Clear Sky Science · en
Nucleic acid-based therapeutics to restore joint homeostasis in age-related and post-traumatic arthritis
Why Aching Joints Need New Kinds of Medicine
Osteoarthritis, the most common form of arthritis, is often dismissed as simple “wear and tear,” but it is actually a complex disease where the joint’s natural balance between damage and repair breaks down. As people live longer and stay active later in life, painful, stiff joints are becoming a major cause of disability, and current treatments mostly mask symptoms or end in costly joint replacement surgery. This review article explores a new class of treatments built from genetic instructions—tiny strands of RNA—that aim not just to ease pain, but to restore a healthy balance inside damaged joints.
From Worn Cartilage to Whole-Joint Breakdown
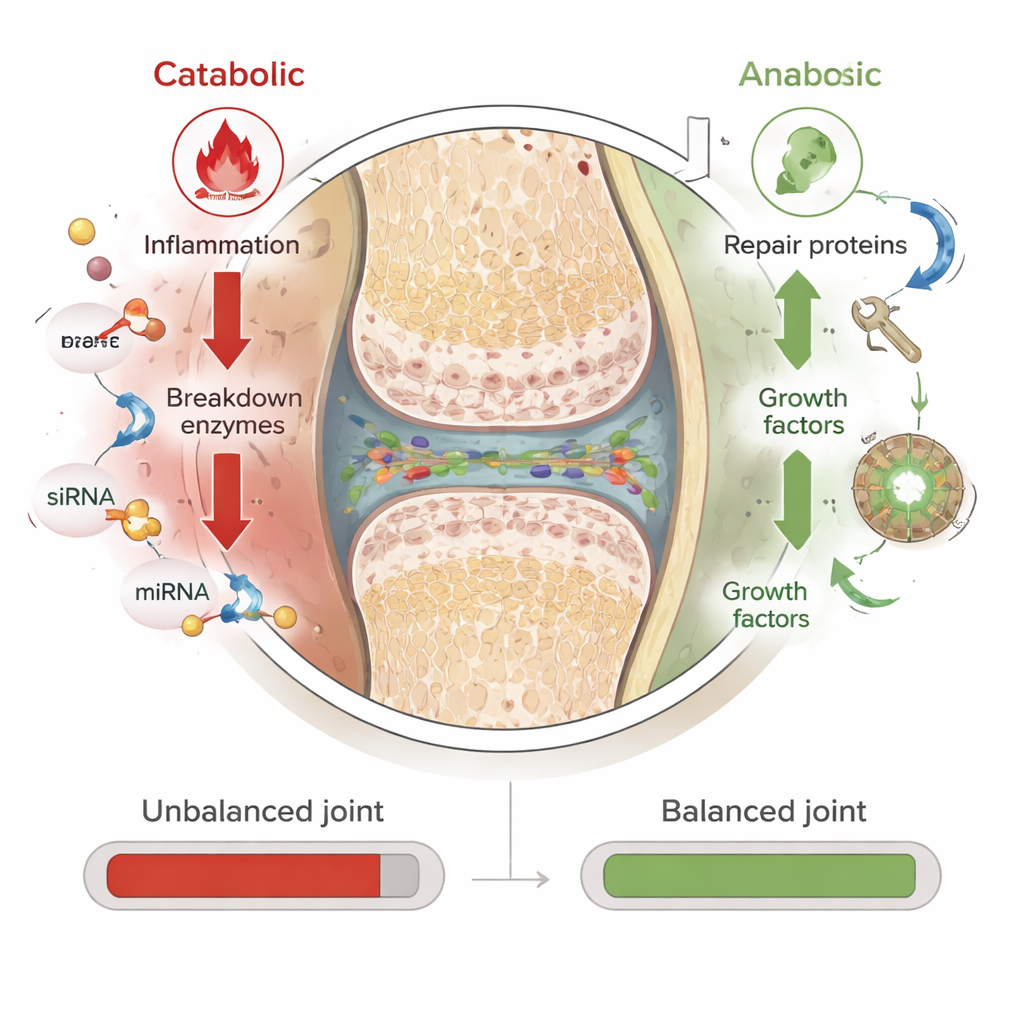
Osteoarthritis is now recognized as a whole-joint disorder, not just a problem of thinning cartilage. In an arthritic knee, for example, the smooth cartilage that cushions bones wears away, the meniscus can fray, the bone underneath thickens, and the joint lining becomes inflamed. This low-grade, chronic inflammation quietly feeds the disease for years. The authors describe how age, injury, excess body weight, and genetics all interact to tip the joint toward “catabolic” processes that break tissue down, overwhelming the “anabolic” processes that repair it. Because each patient’s disease is driven by a slightly different mix of factors, responses to common treatments like painkillers, injections, or even surgery vary widely, and no drug today can truly stop or reverse osteoarthritis.
Why Traditional Treatments Fall Short
The review outlines many current options—from over-the-counter pain pills and steroid shots to physical therapy, braces, and advanced procedures like cartilage implants. Each has benefits, but also clear drawbacks. Pain relievers can irritate the stomach or heart, steroids may damage cartilage with repeated use, and regenerative approaches such as platelet-rich plasma or stem cells are expensive and lack long-term proof. Most importantly, nearly all existing drugs tackle only one side of the problem: they either try to calm inflammation and slow breakdown, or they try to boost repair. Few approaches attack both sides of the see-saw at once, which is likely why they struggle to change the course of the disease.
RNA Medicines: Rewriting the Joint’s Internal Script
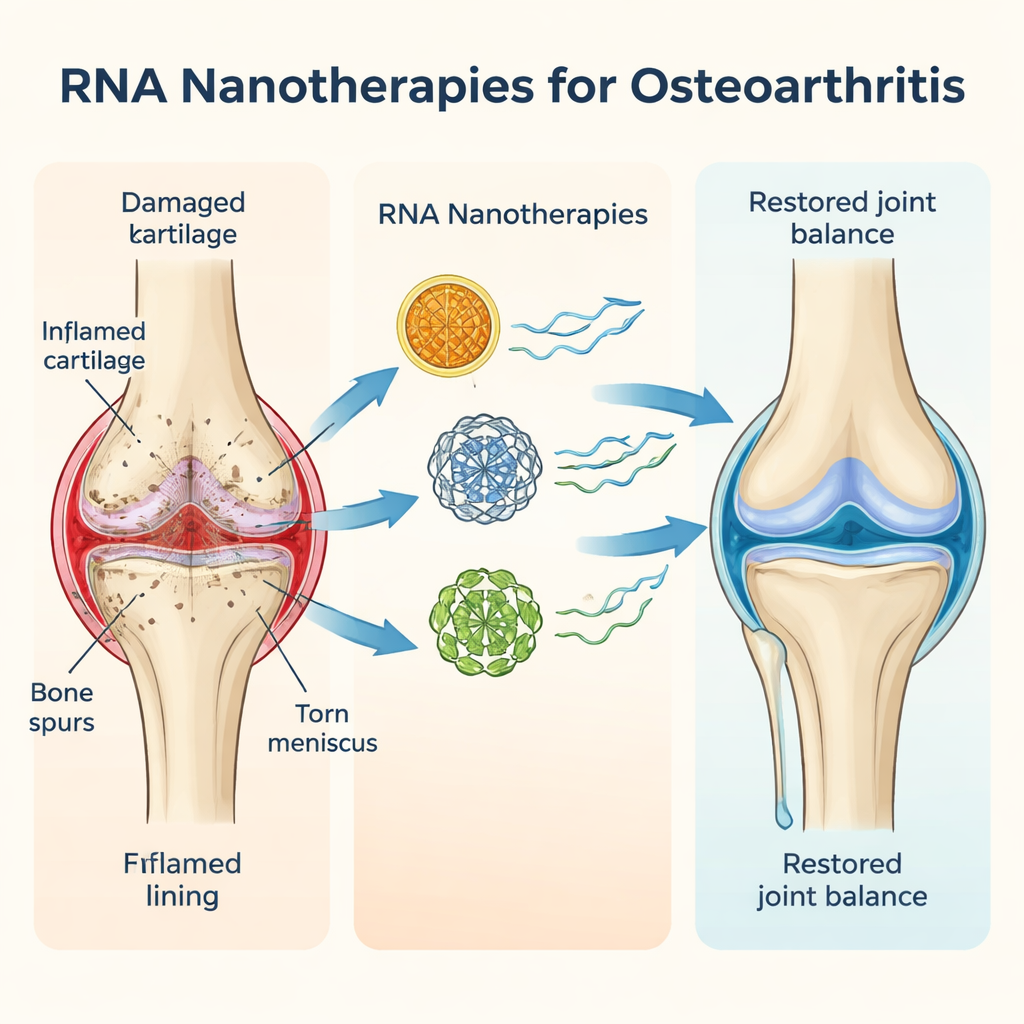
RNA-based therapies work at a different level. Instead of targeting proteins after they are made, they act on the short-lived genetic messages that tell cells which proteins to build. The authors focus on three types. Small interfering RNA (siRNA) can silence a single harmful gene with high precision. MicroRNA (miRNA) can fine-tune entire networks of genes, nudging multiple damaging pathways down at once. Messenger RNA (mRNA) does the opposite: it supplies the instructions to make helpful proteins, such as growth factors that stimulate cartilage repair or molecules that block inflammatory signals. Because RNA acts only temporarily and does not alter DNA, its effects are reversible and, in principle, safer than permanent gene editing. Building on the success of COVID-19 mRNA vaccines, researchers are now testing similar ideas in animal models of arthritis.
Nanoparticles: Getting Fragile Messages to the Right Cells
A major challenge is that naked RNA is fragile and cannot easily slip into cells. The review describes how scientists are packaging RNA inside protective “nanoplatforms”—tiny carriers made from fats, polymers, or short peptides. These nanoparticles shield RNA from breakdown, help it pass through the dense cartilage matrix, and encourage cells in the joint lining and cartilage to take it up. In preclinical studies, siRNA-loaded particles have been used to shut down genes that drive inflammation, oxidative stress, and tissue breakdown, while mRNA-loaded particles have boosted protective proteins that promote lubrication and repair. Early experiments show less cartilage damage and less pain in animal models, suggesting that carefully designed combinations of anti-breakdown and pro-repair RNAs might one day act as true disease-modifying therapies.
Hurdles on the Road to the Clinic
Despite their promise, RNA nanomedicines for osteoarthritis still face important obstacles. The immune system can mistake both the RNA and its carrier for invaders, triggering unwanted inflammation in joints that are already irritated. Injections into the joint are cleared in days to weeks, so long-lasting benefits will likely require smarter slow-release systems or repeated dosing. Manufacturing high-quality RNA and nanoparticles at scale is costly, and regulators have limited experience evaluating such drugs for slowly progressing joint diseases. Finally, because osteoarthritis is so variable from person to person, doctors will need better ways to match specific RNA cocktails to each patient’s unique pattern of joint damage and inflammation.
What This Means for People with Arthritis
In simple terms, this article argues that to truly help people with osteoarthritis, future treatments must do more than cushion pain—they must restore the joint’s lost balance by turning down the genes that drive damage and turning up the genes that support repair. RNA-based medicines, delivered by sophisticated nanoparticle carriers, offer a flexible toolkit to do just that. While they are not yet ready for the clinic and important safety, durability, cost, and regulatory questions remain, the authors see them as a realistic pathway toward personalized, minimally invasive therapies that could delay or even prevent the need for joint replacement in age-related and injury-triggered arthritis.
Citation: Rai, M.F., Pham, C.T., Hou, K. et al. Nucleic acid-based therapeutics to restore joint homeostasis in age-related and post-traumatic arthritis. npj Biomed. Innov. 3, 17 (2026). https://doi.org/10.1038/s44385-026-00068-8
Keywords: osteoarthritis, RNA therapeutics, nanomedicine, joint regeneration, siRNA mRNA miRNA