Clear Sky Science · en
Accelerating personalized medicine: miniaturized patient-derived organoid drug screening for predicting cancer treatment responses and beyond
Bringing the Lab to the Bedside Faster
For many people with advanced cancer, waiting weeks to learn whether a chemotherapy will work can feel agonizing. This study explores a way to speed up that timeline using tiny, lab-grown replicas of a patient’s tumor—called organoids—to test drugs in advance. By shrinking and automating the testing process, the researchers aim to predict which treatments are likely to help, using far less tissue and time than current methods require.
Mini Tumors in a Dish
Patient-derived organoids are three-dimensional clusters of cells grown from a person’s own tumor. Unlike traditional flat cell cultures, these mini tumors keep much of the original cancer’s genetic diversity and behavior. Earlier work has shown that when organoids respond—or fail to respond—to a drug in the lab, patients often show a similar response in the clinic. The problem is that standard organoid drug tests use hundreds of organoids per well, demanding a lot of biopsy material and taking many weeks to complete, which limits their real-world use for patients who need to start treatment quickly.
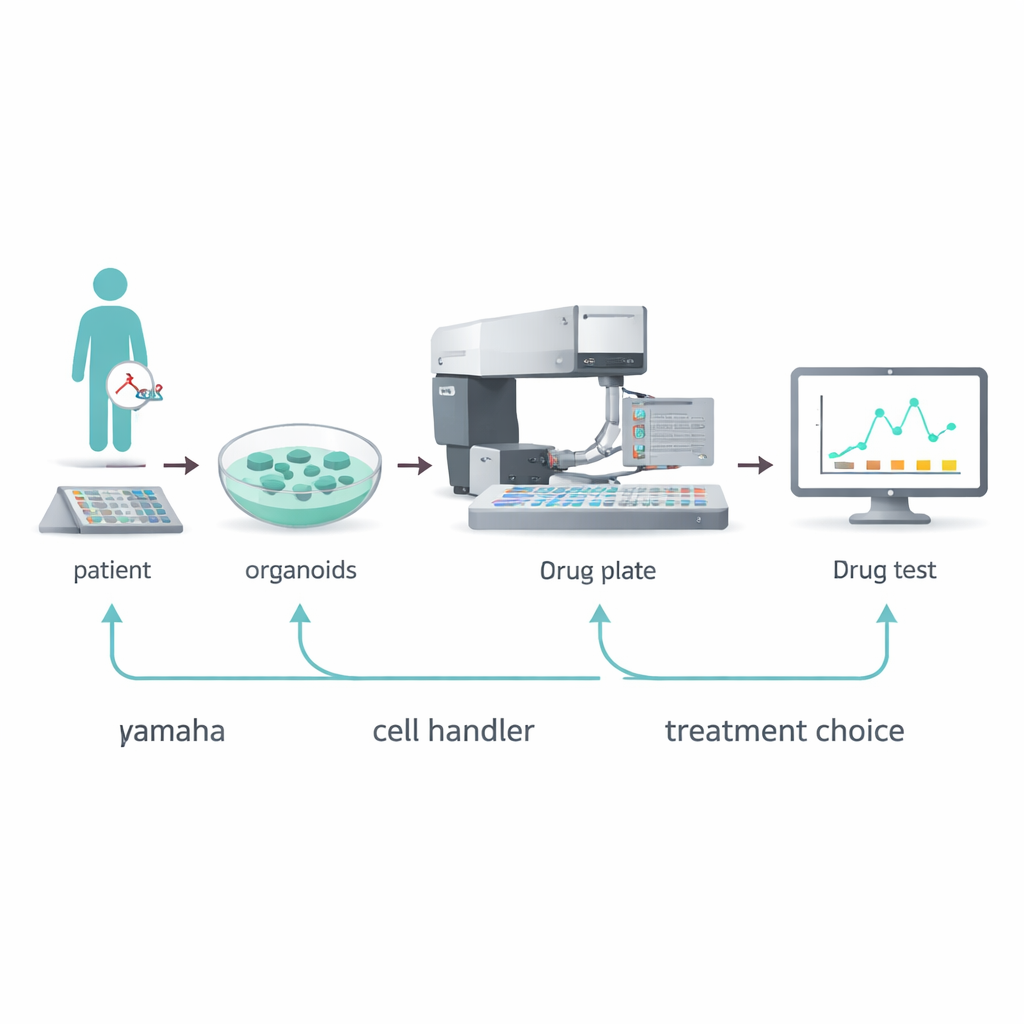
A Robotic Picker for Tiny Tumors
The team turned to an automated device, the Yamaha Cell Handler, to tackle these bottlenecks. This robot uses a camera to spot individual organoids in a source plate, analyzes their size and shape, then gently picks up selected organoids and deposits them into the tiny wells of a 384-well plate. By fine-tuning how many organoids were seeded, how they were supported in a gel-like matrix, and which imaging dyes and readouts were used, the researchers established that just 5 to 10 organoids per well were enough for reliable drug testing. They also optimized a DNA-based fluorescence assay that reports how many cells remain alive after drug exposure, avoiding some of the pitfalls of older energy-based tests.
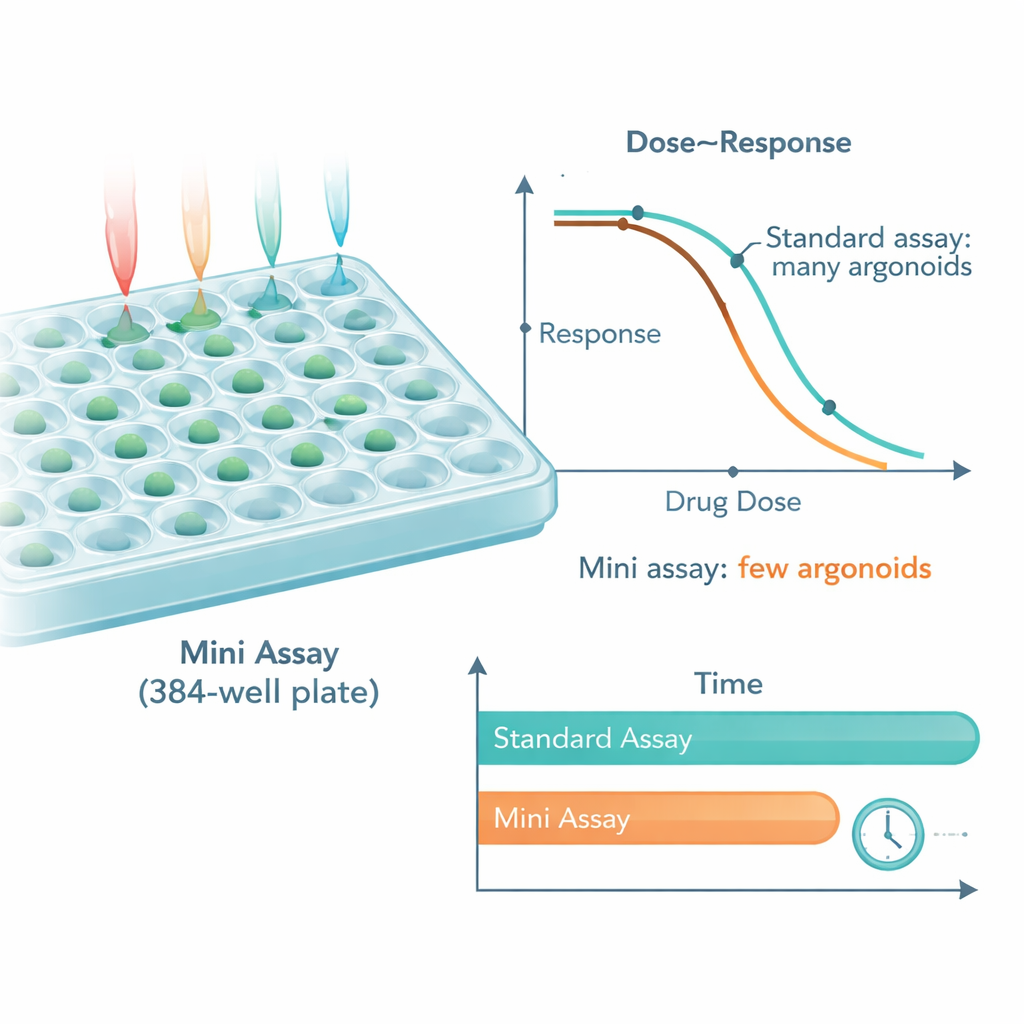
Doing More with Far Fewer Cells
After optimization, the miniaturized system used as little as 0.5% to about 5% of the material needed for conventional screens, slashing organoid requirements by 25-fold. Despite the drastic downscaling, the patterns of drug sensitivity measured with 10 organoids per well closely matched those seen with 250 organoids per well. The platform could even reveal hidden differences within a single patient’s tumor model, such as subgroups of organoids that were sensitive or resistant to a common chemotherapy drug. In an example with a targeted antibody therapy, organoids from tumors with a known resistance mutation behaved as expected—they did not respond—while organoids without that mutation were strongly inhibited, whether tested in the standard or miniaturized format.
From Lab Signals to Patient Outcomes
The researchers then asked whether this leaner assay still reflected real patient experiences. They tested organoids from people with metastatic colorectal cancer who had received standard chemotherapy drugs in the clinic. For both 5-fluorouracil and oxaliplatin, drug responses measured in miniaturized assays (with only 5–10 organoids per well and fewer drug concentrations) strongly agreed with results from larger, traditional screens. More importantly for patients, organoid sensitivity in the mini assay correlated with how long people’s cancers stayed under control and, in many cases, with overall survival. Reducing the number of organoids did not weaken these links, suggesting that the streamlined approach remains clinically informative.
What This Could Mean for Patients
By proving that accurate drug-response testing is possible with far fewer organoids and a faster workflow, this study moves organoid-based precision medicine closer to routine clinical practice. The new automated platform cuts both the number of cells required and the turnaround time by roughly half, making it more realistic to grow organoids from a biopsy, test multiple therapies, and return results in time to guide treatment decisions. While further validation across more cancer types is needed, this miniaturized organoid screening strategy offers a practical path toward choosing cancer drugs based on how an individual’s own tumor behaves in the lab, rather than relying solely on population averages.
Citation: Abouleila, Y., Smabers, L.P., Voskuilen, T. et al. Accelerating personalized medicine: miniaturized patient-derived organoid drug screening for predicting cancer treatment responses and beyond. npj Biomed. Innov. 3, 16 (2026). https://doi.org/10.1038/s44385-026-00067-9
Keywords: cancer organoids, personalized medicine, drug screening, colorectal cancer, automation in oncology