Clear Sky Science · en
An endosymbiosis in plant shoots based on an ankyrin repeat protein
Why tiny partners in plant shoots matter
Chemical fertilizers and pesticides have helped feed the world, but they also pollute water, burn fossil fuels, and harm pollinators and people. Farmers and scientists are searching for living alternatives: friendly microbes that live with plants and help them grow. This study reveals how one such bacterium can move inside plant shoots and roots, quietly reshaping the plant’s inner life to boost growth. By uncovering the “key” molecule that makes this partnership work, the research points toward a new generation of precise, reliable biofertilizers.
A hidden helper inside pine seedlings
Scots pine seedlings in northern forests host a microscopic ally called Methylorubrum extorquens DSM13060. Unlike most helpful microbes, which live on roots or in soil, this bacterium actually enters living plant cells in the shoots and roots and settles near the cell’s nucleus—the control center. Earlier work showed that infected seedlings grow bigger and contain more carbon, even though the bacterium does not supply extra nutrients or produce classic plant hormones. This unusual lifestyle raised a key question: how does the microbe slip inside plant cells without harming them, and how does it steer the plant toward faster growth?
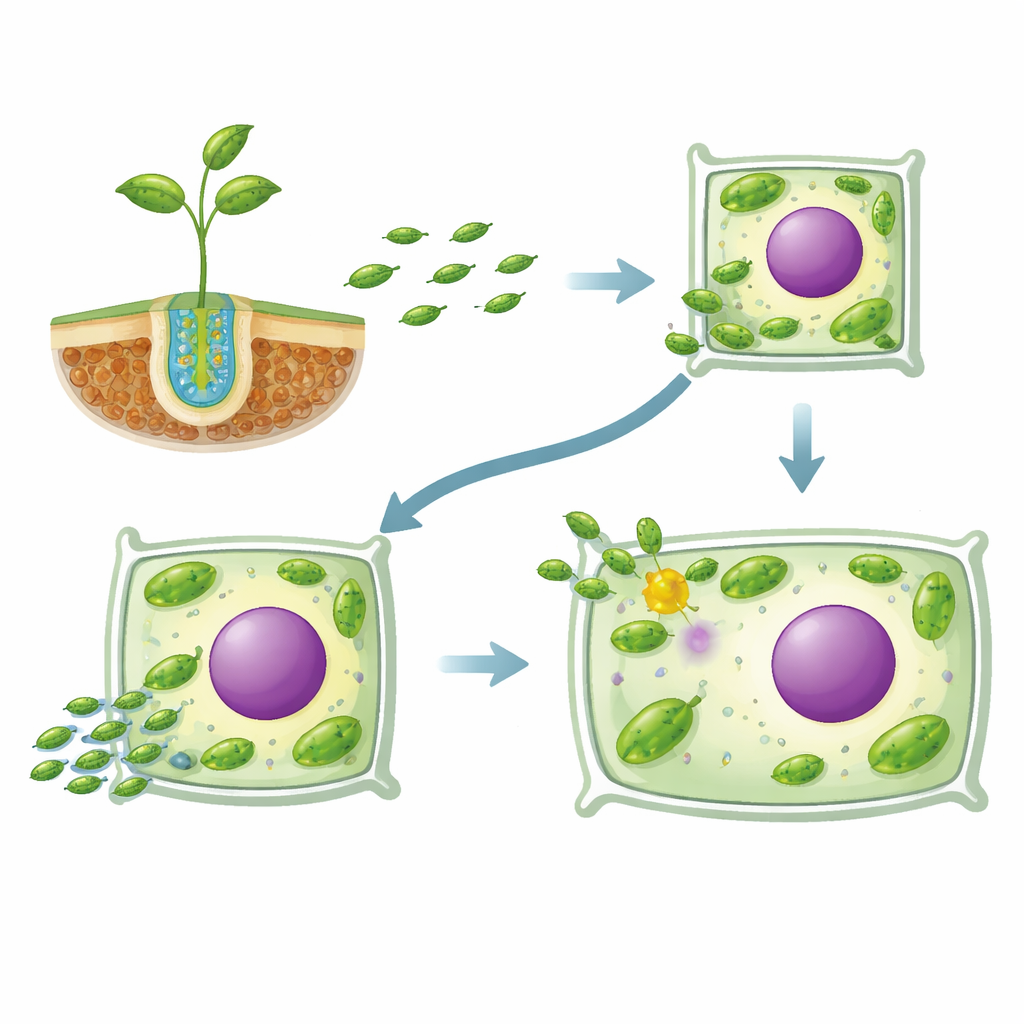
The bacterial “key” that opens plant cells
The authors focused on a single bacterial protein built from ankyrin repeats—modular shapes often used in nature for protein–protein interactions. Using a prediction tool for secreted effectors, they identified this ankyrin protein, called Ank, as likely to be injected by the bacterium into plant cells. They deleted only this one gene and compared the mutant strain, named Δank, with the normal fluorescently tagged strain during long-term colonization of pine seedlings. Under the microscope, the normal strain steadily advanced from the root surface into inner tissues, forming pockets of infection and finally gathering around plant cell nuclei in both roots and shoots. In contrast, Δank largely stalled on the root surface, rarely entered inner tissues, and was almost completely absent from shoots even after months.
When the key is missing, growth benefits vanish
The team then tested what this meant for the plant. Pine seedlings were grown with either water, the normal bacterium, or the Δank mutant, and their dry weights were measured over time. Seedlings hosting the normal strain developed heavier roots and shoots at every time point, confirming its strong growth-promoting effect. Seedlings exposed to Δank, however, grew no better than those given only water and were sometimes even smaller. This tight link between deep colonization and plant growth shows that the beneficial effect is not a side product of simple surface contact, but depends on true endosymbiosis driven by Ank.
How Ank rewires the plant from within
To see what Ank does inside the plant, the researchers used a yeast two-hybrid screen to identify plant proteins that interact with Ank. They found 46 targets, and the vast majority reside inside plant cells, especially in the nucleus and cytoplasm. Many are involved in stress and defense responses, suggesting that Ank helps tone down the plant’s immune alarms so the bacterium can enter without triggering damage. Others are tied to energy metabolism and photosynthesis, including enzymes linked to malate—a favorite carbon source for the bacterium—and components of the light-harvesting machinery. Several targets are nuclear regulators of growth and stress signaling, located right where the bacterium accumulates. Together, these interactions paint Ank as a multitool that softens defenses, adjusts energy flow, and nudges developmental programs in ways that favor both partners.
A protein key for cleaner farming
Viewed from a distance, Ank acts like a molecular key that unlocks the plant interior for a beneficial bacterial guest and turns on the guest’s positive effects: faster root and shoot growth and a calmer response to stress. Without this key, the bacterium remains stuck on the surface and loses its power to help the plant. By revealing this precise mechanism, the study moves biofertilizers from trial-and-error toward rational design. In the future, similar protein keys might allow scientists to engineer reliable, long-lasting plant–microbe partnerships that reduce our dependence on chemical inputs and support more sustainable agriculture.
Citation: Baruah, N., Koskimäki, J.J., Mohammad Parast Tabas, H. et al. An endosymbiosis in plant shoots based on an ankyrin repeat protein. npj Sci. Plants 2, 10 (2026). https://doi.org/10.1038/s44383-026-00026-8
Keywords: plant–microbe symbiosis, biofertilizer, endosymbiont bacteria, sustainable agriculture, plant stress tolerance