Clear Sky Science · en
15 years of longitudinal genetic, clinical, cognitive, imaging, and biochemical measures in DIAN
Why this matters for families and the future of Alzheimer’s treatment
Most people think of Alzheimer’s disease as something that appears late in life, often without warning. But in a small number of families, specific inherited gene changes almost guarantee that a person will develop Alzheimer’s at a relatively young age. The Dominantly Inherited Alzheimer Network (DIAN) Observational Study has spent 15 years closely following these families around the world. By watching how the disease unfolds long before memory problems appear, DIAN is revealing a detailed timeline of brain changes that may help doctors detect, prevent, or delay Alzheimer’s in many more people.
A global effort following high‑risk families
DIAN tracks adults from families that carry rare mutations in three genes linked to an inherited form of Alzheimer’s disease. Some participants have the mutation and some do not, but none of them are told their status by the study unless they choose separate clinical testing. The project began in 2008 with 10 sites in three countries and has grown to 23 performance sites in 11 countries, working in seven languages. Over 660 people have enrolled, with more than 300 still actively taking part. At regular visits, volunteers undergo medical and memory exams, brain scans, spinal fluid and blood tests, and, for those who consent, brain donation after death. Non‑carrier relatives serve as an unusually well‑matched comparison group, helping researchers tease apart normal aging from disease‑related changes.
Seeing the disease decades before symptoms
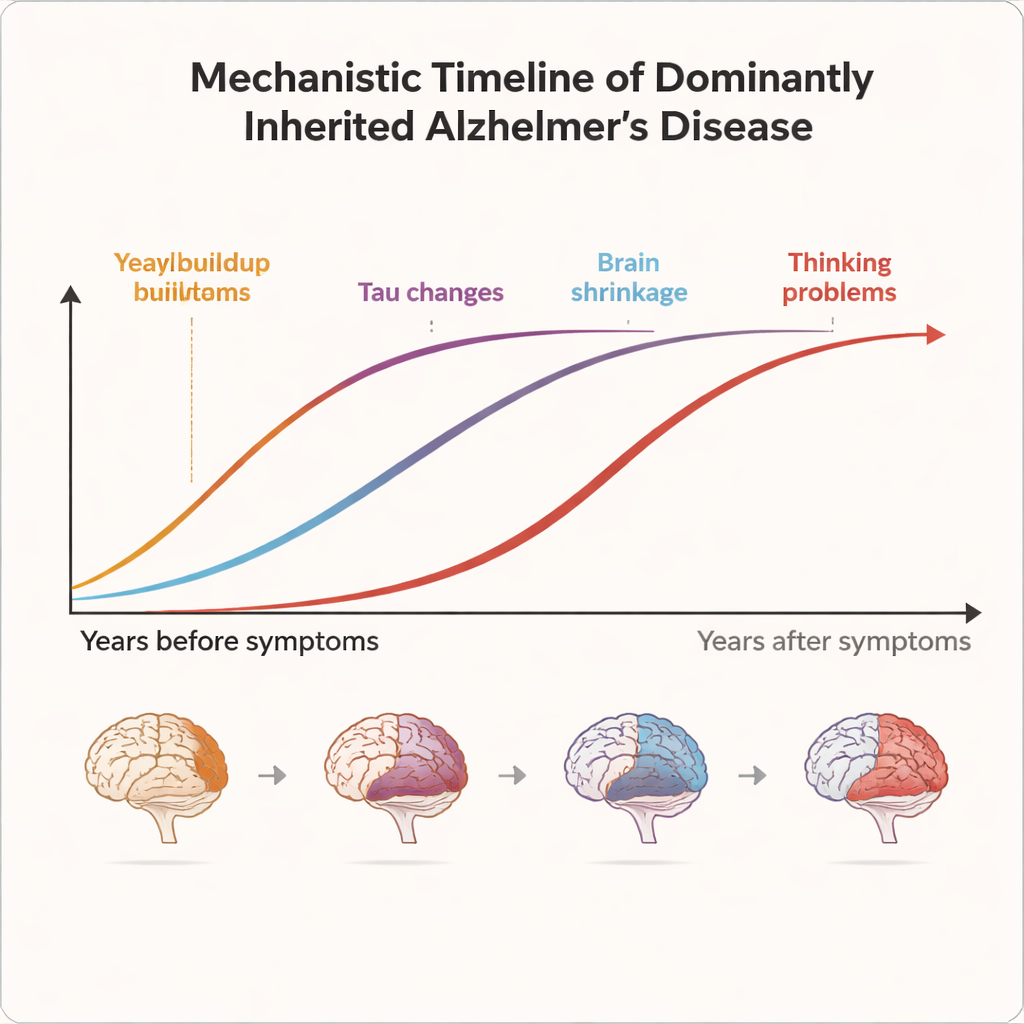
Because the age at which symptoms usually begin is fairly predictable within each family, researchers can estimate how many years each carrier is from their likely onset. This allows them to line up people along a “disease clock,” from more than 20 years before problems start to many years afterward. DIAN’s results show that Alzheimer’s is not a sudden event but a long process. Sticky protein clumps called amyloid plaques begin building up in the brain 15 to 20 years before symptoms, appearing on specialized PET scans and mirrored by falling levels of one amyloid form in the spinal fluid. About 10 to 15 years before expected onset, other warning signs emerge: abnormal tau proteins rise in spinal fluid, brain metabolism slows, and subtle thinning of the cortex appears on MRI scans. Only several years later do changes in thinking and daily function become detectable on detailed cognitive tests.
A living library of measurements and samples
To turn these observations into broadly useful science, DIAN has built one of the richest collections of Alzheimer’s‑related data and tissues in the world. Eight specialized cores manage clinical information, cognitive testing, brain imaging, fluid biomarkers, genetics, brain pathology, statistics, and overall administration. Blood and spinal fluid samples are processed in a central biobank, where advanced tools measure dozens of proteins linked to amyloid, tau, inflammation, and nerve cell injury. Genetic and “multi‑omic” analyses examine DNA, RNA, proteins, fats, and other molecules in blood, brain tissue, and cell models derived from participants. A growing neuropathology program carefully examines donated brains to confirm diagnoses and stage disease. All of this information is cleaned, quality‑checked, and frozen into yearly data sets that outside investigators can request under strict privacy safeguards.
Driving trials and next‑generation tools
DIAN is not only a study of what happens naturally; it is also the backbone of a companion clinical‑trial program that tests drugs aimed at slowing or preventing inherited Alzheimer’s. Because observational and treatment studies use harmonized procedures, their results can be combined and compared. The detailed DIAN timeline of brain changes has already helped shape international criteria for defining Alzheimer’s biologically, based on amyloid, tau, and neurodegeneration markers rather than symptoms alone. Looking ahead, the network is expanding into remote assessments using smartphone apps, home‑health visits, and wearable devices to detect very early thinking changes and track daily function. It is also investing heavily in blood‑based tests and deeper molecular profiling to find new treatment targets and understand why the disease progresses differently across individuals and populations.
What this means for people worried about Alzheimer’s
The DIAN study shows that in inherited Alzheimer’s, the brain begins to change many years before memory fails—and that these hidden shifts can be measured in blood, spinal fluid, and scans. Although the studied mutations are rare, the underlying disease process appears to closely resemble that of the far more common late‑onset form of Alzheimer’s. This makes DIAN a powerful model for the entire field. By mapping the step‑by‑step journey from healthy brain to dementia, and by sharing its carefully gathered resources worldwide, DIAN is helping scientists design earlier and more precise interventions. For families facing inherited risk, and for the millions affected by Alzheimer’s more broadly, these insights offer a clearer path toward earlier detection, better monitoring, and ultimately more effective prevention and treatment.
Citation: Daniels, A.J., McDade, E., Llibre-Guerra, J.J. et al. 15 years of longitudinal genetic, clinical, cognitive, imaging, and biochemical measures in DIAN. npj Dement. 2, 13 (2026). https://doi.org/10.1038/s44400-025-00047-7
Keywords: Alzheimer’s disease, genetic dementia, brain biomarkers, longitudinal study, early detection