Clear Sky Science · en
Optimising the induction of inflammation within preterm infant-derived intestinal epithelial organoids
Why this matters for fragile newborns
Babies born very early face a dangerous combination: an immature gut and a rapidly changing community of microbes. Together these can spark runaway inflammation that damages the intestine and leads to life‑threatening conditions such as necrotising enterocolitis. Because we cannot experiment directly on preterm babies, scientists need realistic lab models of their guts to test what drives inflammation and how to stop it. This study refines such a model using tiny lab‑grown gut tissues, offering a clearer way to probe which microbial signals tip the balance from normal defence into harmful inflammation.
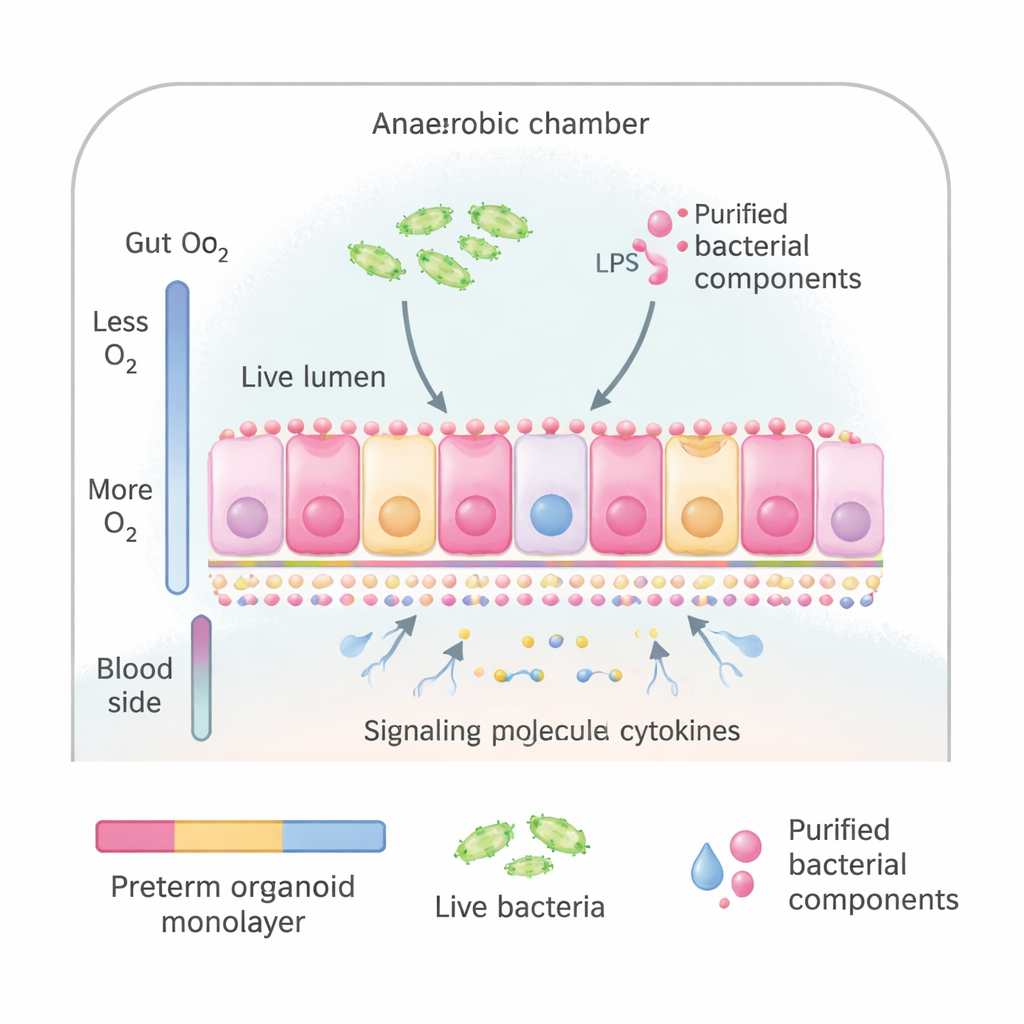
Growing a preterm gut in a dish
The researchers used intestinal organoids—miniature versions of gut lining grown from stem cells taken from preterm infants during surgery. These organoids can be flattened into a thin sheet of cells that looks and behaves much like the inner surface of the small intestine, including the correct top (facing the gut contents) and bottom (facing the bloodstream). The team placed these sheets into a special culture system that mimics the natural oxygen gradient of the gut: low oxygen on the side facing microbes and higher oxygen on the blood side. This allowed them to expose the “gut” surface either to whole bacteria taken from preterm babies or to purified bacterial components that are known to alert the immune system.
Testing what best triggers an inflammatory alarm
The scientists compared several ways of provoking inflammation: a mix of live bacteria commonly found in sick preterm infants; the same bacteria that had been killed by heat; and two purified bacterial signals, lipopolysaccharide (LPS) from bacterial cell walls and flagellin from bacterial tails. They measured release of IL‑8, a signalling protein that attracts immune cells and serves as a general marker of inflammation, and used large‑scale protein analysis to see how the gut cells’ internal machinery changed. Surprisingly, live or dead bacteria did not strongly increase IL‑8 in this system, even after 24 hours. In contrast, flagellin and especially the combination of flagellin and LPS triggered a clear rise in IL‑8 within just three hours, showing that these purified signals could reliably switch on an inflammatory state.
Finding the most realistic and efficient setup
To better match real gut biology, the team next paid attention to where each signal naturally acts. LPS is usually sensed on the side of the gut lining that faces the intestinal contents, whereas flagellin is mainly detected on the tissue side. Using much lower, more physiologically realistic doses than earlier experiments, they applied LPS to the top (lumen‑facing) side and flagellin to the bottom (blood‑facing) side of the organoid sheet. This simple change produced a strong and broad response within three hours: multiple inflammatory messengers—including IL‑8, TNF and several chemokines—were secreted from both sides of the tissue. At the same time, hundreds of cellular proteins changed in abundance, indicating large‑scale activation of defence pathways.
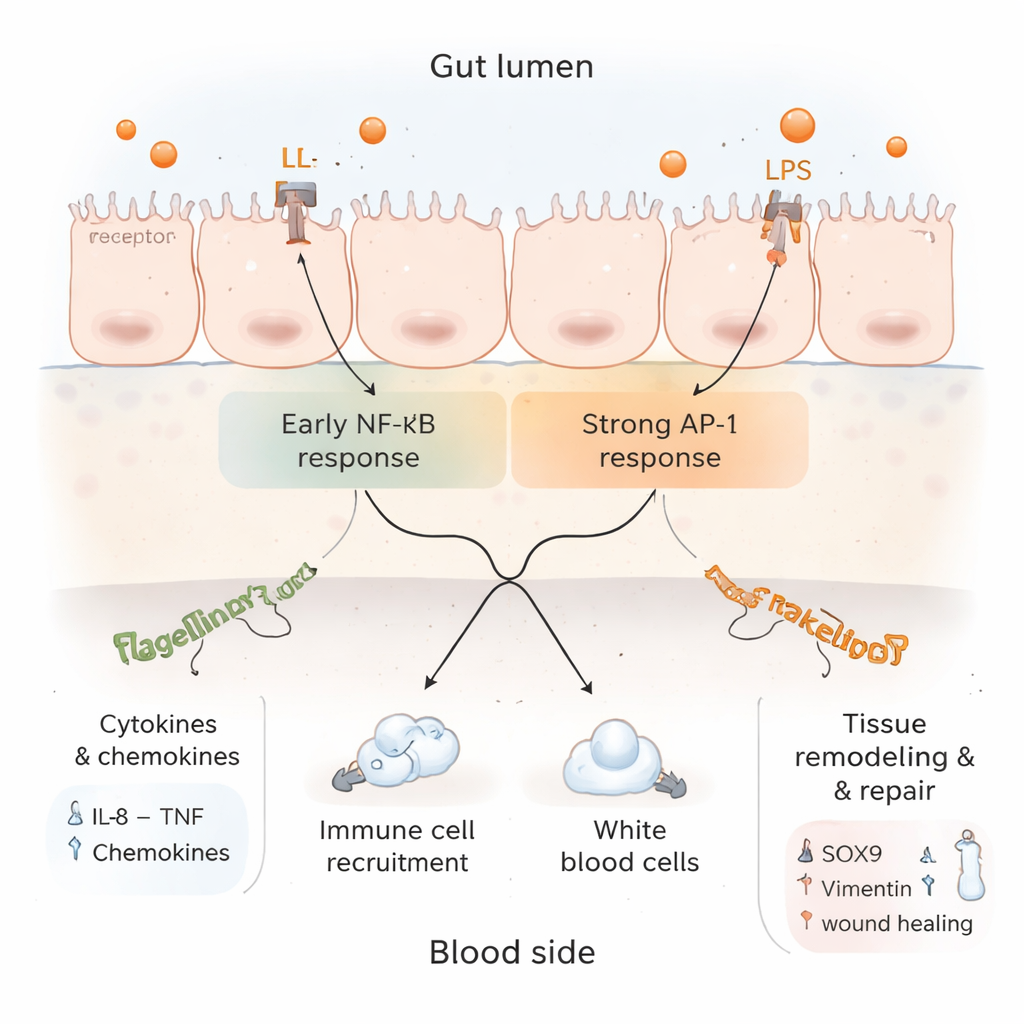
Inflammation, brakes, and repair in the same model
Digging deeper into the protein data, the researchers found a complex picture. On one hand, markers of active inflammation and immune cell recruitment were boosted, and signalling routes linked to TNF and IL‑17—both central to gut immune defence—were switched on. On the other hand, several components of the classic NF‑κB pathway, which drives many inflammatory genes, were dialled down, while an alternative route involving AP‑1 remained active. This suggests that after an initial alarm, the tissue begins to apply brakes to prevent uncontrolled damage. At the same time, proteins associated with tissue regeneration, structural reshaping, and controlled cell death were increased, hinting that the gut lining was not only inflamed but also attempting to remodel and heal itself.
What this means for future treatments
By systematically comparing different microbial stimuli, doses, positions, and exposure times, the authors conclude that a three‑hour exposure to low‑dose LPS on the gut side together with flagellin on the blood side is the most robust and reproducible way to induce inflammation in preterm‑derived intestinal organoids. This refined model captures not just the flare of inflammation, but also the built‑in checks, tolerance mechanisms, and repair responses of the fragile preterm gut. It provides a practical blueprint for other laboratories to study how specific microbes, drugs, or nutritional factors might worsen or calm gut inflammation in very premature infants—an essential step toward safer, targeted strategies to prevent devastating intestinal diseases in this vulnerable group.
Citation: Chapman, J.A., Frey, A.M., Dueñas, M.E. et al. Optimising the induction of inflammation within preterm infant-derived intestinal epithelial organoids. npj Gut Liver 3, 5 (2026). https://doi.org/10.1038/s44355-026-00054-2
Keywords: preterm infant gut, intestinal organoids, gut inflammation, microbiome, necrotising enterocolitis