Clear Sky Science · en
Plastid-encoded Ycf10 maintains chloroplast proton homeostasis essential for photosynthesis in Chlamydomonas reinhardtii
Why tiny algae matter for life on Earth
Every breath of oxygen we take depends on photosynthesis, the way plants and algae turn light into chemical energy. In this study, scientists zoomed in on a single protein, called Ycf10, inside the chloroplasts of a microscopic green alga named Chlamydomonas reinhardtii. They discovered that this protein acts as a kind of pH balancer, helping chloroplasts keep just the right proton levels so that light energy can be used safely and efficiently. Understanding this hidden regulator may one day help us design crops and algae that grow better under strong light or fluctuating climates.
Keeping the cell’s solar panels in balance
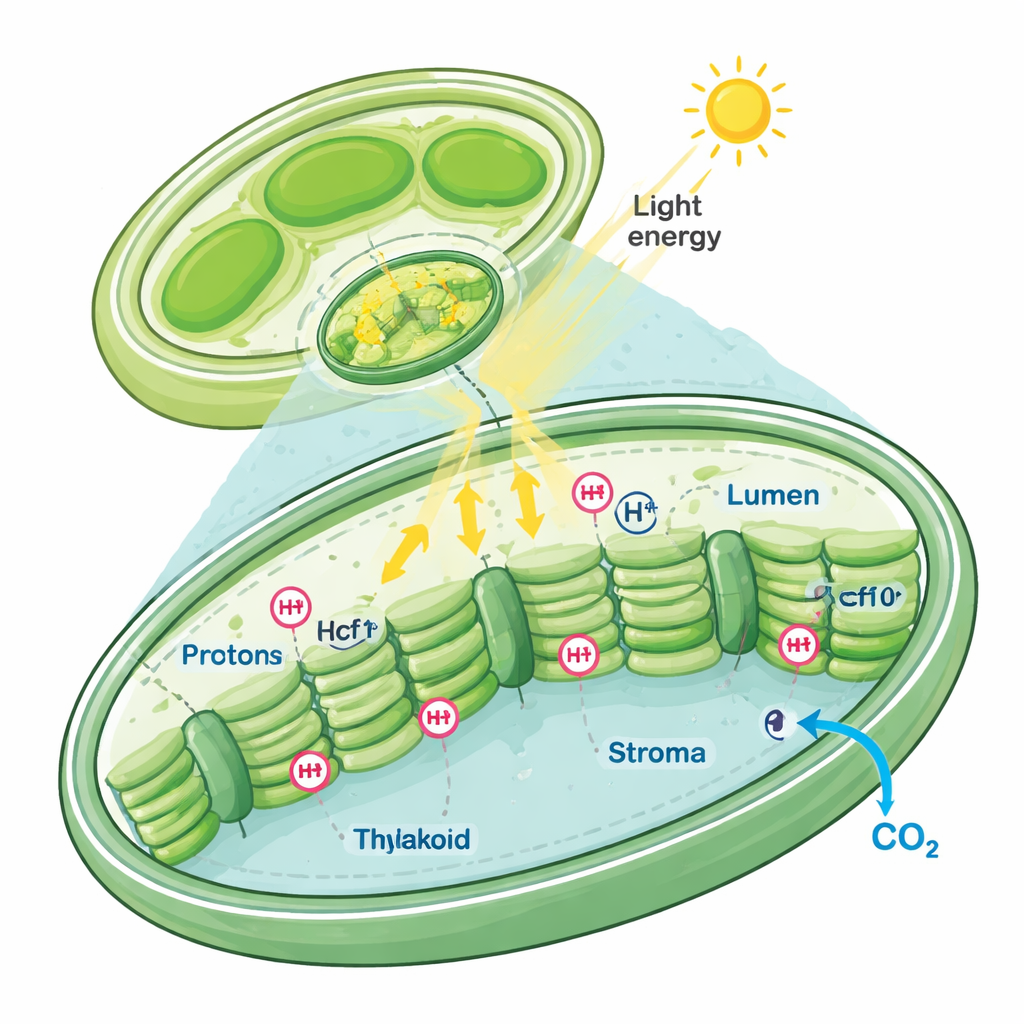
Chloroplasts are the “solar panels” of plant and algal cells, and their internal chemistry must be finely tuned. When light is absorbed, it drives electrons along a chain of protein complexes and pumps protons to create a gradient, which in turn powers the production of the energy-rich molecule ATP and supports carbon dioxide (CO2) fixation. If this balance tips too far under intense light, harmful reactive oxygen species (ROS) form and damage the chloroplast. To prevent this, cells use a safety valve called non-photochemical quenching (NPQ), which harmlessly releases excess light energy as heat. The authors suspected that Ycf10, a little-studied, chloroplast-encoded membrane protein, might help control proton levels and, through that, influence both light protection and CO2 use.
Disabling Ycf10 exposes a hidden weak point
To probe Ycf10’s role, the team engineered mutant Chlamydomonas strains in which the ycf10 gene was disrupted but neighboring photosynthesis genes remained largely unaffected. They confirmed that Ycf10 is a membrane protein embedded in the chloroplast envelope and that its amount drops under high light in normal cells. On rich medium, the mutants grew almost as well as the wild type, but they contained less chlorophyll and their growth faltered when forced to rely purely on photosynthesis. Careful measurements of chlorophyll fluorescence and gas exchange showed that their ability to transport electrons, evolve oxygen, and consume oxygen in respiration all declined, especially after several hours of strong light. NPQ, the light safety valve, was also much weaker in the mutants, leaving them more vulnerable to light stress.
Proton balance and carbon uptake fall out of tune
The researchers then asked directly whether the internal proton balance was disturbed. Using a sensitive optical signal that reports on the proton motive force, they found that under normal light the overall “battery” was similar in mutants and wild type, but the split between electrical potential and pH difference was changed. After high light treatment, the total proton motive force and particularly the pH difference across the thylakoid membrane dropped sharply in the mutants, indicating poor lumen acidification. Dyes that glow in acidic environments revealed extra acidic spots in the cytoplasm of mutant cells after strong light, hinting that protons were in the wrong place. Non-invasive microelectrodes showed that, unlike wild-type cells, mutants tended to take up protons from the medium under high light. When grown at different external pH values, the mutants struggled most in acidic conditions, and their growth improved as the medium became more alkaline, consistent with a defect in proton homeostasis.
From disturbed pH to poor CO2 use and self-digestion
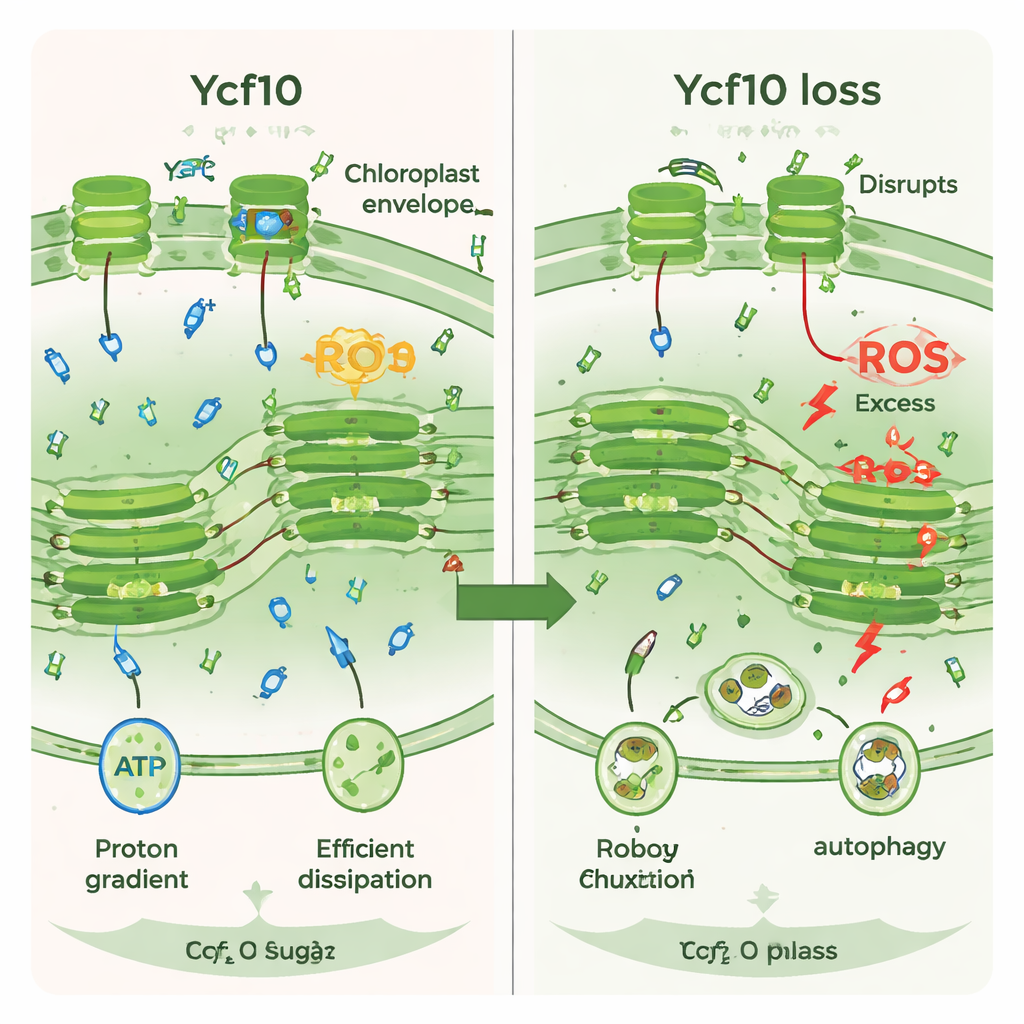
Because CO2 and bicarbonate interconvert in a proton-dependent way, the team next examined how Ycf10 loss affected inorganic carbon use. Under acidic conditions, mutant cells showed a lower affinity for inorganic carbon during photosynthesis than wild type, though this difference largely disappeared at neutral or alkaline pH. Genes that belong to the carbon concentrating mechanism—a system that helps raise CO2 levels around the CO2-fixing enzyme—were switched on more strongly in mutants, suggesting the cells were trying to compensate. Direct measurements confirmed that CO2 fixation capacity dropped in the mutants after high light exposure. At the same time, ROS levels rose and markers of autophagy, the cell’s self-cleaning and recycling pathway, increased, and fluorescent staining revealed more autophagosomes. Altogether, the cells appeared to slide into photooxidative damage and begin dismantling their own chloroplasts.
A small protein with a big protective job
In plain terms, this work shows that Ycf10 helps chloroplasts keep their “acid–base” balance just right during photosynthesis. When Ycf10 is impaired, protons accumulate where they should not, the light-driven proton gradient weakens, the light safety valve fails to open fully, and CO2 is not used efficiently. Under strong light, this runaway situation leads to excess reactive molecules and triggers a cellular cleanup response that can degrade chloroplasts. By uncovering Ycf10’s role as a central coordinator linking proton balance, light protection, and carbon capture, the study highlights a subtle control point that could be targeted to make plants and algae more resilient and productive in a changing environment.
Citation: Lv, K., Pan, J., Yang, H. et al. Plastid-encoded Ycf10 maintains chloroplast proton homeostasis essential for photosynthesis in Chlamydomonas reinhardtii. npj Sci. Plants 2, 7 (2026). https://doi.org/10.1038/s44383-026-00025-9
Keywords: chloroplast proton homeostasis, photosynthesis, Chlamydomonas reinhardtii, carbon concentrating mechanism, non-photochemical quenching