Clear Sky Science · en
AI driven 3D subcellular RPE map discovers cell state transitions in establishment of apical-basal polarity
Why the Eye’s Support Cells Matter
The sharp vision we enjoy every day depends on a thin sheet of support cells at the back of the eye called the retinal pigment epithelium (RPE). When these cells lose their orderly internal structure, sight-stealing diseases such as age-related macular degeneration can follow. This study combines stem cell biology, advanced microscopy, artificial intelligence, and mathematical modeling to build a detailed three-dimensional “digital twin” of an RPE cell, revealing how its internal parts rearrange as it matures and what goes wrong when that process is disturbed.
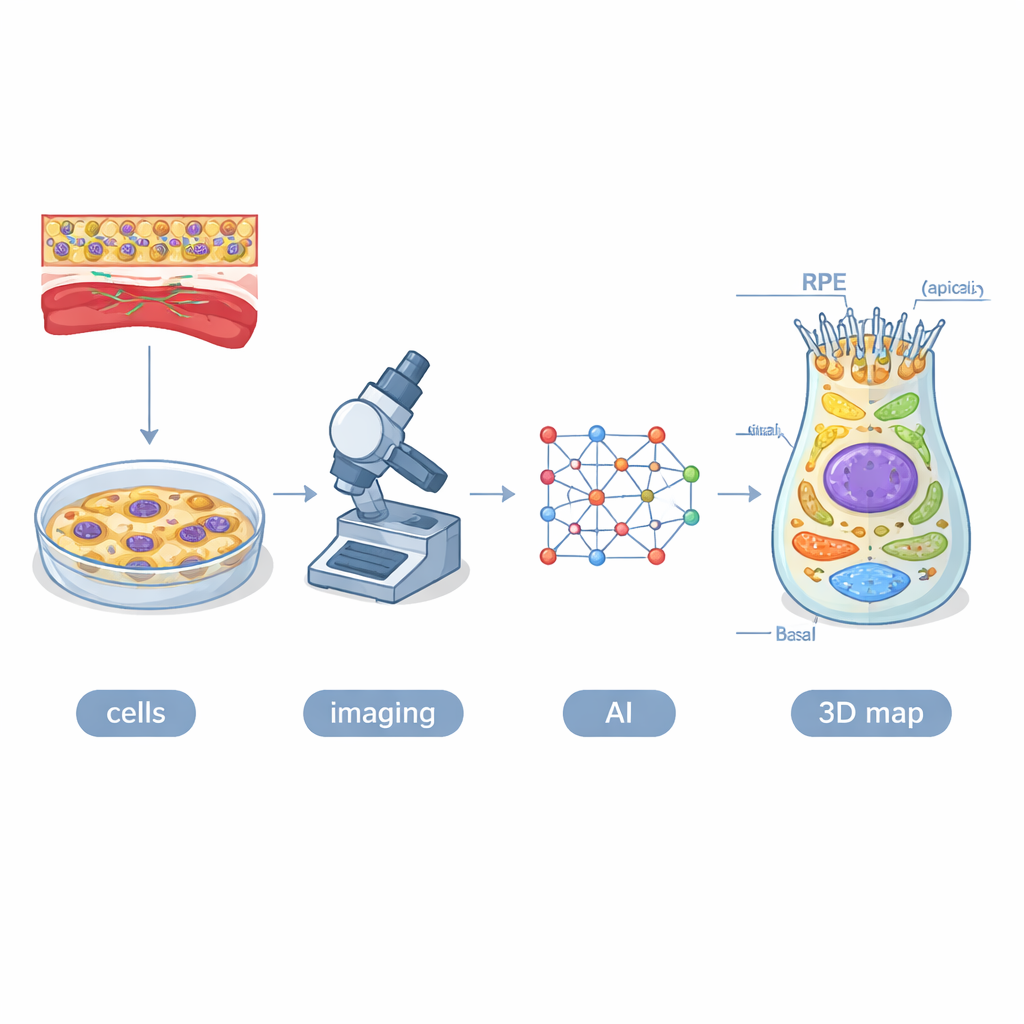
Building a Digital Twin of Eye Cells
The researchers started with human induced pluripotent stem cells—cells reprogrammed to behave like embryonic cells—and coaxed them into becoming RPE. They used 16 engineered cell lines in which different cellular structures, such as mitochondria, lysosomes, and the cell’s outer skeleton, glowed green under the microscope. Over four weeks, they imaged roughly 1.3 million cells in 3D using high-content confocal microscopy. To make sense of this enormous dataset, they created an AI system called POLARIS, based on a type of neural network that can automatically outline each cell, its nucleus, and the tagged organelles in every image slice. Human experts checked and refined the machine’s work, then the team combined the results into an average three-dimensional model—a digital twin—of a typical RPE cell at each stage.
Two Paths: Healthy Maturation vs. Blocked Polarity
RPE cells must become “polarized,” with distinct top (apical) and bottom (basal) sides, to shuttle nutrients, clear waste, and talk to both the retina and the blood supply. To encourage this, the team treated some cultures with a molecule called PGE2, which promotes formation of a tiny sensory structure known as the primary cilium and enhances polarity. Other cultures received HPI4, which interferes with cilia and prevents proper polarization. Using POLARIS-derived measurements, the researchers showed that in the healthy path, cells grew taller and narrower and their nuclei became more compact and rounded. Under HPI4, cells stayed flatter and wider, with more irregular shapes. Statistical models revealed that in successfully polarizing cells, these shape changes followed a predictable, non-random sequence, while blocked cells drifted into more variable, disordered states.
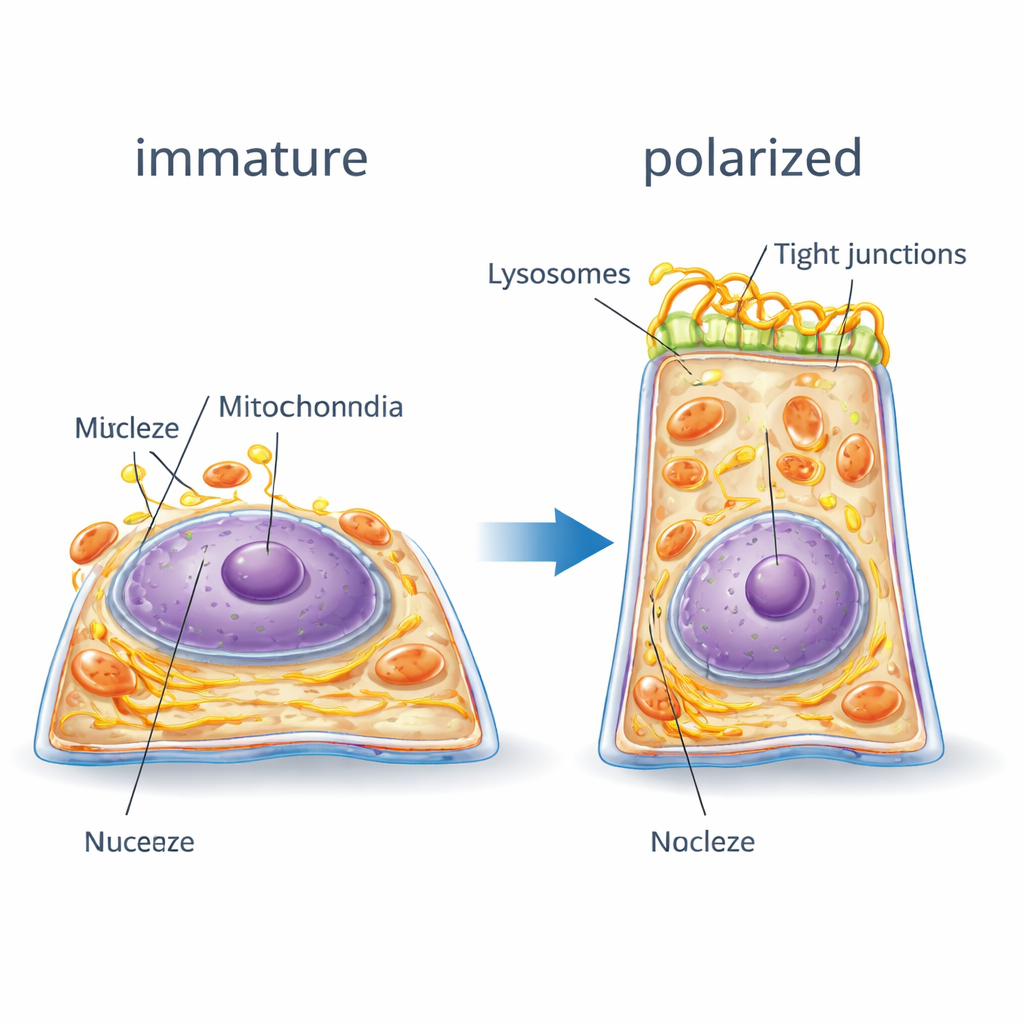
How the Cell’s Interior Rearranges
The digital twin made it possible to track how the cell’s internal architecture reorganizes. As RPE cells polarized, their internal scaffolding of actin and myosin fibers shifted from many small fragments to fewer, thicker strands that ringed the cell borders—like tightening a belt—to support the new tall shape. The nuclear envelope developed deep folds, and cells tended to end up with fewer nucleoli, signs of a more mature, stable gene expression program. Junction proteins that knit neighboring cells together moved from scattered locations inside the cell to well-defined bands along the side walls, strengthening the barrier. Meanwhile, energy-producing mitochondria and the endoplasmic reticulum, which helps process proteins and lipids, enlarged and migrated toward the nucleus, forming more coherent networks. Lysosomes, the cell’s recycling centers, shifted toward the central, upper region of the cell. When polarity was blocked, many of these relocations were incomplete or absent, and organelles stayed more randomly dispersed.
Conversations Between Organelles
The team also asked which organelles tend to occupy the same neighborhoods inside the cell, a sign that they may interact functionally. By overlaying organelle maps onto the average cell and calculating how strongly their positions correlated, they found that in well-polarized cells, structures grouped into coordinated clusters. For example, peroxisomes, mitochondria, endoplasmic reticulum, certain junctions, and parts of the cytoskeleton formed a tightly connected network linked to energy use and membrane remodeling. Lysosomes settled near the cell’s central organizing hub, the centriole, suggesting a role in controlling cilia and surface renewal. In contrast, when polarization was blocked, these spatial relationships weakened, and the overall “conversation” between organelles appeared fragmented. Machine-learning classifiers trained on these features indicated that the lateral distribution of mitochondria and the vertical placement of tight junctions were especially powerful markers of whether a cell was properly polarized.
Why This Matters for Vision and Disease
By fusing AI, large-scale imaging, and mathematical analysis, this work delivers a richly detailed 3D reference of how healthy human RPE cells organize themselves in space and time, and how that organization breaks down when polarization fails. For non-specialists, the takeaway is that the health of the retina depends not just on what molecules are present, but on where and when cellular components are arranged inside each support cell. The digital twin produced here offers a quantitative yardstick for spotting subtle defects in patient-derived RPE, opening new avenues for diagnosing early cellular problems in retinal diseases and for testing therapies that aim to restore proper cell structure before vision is irreversibly lost.
Citation: Ortolan, D., Sathe, P., Volkov, A. et al. AI driven 3D subcellular RPE map discovers cell state transitions in establishment of apical-basal polarity. npj Artif. Intell. 2, 20 (2026). https://doi.org/10.1038/s44387-026-00074-6
Keywords: retinal pigment epithelium, cell polarity, artificial intelligence, 3D cell mapping, age-related macular degeneration