Clear Sky Science · en
Developing a protocol to counteract spontaneous in vitro activation of intestinal fibroblasts using design of experiments
Why keeping gut repair cells calm matters
In chronic gut diseases like Crohn’s, scar tissue can slowly narrow the intestine until food can barely pass, often leading to surgery. This scarring, called fibrosis, is driven by support cells known as fibroblasts that become overactive and lay down too much connective tissue. To understand and one day prevent this process, scientists rely on lab-grown fibroblasts. But there is a catch: the simple plastic dishes used in most labs push these cells into an unnaturally activated state all by themselves, making it hard to study the earliest steps that lead from a healthy to a fibrotic gut.
When the lab dish sends the wrong signal
The authors first showed how quickly intestinal fibroblasts get stirred up on standard tissue-culture plastic. Within just 24 hours, both an established cell line and primary cells from male and female donors switched from a quiet state to an activated, muscle-like form. They produced high levels of proteins associated with contraction and scarring, such as α-smooth muscle actin and fibroblast activation protein, even without any added inflammatory triggers. This means many existing lab models may be starting from a state that already resembles late-stage disease rather than early, reversible changes.
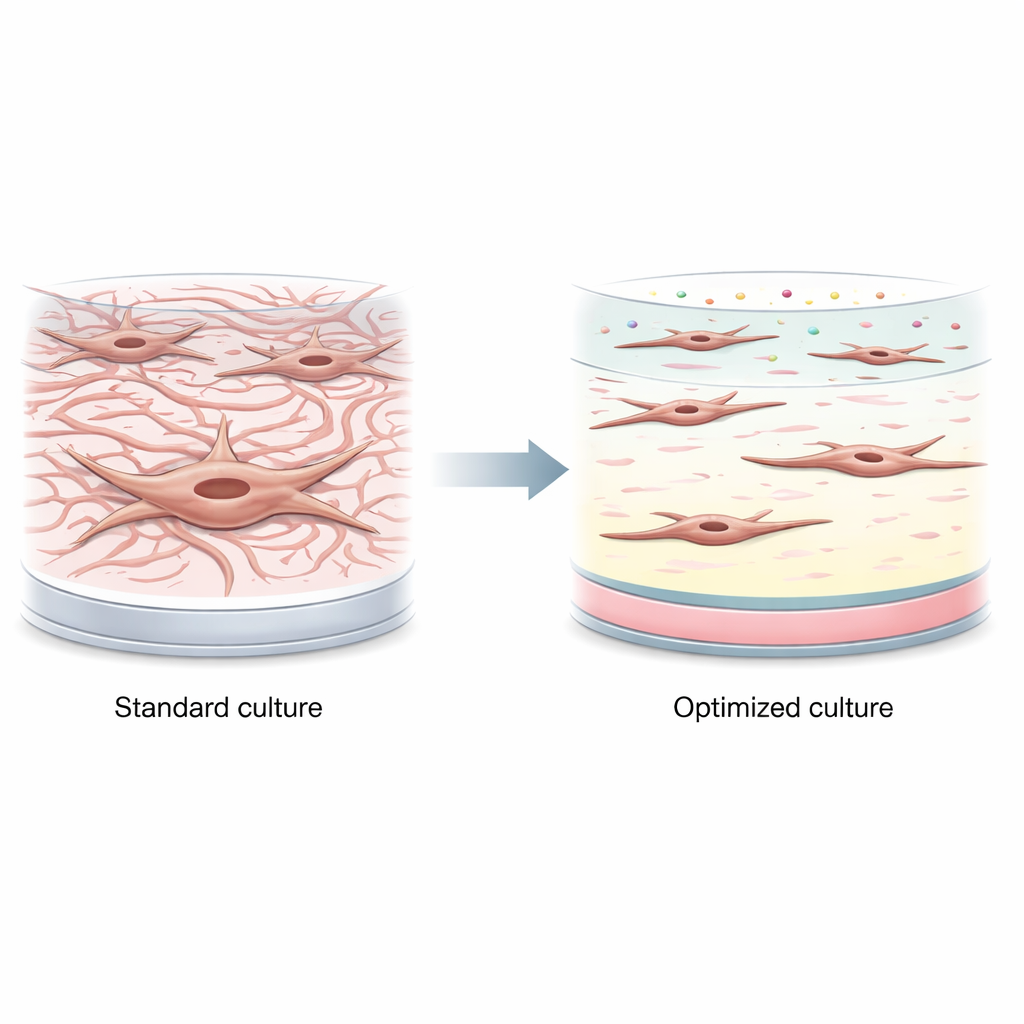
Testing single ingredients is not enough
To calm the cells back down, the team explored components of the fibroblasts’ natural surroundings in the gut, including key scaffolding proteins (collagen types I and III, and laminin) and soluble factors (vitamin D and basic fibroblast growth factor, or FGF). They tried these one at a time, either as surface coatings or as additives to the culture medium. Some conditions nudged activation markers downward: for example, moderate doses of vitamin D lowered a major scarring marker several-fold, and certain coating densities had mild effects. But these benefits were offset by problems such as reduced cell survival at higher vitamin D levels, and overall the single tweaks did not reliably keep fibroblasts in a low-activity state.
Letting statistics search the space of possibilities
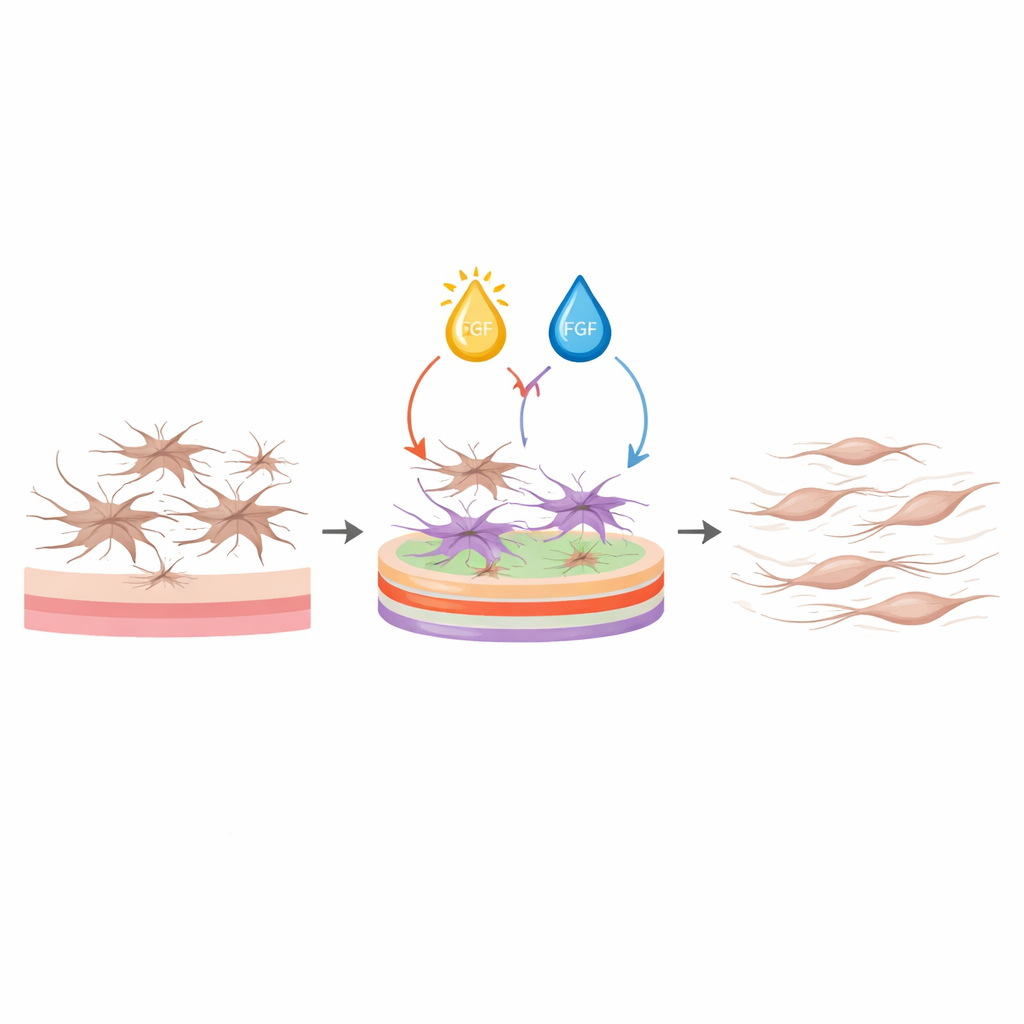
Instead of adjusting one knob at a time, the researchers used a formal “design of experiments” approach to search many combinations efficiently. They varied five inputs—the three matrix proteins plus vitamin D and FGF—across a wide range of doses, generating 42 carefully chosen culture conditions. For each, they measured four outcomes linked to scarring: a key activation marker, secretion of a major matrix protein (fibronectin), cell growth, and cell survival. Statistical models then revealed which factors, and which pairings between them, had the biggest influence. Vitamin D emerged as a powerful but dose-sensitive player, affecting all readouts, while FGF tended to lower activation and support cell growth and survival. Collagen I, collagen III, and laminin shaped how these soluble cues were interpreted, with certain combinations dampening activation far more than any single factor could.
Building a recipe for calm but healthy fibroblasts
Using these models, the team asked the software to identify conditions that would simultaneously lower activation markers and matrix production while keeping the cells alive, without insisting that proliferation be low. The best recipe combined a high density of collagen I, low collagen III, and high laminin on the culture surface, together with mid-range vitamin D and high FGF in the medium. Under this protocol, fibroblasts stayed slim and spindle-shaped instead of spread and star-like, expressed much lower levels of genes and proteins linked to contraction and fibrosis, and secreted far less collagen and fibronectin. At the same time, their viability remained as high as, or better than, that of cells on plain plastic. Importantly, these effects held not only for the model cell line but also for primary fibroblasts from both male and female human donors, showing the protocol is robust.
What this means for future gut fibrosis research
By carefully tuning the “neighborhood” around intestinal fibroblasts—both the surface they attach to and the factors bathing them—this study delivers a straightforward protocol that keeps these cells in a non-activated, yet healthy, state using standard lab materials. For researchers, this provides a clean starting point to add specific inflammatory or mechanical cues and watch how fibrosis begins, rather than working with cells that are already scar-like. More broadly, the work demonstrates how statistical design can uncover subtle, non-intuitive ingredient combinations that tame complex cell behaviors, paving the way for better models of scarring and, eventually, more targeted strategies to prevent or reverse gut fibrosis.
Citation: Mohammadalizadeh, Z., Sudi, S., Basto Moreno, D.A. et al. Developing a protocol to counteract spontaneous in vitro activation of intestinal fibroblasts using design of experiments. npj Biomed. Innov. 3, 25 (2026). https://doi.org/10.1038/s44385-026-00079-5
Keywords: intestinal fibrosis, fibroblast activation, cell culture protocol, vitamin D and FGF, design of experiments