ., Parikh, S. et al. White matter aging across the lifespan in Down syndrome: developmental origins, Alzheimer's disease progression, and therapeutic implications. npj Dement. 2, 16 (2026). https://doi.org/10.1038/s44400-026-00062-2
Keywords: Down syndrome, white matter, Alzheimer’s disease, myelin, brain aging