Clear Sky Science · en
3D biofabricated in vitro models as new approach methodologies for animal alternatives
Rethinking How We Test New Medicines
For nearly a century, most new drugs have been tested in animals before reaching human trials. Yet more than 9 out of 10 drug candidates that look safe in animals still fail in people. This article explores how a new generation of 3D "bioprinted" human tissues could change that story—offering more accurate, humane ways to predict how our bodies respond to medicines, and potentially speeding safer treatments to patients.

From Lab Animals to Human-Centered Testing
Recent US legislation, known as the FDA Modernization Act 2.0, has removed the automatic requirement that every new drug be tested in animals. Instead, regulators can now accept "New Approach Methodologies"—human-relevant test systems designed to better forecast real patient responses. Among these, 3D bioprinting stands out. It uses specialized printers to place living human cells and soft, gel-like materials into intricate shapes that mimic real tissues and mini-organs. The process often begins with medical images such as CT or MRI scans, which provide a blueprint. Scientists then choose suitable biomaterials, mix in the right types of cells to create a printable "bioink," and print layered or volumetric structures that mature into functional tissue models in bioreactors.
How 3D Bioprinting Builds Living Tissues
Bioprinting is not a single technology but a family of methods. Inkjet-style printers spray tiny droplets of cell-containing fluid to build thin layers with fine detail, making them useful for miniature tissues such as skin patches or lung barriers. Extrusion printers squeeze continuous strands of thicker bioinks, which can include many cells and supporting fibers, allowing construction of larger, more robust structures such as heart valves, liver tumor models, or vascular networks. A newer class, called vat photopolymerization, shines patterned light into a liquid to solidify complex shapes without squeezing cells through nozzles. Variants of this method can sculpt micron-scale features, print an entire small organ in seconds, or even shape tissues inside a transparent bath, all while keeping cells alive.
Replacing and Refining Drug Tests
These printed tissues are already being used to rethink the drug pipeline. In early discovery, patient- or disease-specific constructs—such as bioprinted tumor models—let researchers test many drug candidates in realistic 3D environments that better resemble human disease than flat cell layers in a dish. In preclinical testing, printed skin, lung, and liver tissues are proving more accurate than animals for spotting human-specific toxicity or side effects. Some studies go further by printing "clinical trials on a chip," where tissues made from cells of different donors are exposed to a drug in parallel, revealing who might benefit or be harmed. Regulators now encourage companies to submit data from these models alongside animal studies, building up the evidence needed for broader acceptance.
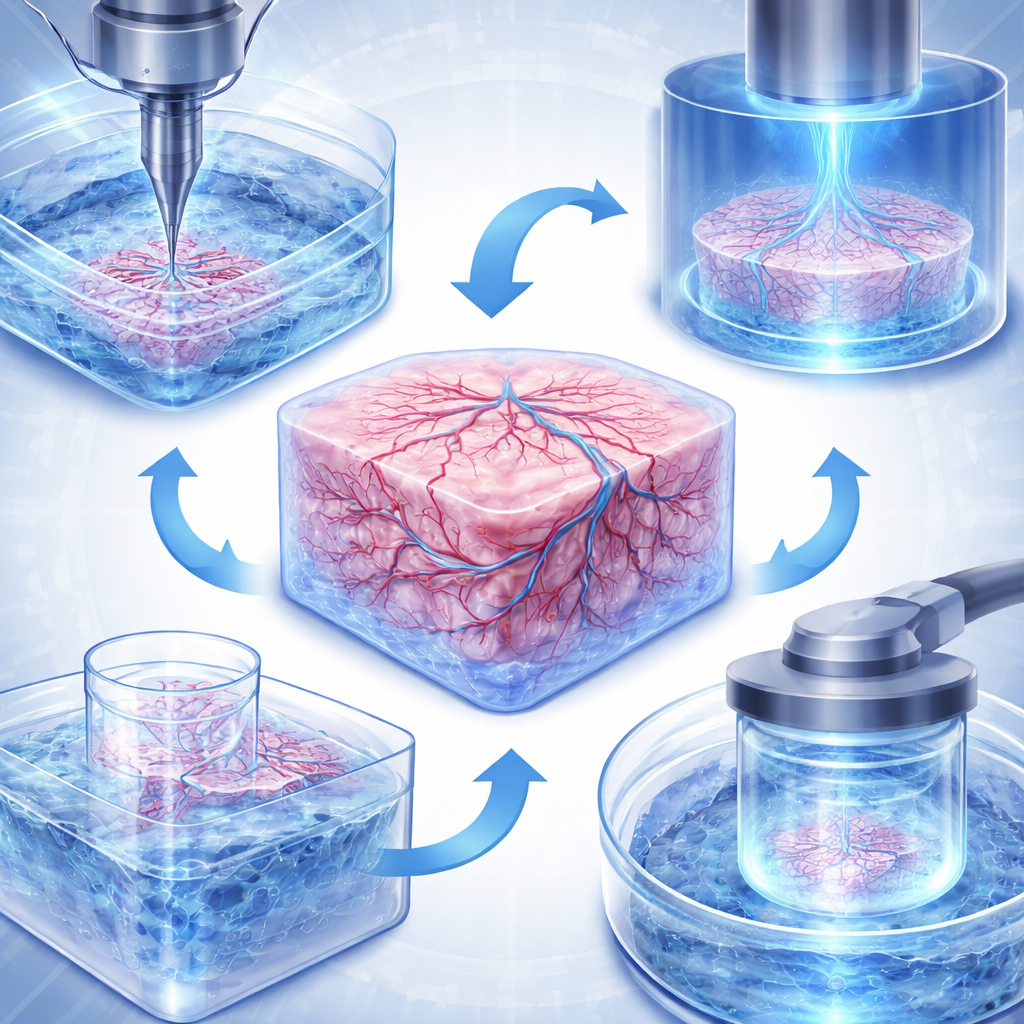
Engineering Organs Without a Body
Despite rapid progress, several hurdles remain before bioprinted tissues can routinely stand in for animal tests. One major challenge is blood supply: real organs contain vessel networks that range from large arteries down to hair-thin capillaries, while printed constructs must overcome a natural limit on how far oxygen and nutrients can diffuse. Researchers are tackling this with new printing strategies that create branching channels, with support baths that hold soft tissues in place while tiny vessels are formed, and with lightning-fast volumetric printers that make thick, cell-dense structures before cells begin to suffer. Another challenge is realism: living tissues are mixtures of many cell types bathed in complex chemical signals and, often, influenced by the microbiome and distant organs. Advanced models now combine multiple cell populations, controlled gradients of oxygen and pH, and even linked "multi-organ" systems that connect gut, liver, immune, and brain tissues through flowing fluid to mimic whole-body responses.
A Humane, Predictive Future for Drug Development
Taken together, these advances suggest a future in which high-fidelity, human-based tissue models become central to how we evaluate medicines. The article concludes that 3D bioprinting is moving from a niche laboratory technique to a core tool for building standardized, regulator-ready test systems. To fully realize this promise, scientists and regulators must agree on where and how these models should be used, prove that they reliably predict human outcomes, and solve remaining technical challenges such as building stable vasculature and anatomically complex organs. If successful, this shift could reduce reliance on animal testing, cut costly late-stage drug failures, and deliver safer, more effective therapies while better reflecting the biology that matters most: our own.
Citation: Hua, W., Gaharwar, A.K. 3D biofabricated in vitro models as new approach methodologies for animal alternatives. npj Biomed. Innov. 3, 20 (2026). https://doi.org/10.1038/s44385-026-00073-x
Keywords: 3D bioprinting, animal testing alternatives, drug development, tissue models, volumetric bioprinting