Clear Sky Science · en
Use of in vitro co-culture models to inform bacterial engineering for the treatment of solid tumours
Why bacteria in cancer might matter to you
Cancer treatments like surgery, chemotherapy, and radiotherapy save lives but often come with harsh side effects and, too often, treatment failure. An intriguing new idea is to enlist living bacteria as precision tools that can find and attack solid tumors from the inside. This article explains how scientists are building laboratory test systems—where human cells and bacteria grow together—to design safer, smarter bacterial therapies before they ever reach patients.
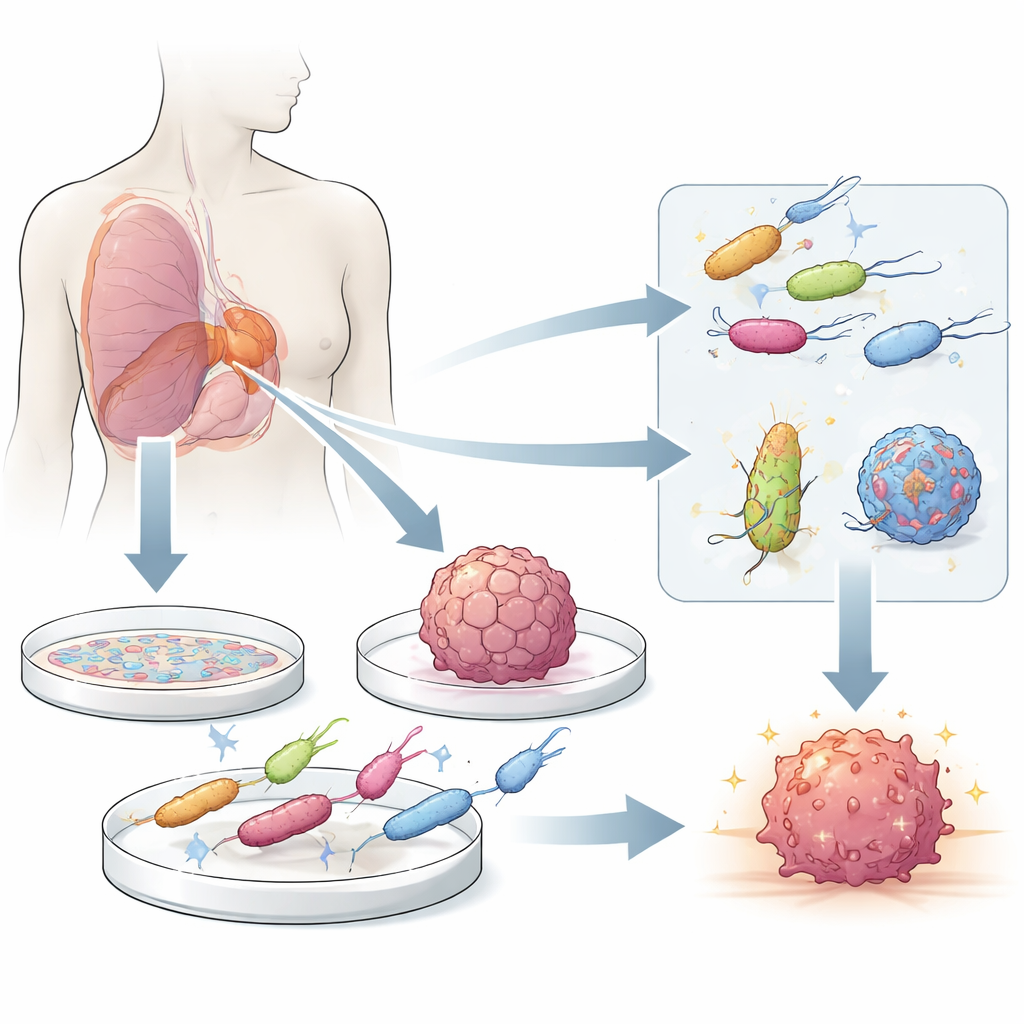
Tiny helpers that seek out tumors
Certain bacteria naturally prefer the harsh, oxygen-poor environment inside solid tumors over healthy tissues. After slipping through leaky tumor blood vessels or hitching a ride with immune cells, these microbes move toward dead and dying cells in the tumor core, where they can grow and sometimes trigger cancer cell death and local immune attacks. Modern genetic engineering turns these natural "tumor-homing" strains into living delivery vehicles that can release drugs, immune stimulants, or other payloads directly where they are needed most, while efforts to weaken their harmful traits improve safety.
Building realistic lab mini-tumors
To understand and improve these living medicines, scientists need testbeds that mimic what happens in the human body as closely as possible. Simple flat cell layers are easy to grow and useful for quick screening, but they miss the complex structure and oxygen gradients of real tumors. Three-dimensional models, such as spheroids made from cancer cell lines and organoids grown from patient tissue, better reproduce key features like a living outer shell, a quiet inner zone, and a dead core. When bacteria are mixed directly with these mini-tumors, researchers can watch how they invade, where they accumulate, and how well engineered drug circuits work in conditions that resemble a patient’s tumor.
Separating cells to study signals
Other co-culture designs focus less on physical contact and more on the invisible chemical exchanges between bacteria and human cells. In transwell systems, a porous membrane divides two chambers so that small molecules pass through while cells stay put. This lets scientists test, for example, how a bacterial strain converted a harmless compound into a tumor-killing gas that diffused across to nearby cancer cells. Microfluidic chips—tiny devices with channels and compartments—add another layer of realism. They can control fluid flow, oxygen levels, and timing, allowing engineered bacteria to grow, burst in synchronized cycles, and repeatedly bathe cancer cells in therapeutic molecules, closely tracking how both partners respond over time.
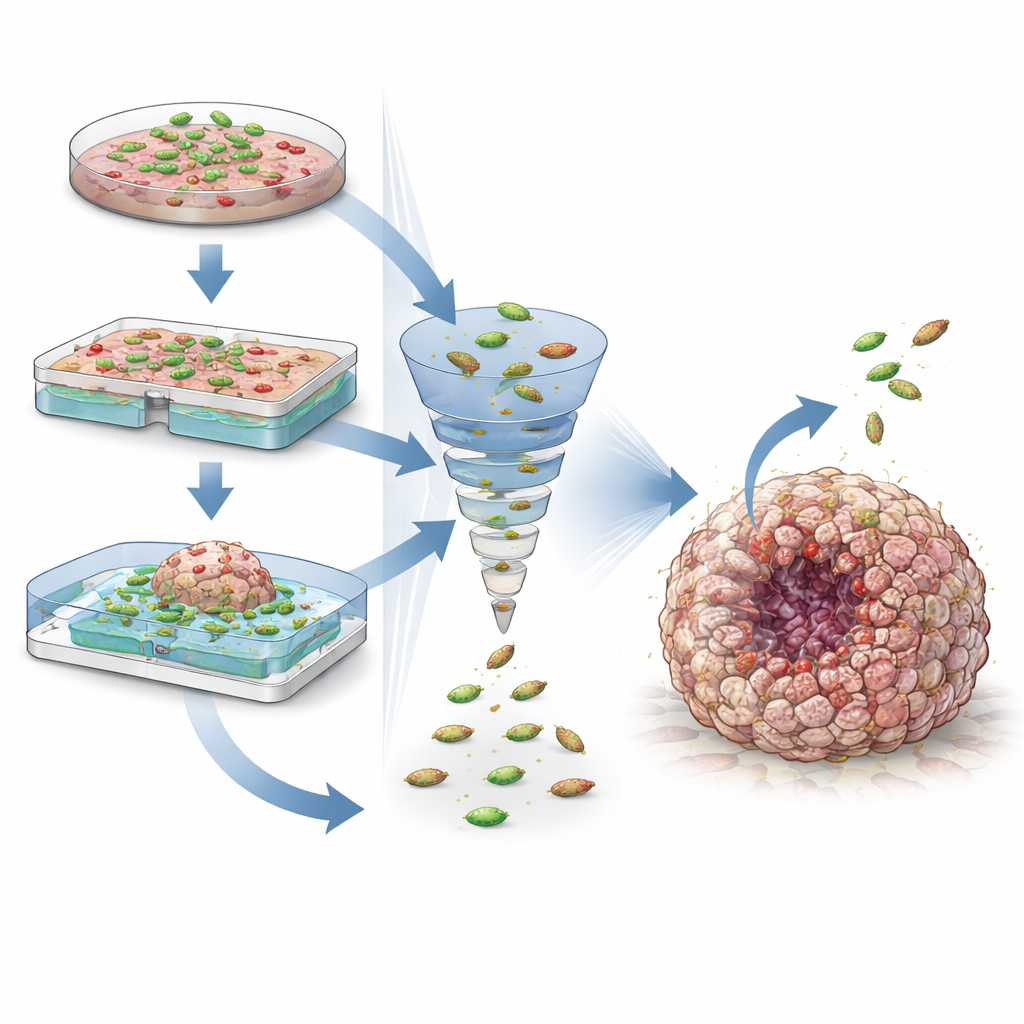
Peering inside the tumor ecosystem
Co-culture models are also being used to study how bacteria interact with the broader tumor ecosystem. Organoids derived from individual patients can preserve many of the genetic quirks and drug responses of their original tumors, opening the door to personalized testing of bacterial therapies. Adding immune cells to these cultures reveals how bacteria can boost or dampen anti-cancer immunity and how they might combine with checkpoint inhibitor drugs. At the same time, these models flag safety concerns, such as bacterial toxins that damage DNA and could help trigger cancer in the first place. By adjusting culture conditions, oxygen levels, and measurement methods—from live imaging to DNA sequencing—researchers can systematically explore these benefits and risks.
From lab bench to bedside
Despite exciting animal results, very few bacterial cancer therapies have shown clear benefits in human trials. The authors argue that smarter use of co-culture systems can bridge this gap. By better capturing patient-to-patient differences, tumor complexity, and prior treatments, these models can identify which engineered strains are most promising, how to combine them with existing drugs, and where they might fail. They also highlight the need for shared standards so that labs around the world can compare results and regulators can judge safety and effectiveness more confidently. In plain terms, carefully designed lab mini-tumors grown side-by-side with bacteria offer a powerful way to refine these “smart bugs,” increasing the chances that future patients will receive bacterial therapies that are both safer and more effective.
Citation: Foschini, S., Wright, J.A., Woods, S.L. et al. Use of in vitro co-culture models to inform bacterial engineering for the treatment of solid tumours. npj Biomed. Innov. 3, 21 (2026). https://doi.org/10.1038/s44385-026-00075-9
Keywords: bacterial cancer therapy, tumor organoids, co-culture models, synthetic biology, tumor microenvironment