Clear Sky Science · en
A foundation model for breast and lung cancer screening using non-contrast computed tomography
Why One Scan for Two Cancers Matters
Cancer screening usually works like a series of separate checkups: one test for the lungs, another for the breasts, each adding time, cost and extra radiation. This study explores a different idea—whether a single, widely used chest scan could quietly check for both lung and breast cancer at once, with help from artificial intelligence (AI). If successful, such an approach could turn routine scans into a double-duty safety net, especially in busy hospitals and regions with limited resources.
A New Kind of Digital Cancer Scout
The researchers developed an AI system called OMAFound that learns to "read" three-dimensional chest CT scans. Unlike traditional tools trained narrowly on one organ, this system first studies more than 200,000 unlabeled scans from over 58,000 people, simply learning what healthy and diseased chests look like in general. This broad pretraining lets the AI pick up subtle patterns across the whole chest. The team then adds smaller, labeled datasets indicating whether each patient eventually proved to have breast cancer, lung cancer or neither, teaching the model to turn its general image understanding into concrete cancer predictions.
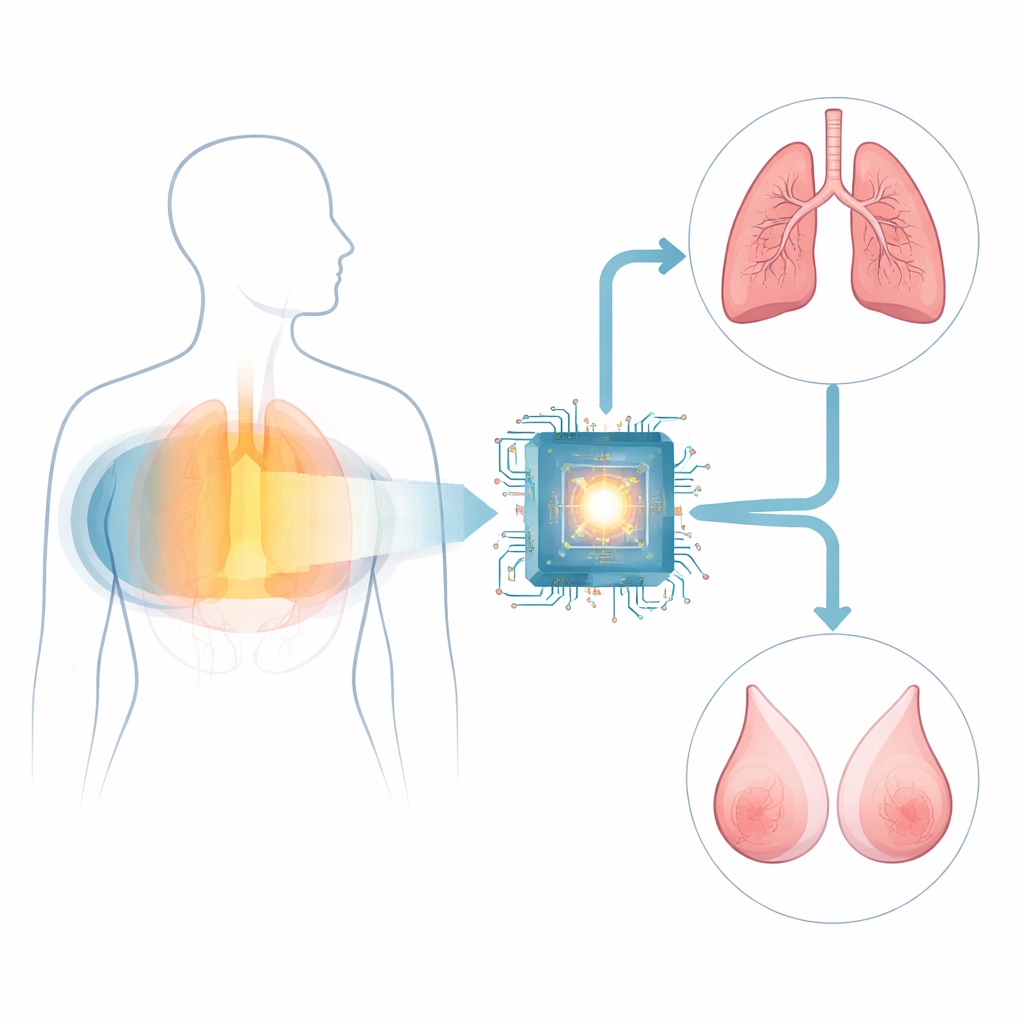
Teaching One System to Watch Two Organs
On top of the shared image-reading core, the team built two focused branches: one tuned to signs of breast cancer and one tuned to signs of lung cancer. They trained and tested these branches on CT scans from more than 150,000 patients across several Chinese hospitals and international datasets. For breast cancer, they also built a separate AI system that reads mammograms, the current standard screening test, so they could fairly compare CT-based AI with established practice in the same women. In side-by-side tests using women who had both kinds of images, the mammogram AI was slightly more accurate overall, but the CT-based breast branch was more sensitive—catching more cancers—while mammography was better at avoiding false alarms.
From Organ Findings to Whole-Patient Answers
Looking at each organ in isolation can mislead: if both branches say "possible cancer," the combination might unrealistically suggest two separate tumors in the same person. To avoid this, the researchers created a third AI module that blends information from both breast and lung views of the same CT scan and decides whether the patient overall is likely to have cancer. This patient-level view follows clinical reality, where people far more often have a single cancer, if any. Among women in the test sets, this combined strategy offered the best balance—high sensitivity to existing cancers while keeping false positives in check—outperforming simple mathematical ways of merging the organ-level outputs.
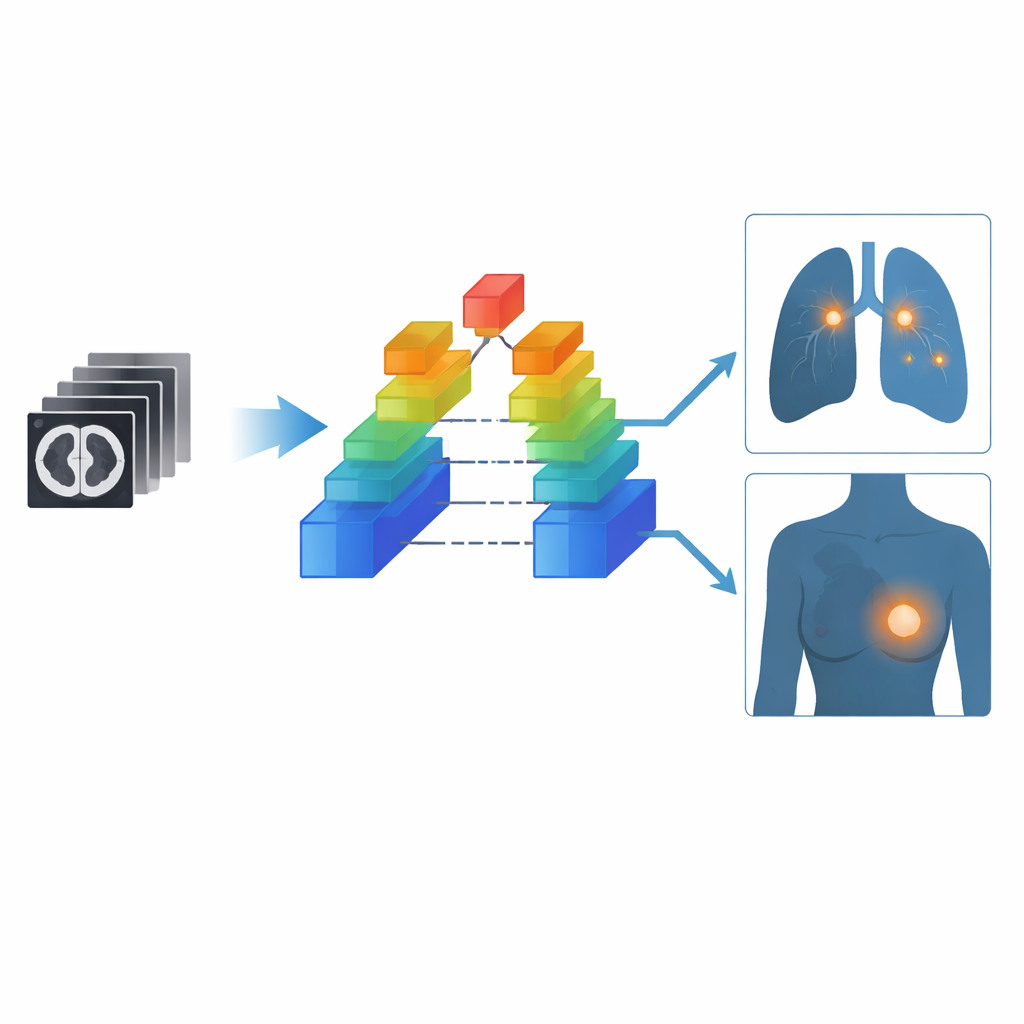
Testing the System in Real-World Screening
To see how OMAFound works outside the lab, the team ran a prospective study in four medical centers, following more than 21,000 people coming for low-dose chest CT screening. In men, where only lung cancer is relevant, the system correctly balanced cancer detection and false alarms about 86% of the time. In women, it reached balanced accuracies of about 82% for breast cancer, 88% for lung cancer and 83% when deciding whether a woman had any cancer at all. The researchers also asked seven generalist radiologists to read a challenging sample of scans, first on their own and then with OMAFound’s risk scores and heatmaps highlighting areas of concern. With AI assistance, the radiologists’ ability to catch cancers rose sharply—especially for breast tumors—while their tendency to overcall non-cancer cases did not increase.
What This Could Mean for Everyday Care
Overall, the study suggests that a single low-dose chest CT, already used widely for lung checks, could also offer an additional layer of breast cancer screening when paired with a strong AI system. OMAFound does not replace mammography or expert judgment, but it could flag high-risk people earlier and help overworked clinicians focus their attention where it is most needed. By turning one common scan into a multi-cancer lookout, the approach points toward screening programs that are more efficient, more accessible and potentially more lifesaving, without adding extra tests, cost or radiation exposure.
Citation: Liang, Z., Niu, Q., Wang, J. et al. A foundation model for breast and lung cancer screening using non-contrast computed tomography. Nat. Health 1, 403–415 (2026). https://doi.org/10.1038/s44360-026-00055-8
Keywords: multi-cancer screening, low-dose CT, breast cancer, lung cancer, medical AI