Clear Sky Science · en
Engineering macrophages for cancer immunotherapy: emerging insights and therapeutic potential
Turning the Body’s Cleanup Crew into Cancer Fighters
Our bodies are full of macrophages—roaming “cleanup” cells that swallow germs and dead tissue. This review article explores how scientists are learning to redirect these cells against cancer. Because macrophages are plentiful inside tumors, including those that resist today’s leading immunotherapies, understanding and engineering them could open new options for patients with hard-to-treat cancers.
The Many Faces of a Single Cell Type
Macrophages are not a single fixed cell type; they are shape-shifters. They can arrive from the bloodstream or already live in tissues, and once settled they respond to local signals. In tumors, these tumor-associated macrophages can either help kill cancer cells or, confusingly, help tumors grow and spread. Earlier, researchers tried to squeeze them into two boxes—“M1” (attack mode) and “M2” (repair or support mode). New genetic and spatial mapping tools now show a much richer spectrum of states, with multiple subgroups and behaviors that vary from one tumor and location to another. Even cells once thought to be firmly “pro-tumor” can, in some contexts, rally T cells and support anti-cancer immunity.
Feeling Their Surroundings and Eating Cancer Cells
Macrophages do not just listen to chemical signals; they also feel the physical world around them. As they crawl through a tumor’s dense mesh of proteins, squeeze between cells, and endure fluid forces, they sense stiffness and strain through specialized receptors such as Piezo1. These mechanical cues, together with classic immune signals, can tilt macrophages toward more inflammatory or more calming roles. Functionally, macrophages can engulf cancer cells, then chop them up and display fragments (antigens) to T cells, bridging innate and adaptive immunity. Cancer cells fight back by sending “don’t eat me” signals, notably through a surface molecule called CD47 that binds a brake on macrophages. Drugs that block this interaction are in many clinical trials, boosting cancer cell clearance but needing careful tuning to avoid harming healthy cells.
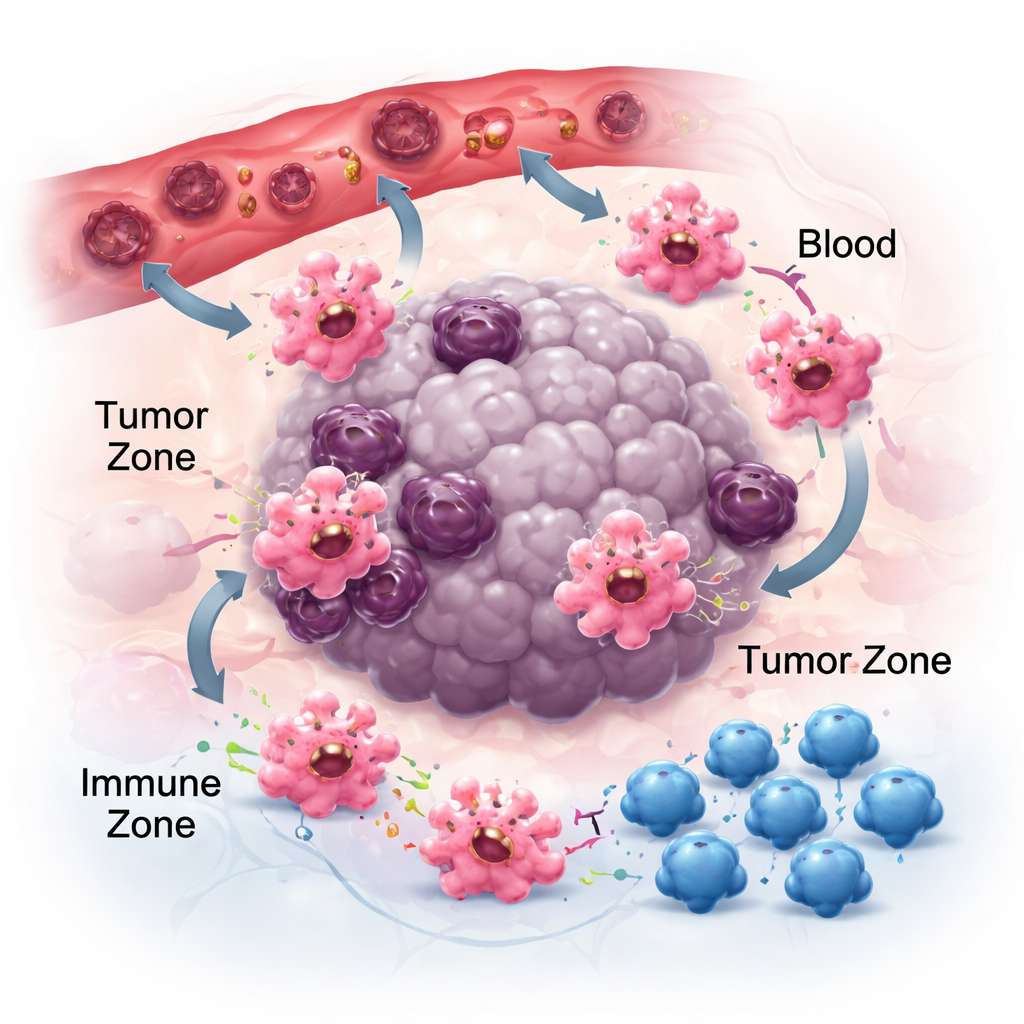
Moving, Sticking, and Talking in the Tumor Neighborhood
To matter in cancer, macrophages must first reach tumors and then position themselves in the right niches. They follow chemical trails—chemokines and growth factors—released by cancer and surrounding cells. Pairs such as CCR2–CCL2 help draw them into tumors, and drugs that block these signals can cut macrophage buildup, though not completely, pointing to other attractants like CSF-1 and VEGF. Once in place, how macrophages stick to the local matrix also shapes their behavior. Their adhesion receptors, called integrins, don’t just act as glue; they amplify inflammatory signals and can nudge cells toward more tumor-fighting states. At the same time, macrophages shed tiny packages called extracellular vesicles that carry RNA and proteins to nearby cells. Depending on the tumor type, these vesicles can either blunt immunity and aid spread or bolster anti-tumor responses, highlighting the context-dependent nature of macrophage communication.
Reprogramming Macrophages with Genes and Materials
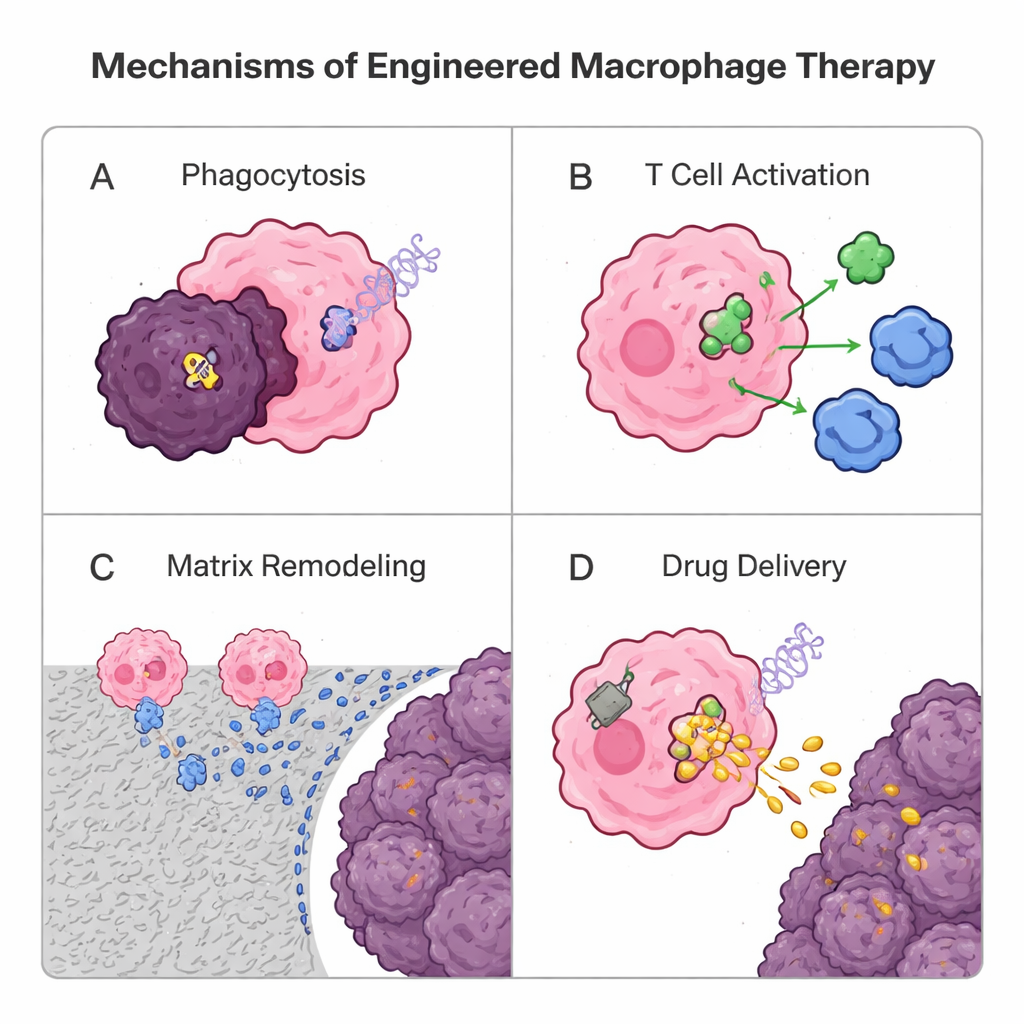
Because macrophages naturally infiltrate solid tumors better than many T cells, researchers are now re-engineering them as living medicines. One genetic strategy borrows from chimeric antigen receptor (CAR) T cell therapy: adding a synthetic receptor to create CAR macrophages (CAR-M). These designer cells recognize tumor markers, engulf cancer cells more efficiently, digest the surrounding matrix, release inflammatory signals, and activate T cells by presenting tumor antigens. Early human trials of HER2-targeted CAR-Ms show encouraging safety and biological activity, although more work is needed to prove strong, lasting benefits. In parallel, non-genetic approaches use particles and “backpacks” made from lipids or polymers. Macrophages can swallow drug-loaded nanoparticles and carry them deep into tumors like Trojan horses, or wear surface-attached patches that slowly release immune-boosting signals, helping them maintain a tumor-fighting identity without overloading their interior. Macrophage-derived vesicles themselves are also being tested as natural, cell-free delivery vehicles.
Challenges Ahead and Reasons for Hope
Despite their promise, macrophage therapies must overcome practical hurdles. It is time-consuming and costly to generate enough consistent cells from patients, and once infused, these cells do not live or multiply indefinitely. Their built-in flexibility, while biologically useful, can make it hard to keep them locked in an anti-tumor mode inside a complex, suppressive tumor microenvironment. The authors argue that advanced bioengineered models—3D cultures, organoids, and organ-on-a-chip systems that mimic real tissue mechanics—will be crucial for understanding and predicting how engineered macrophages behave in patients. Even so, their natural ability to enter solid tumors, eat cancer cells, and awaken T cells, combined with an early track record of safety, suggests that macrophage-based therapies could become an important new weapon, especially against “cold” tumors that currently resist most immune treatments.
Citation: Block, A., Liu, X., Zhang, D. et al. Engineering macrophages for cancer immunotherapy: emerging insights and therapeutic potential. npj Biomed. Innov. 3, 18 (2026). https://doi.org/10.1038/s44385-026-00072-y
Keywords: macrophage immunotherapy, tumor microenvironment, CAR macrophages, phagocytosis, nanoparticle drug delivery