Clear Sky Science · en
Surface modified electrospun scaffold supports iPSC-derived limbal stem cell function
Giving Sight a Second Chance
When the clear window at the front of the eye—the cornea—is damaged, people can lose their vision permanently. A major culprit is the loss of special cells at the edge of the cornea, called limbal stem cells, which normally keep the surface clear and smooth. This paper describes a new, lab‑made “living bandage” that could one day carry replacement stem cells to the eye and help restore sight more safely and reliably than current methods.
Why the Eye’s Edge Matters
The cornea is made of several layers and must stay smooth and transparent to bend light properly. A small ring of stem cells sitting at the border between the clear cornea and the white of the eye constantly renews its outermost layer. Injury, infection, or autoimmune disease can destroy these limbal stem cells, causing a condition known as limbal stem cell deficiency. Without them, blood vessels grow into the normally clear cornea, the surface becomes scarred and irregular, and patients can suffer severe vision loss or blindness. Doctors can transplant healthy stem cells, but they still need a safe, strong, and transparent carrier to deliver these fragile cells to the damaged eye.
Building a Synthetic “Living Bandage”
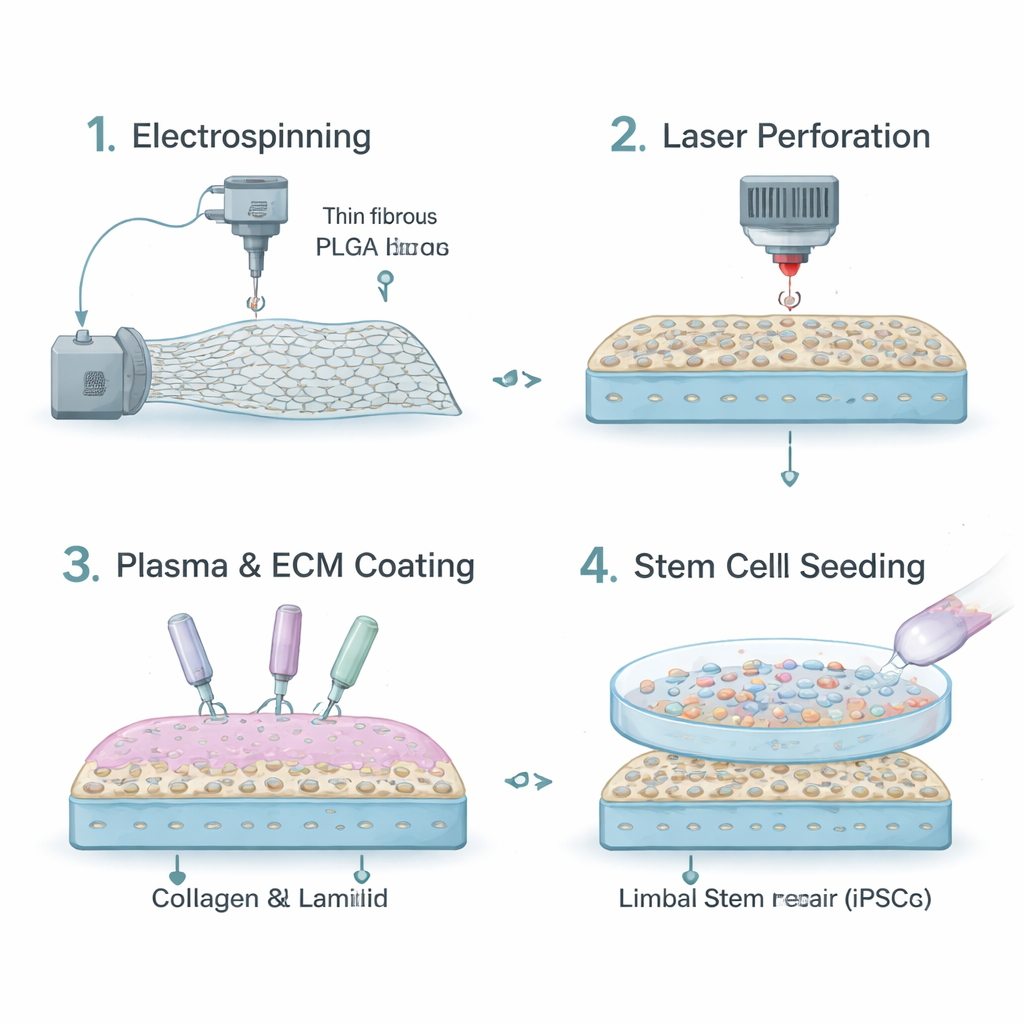
Today, many surgeons rely on natural tissues such as amniotic membrane or collagen sheets as carriers. While these materials are biocompatible, they vary from donor to donor, can be in short supply, and do not allow precise control over how long they last or how stiff they are. The researchers instead turned to a biodegradable plastic called PLGA, already used in some medical devices. Using a technique called electrospinning, they drew PLGA into a mat of tiny fibers that resembles the eye’s own supporting layer. This fibrous sheet is strong enough to handle during surgery and can be tuned to slowly dissolve over time as the eye heals.
Making the Scaffold Cell-Friendly and Clear
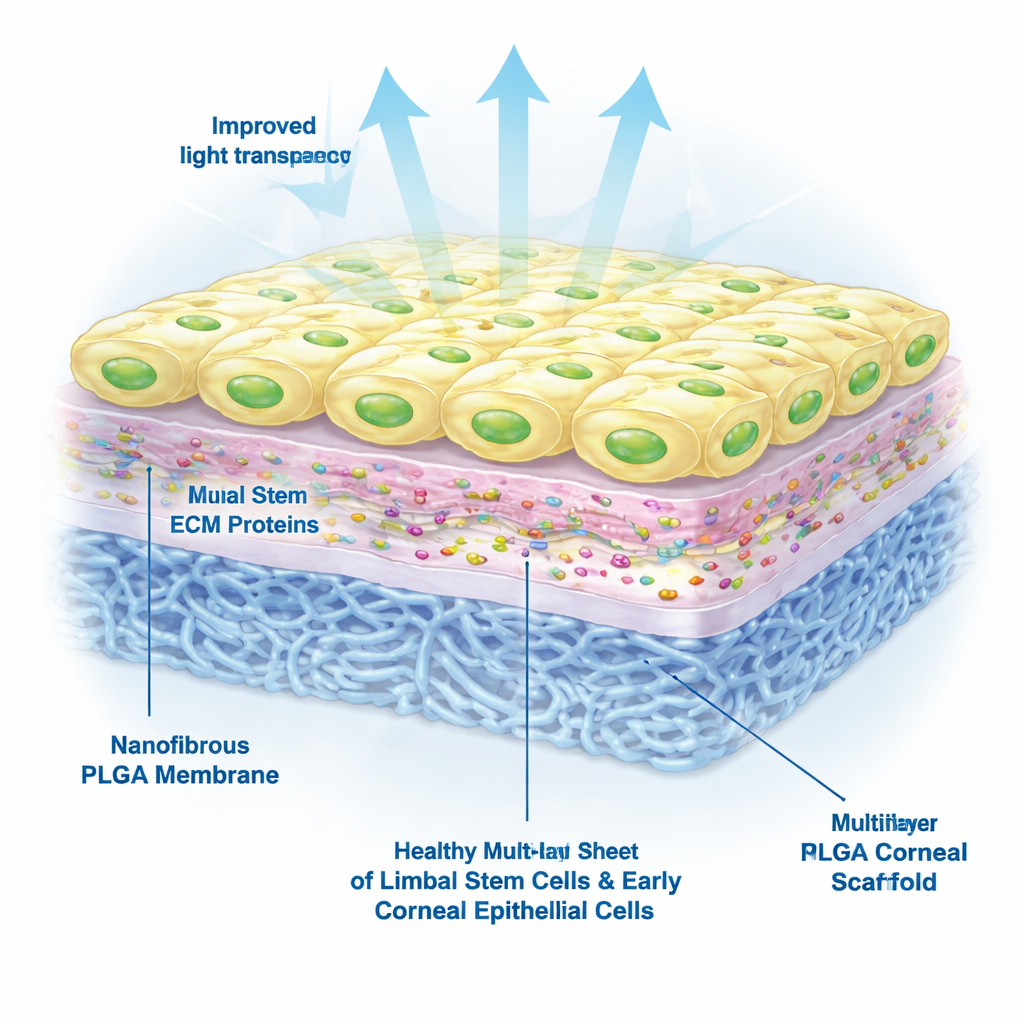
Plain PLGA, however, repels water and does not naturally attract cells. To make its surface more welcoming, the team treated the fibers with atmospheric plasma, which adds chemical groups that can grab onto proteins. They then coated the scaffold with collagen IV and laminin‑521, two key components of the natural stem cell “home” around the cornea. To improve transparency, they used a precise laser to punch microscopic holes through the membrane. These micro‑perforations increased the amount of light passing through the scaffold from about 44% to around 60%, bringing it closer to the clarity of real cornea while keeping enough fibers intact for cell attachment.
Testing Stem Cells on the New Surface
The team next tested how well limbal stem cells derived from induced pluripotent stem cells—adult cells reprogrammed back into a flexible, stem‑like state—would grow on the scaffold. They compared different surface treatments and found that laminin‑521 was essential: scaffolds that were plasma‑treated and coated with laminin‑521, with or without collagen IV, supported healthy cell attachment, survival, and growth for at least a week. In contrast, cells placed on untreated PLGA or on PLGA coated with collagen IV alone often died or detached. Under the microscope, cells on the optimized scaffold formed tightly packed, “cobblestone” sheets similar to a normal corneal surface.
Keeping Stem Cells in a Repair-Ready State
Beyond simply surviving, the transplanted cells must stay in a state where they can both renew themselves and form new corneal surface cells. The researchers used protein staining and gene tests to examine the identity of the cells on their scaffold. They found strong signals for markers associated with limbal stem cells and early corneal cells, and only weak signals for markers of fully mature cells. This pattern suggests a healthy mix of active stem cells and early offspring cells that are primed to rebuild the corneal surface, rather than a tired, worn‑out population.
What This Could Mean for Patients
Overall, the study shows that a carefully engineered, fully synthetic scaffold can mimic many features of the eye’s natural stem cell environment. By combining a tunable fibrous material, micro‑perforations for better vision, and a protein coating that specifically supports limbal stem cells, the authors created a promising carrier for future stem cell transplants. While this work was done in the lab and animal and clinical trials are still ahead, the approach could eventually offer a more consistent, scalable, and personalized way to restore a clear, healthy corneal surface and help people with otherwise untreatable vision loss see again.
Citation: Mahmood, N., Zha, D., Gullion, S. et al. Surface modified electrospun scaffold supports iPSC-derived limbal stem cell function. npj Biomed. Innov. 3, 14 (2026). https://doi.org/10.1038/s44385-026-00066-w
Keywords: corneal regeneration, limbal stem cells, electrospun scaffold, biomaterials, induced pluripotent stem cells