Clear Sky Science · en
Preferential HER4 stimulation preserves neuregulin-induced improvement of myocardial function
New Hope for Failing Hearts
Heart failure affects millions of people worldwide and often keeps getting worse even with today’s best medicines. This study explores a new type of biological drug, called JK07, designed to help the heart repair itself while avoiding a serious worry: accidentally feeding hidden cancers. By tuning a natural healing signal in the heart and steering it away from cancer-linked pathways, the researchers aim to deliver the good without the bad.
How the Heart’s Own Repair Signal Works
The work centers on neuregulin‑1 (NRG‑1), a protein our bodies make that helps heart muscle cells grow, survive, and recover from damage. NRG‑1 sends its instructions through docking stations on cells called HER3 and HER4. When NRG‑1 lands on these receptors, they pair up with a partner called HER2 and launch internal survival and repair signals. Earlier animal and human studies with lab‑made NRG‑1 showed meaningful boosts in heart pumping strength, but they also brought side effects such as digestive upset and liver problems. Even more worrisome, HER3 is known to drive the growth of many cancers, raising fears that long‑term NRG‑1 treatment could awaken silent tumors.
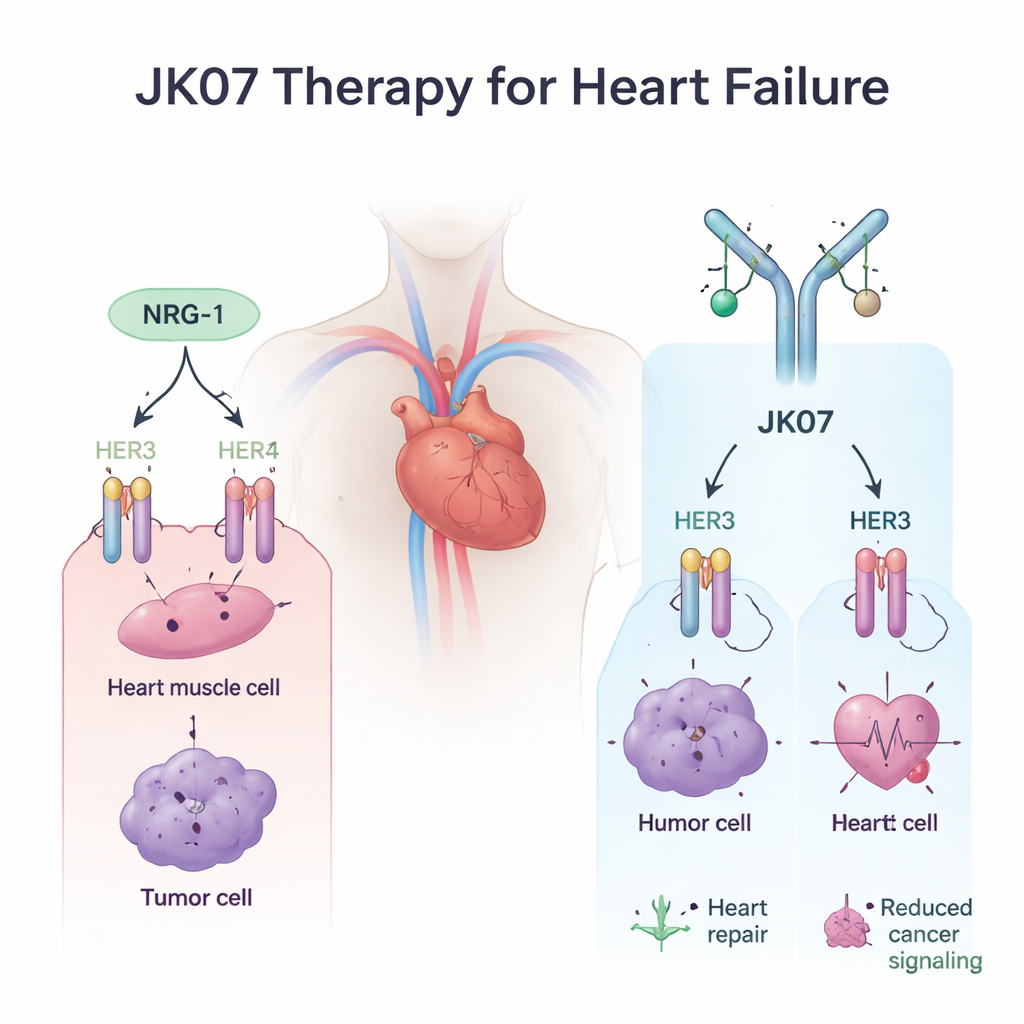
A Designer Molecule with a Safety Lock
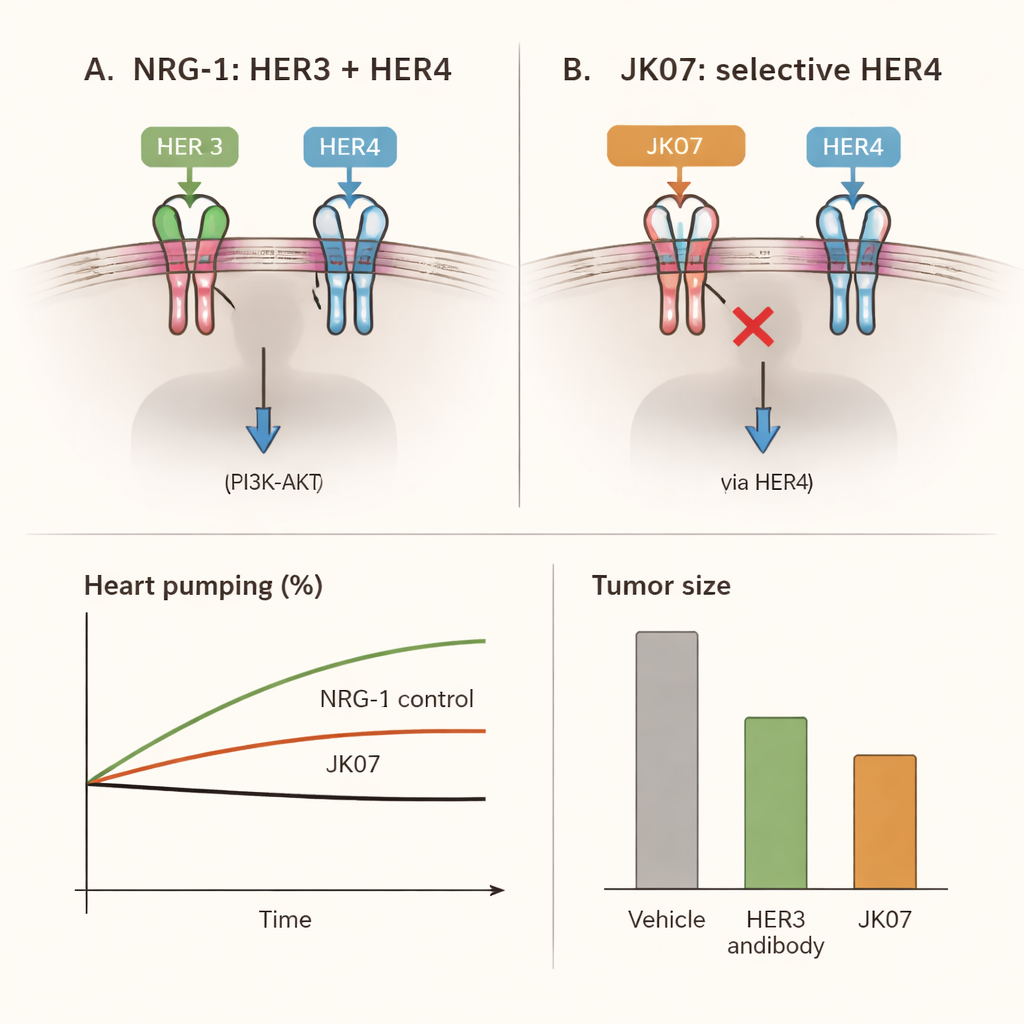
To solve this problem, the scientists engineered JK07, a fusion molecule with two carefully chosen parts. One end is a fully human antibody built to block HER3, the cancer‑linked receptor. The other end is the active piece of NRG‑1 that triggers helpful signals, attached in duplicate so it can still reach heart cells. In test‑tube experiments, JK07 firmly grabbed HER3 and at the same time kept the NRG‑1 fragment properly folded and active. In cancer cell systems where growth depends on HER3, JK07 sharply cut the signals that require HER3, slowed cell growth compared with plain NRG‑1, and in mice bearing HER3‑driven tumors it shrank tumors in a dose‑dependent way. These findings show that the antibody portion really does act as a safety lock on HER3.
Keeping the Heart‑Friendly Signal Alive
Crucially, JK07 still had to preserve the benefits of HER4, the receptor linked mainly to heart repair rather than cancer. Using specialized cell lines and heart muscle cells grown from human stem cells, the team showed that JK07 drove pairing of HER4 with HER2 and switched on a key protective pathway inside heart cells, known as AKT, just as strongly as unmodified NRG‑1. Rat heart cells responded in the same way. In other words, JK07 did not blunt the heart’s rescue signal; it simply redirected it away from HER3 and toward HER4.
From Rodents to Monkeys: What JK07 Does to the Heart
The researchers then asked whether this selective signal could actually rescue failing hearts. In rats given a surgically induced heart attack, heart pumping ability fell by about one‑third, closely mimicking human heart failure. Animals treated with JK07 several times over four weeks showed clear recovery: ejection fraction, a standard measure of how much blood the heart pushes out with each beat, rose by up to about one‑third compared with their pre‑treatment state. The heart’s enlarged chambers shrank toward normal size, scar‑related collagen in damaged areas decreased, and the microscopic structure of heart muscle looked more organized and healthy. These improvements matched those seen with a control NRG‑1‑based drug that does not block HER3, indicating that HER4 alone is enough to deliver the cardiac benefits.
Closer to the Clinic: Results in Monkey Heart Failure
To test JK07 in an animal model that resembles human disease more closely, the team studied middle‑aged rhesus macaques that had naturally developed long‑standing heart failure, including both the reduced‑pumping type (HFrEF) and the stiff‑heart type (HFpEF). In monkeys with weak pumping, two independent studies showed that weekly JK07 infusions led most animals to gain at least five percentage points in ejection fraction, with some approaching 20‑point absolute improvements—similar to animals treated with the established drug sacubitril/valsartan. In monkeys whose main problem was filling and relaxation rather than squeezing, JK07 improved the grade of diastolic dysfunction in most animals, whereas sacubitril/valsartan helped only rarely. Across studies, no major safety issues emerged at the doses tested.
What This Could Mean for Patients
For non‑specialists, the key message is that this study shows it may be possible to harness the heart’s own repair system in a more targeted and safer way. JK07 is built to stimulate the “good” receptor (HER4) that supports heart healing while blocking the “risky” receptor (HER3) tied to cancer. In both rodents and monkeys, this selective approach improved heart pumping, reduced structural damage and scarring, and even showed anti‑tumor activity in a cancer model. While human trials are still needed to confirm benefits and long‑term safety, JK07 points toward a future in which treatments for heart failure not only work better but do so with a lower risk of fueling cancer.
Citation: Murphy, S.L., Tang, W.H.W., Zhuang, X. et al. Preferential HER4 stimulation preserves neuregulin-induced improvement of myocardial function. npj Drug Discov. 3, 10 (2026). https://doi.org/10.1038/s44386-026-00038-5
Keywords: heart failure, neuregulin-1, HER4, biologic therapy, cardiac remodeling