Clear Sky Science · en
Hydrogen-based ore-to-part manufacturing of near-net-shape stainless steel
Turning Rocks into Ready-to-Use Steel Parts
Most everyday objects—from cars and bridges to kitchen appliances—depend on steel, but making it the traditional way releases vast amounts of carbon dioxide. This study explores a cleaner route: starting from mineral powders that resemble crushed rock and using hydrogen gas to transform them directly into finished stainless-steel parts, potentially cutting both emissions and manufacturing steps.
A New Shortcut in the Steelmaking Journey
Conventional steel production is a long chain: ores are mined, purified at high temperatures using coal or coke, melted, cast, rolled, forged, and then machined into shape. Every stage consumes energy and usually burns fossil fuels. The authors propose collapsing much of this chain into a single integrated route they call “ore-to-part.” Instead of first making pure metal and then shaping it, they mix oxide powders that represent real ore ingredients for stainless steel—iron, chromium, nickel, and molybdenum oxides—plus the natural “dirt” (silicates and alumina) that accompanies them. These powders are shaped close to the final geometry and then exposed to hot hydrogen, which strips away oxygen and turns the mixture into solid metal.
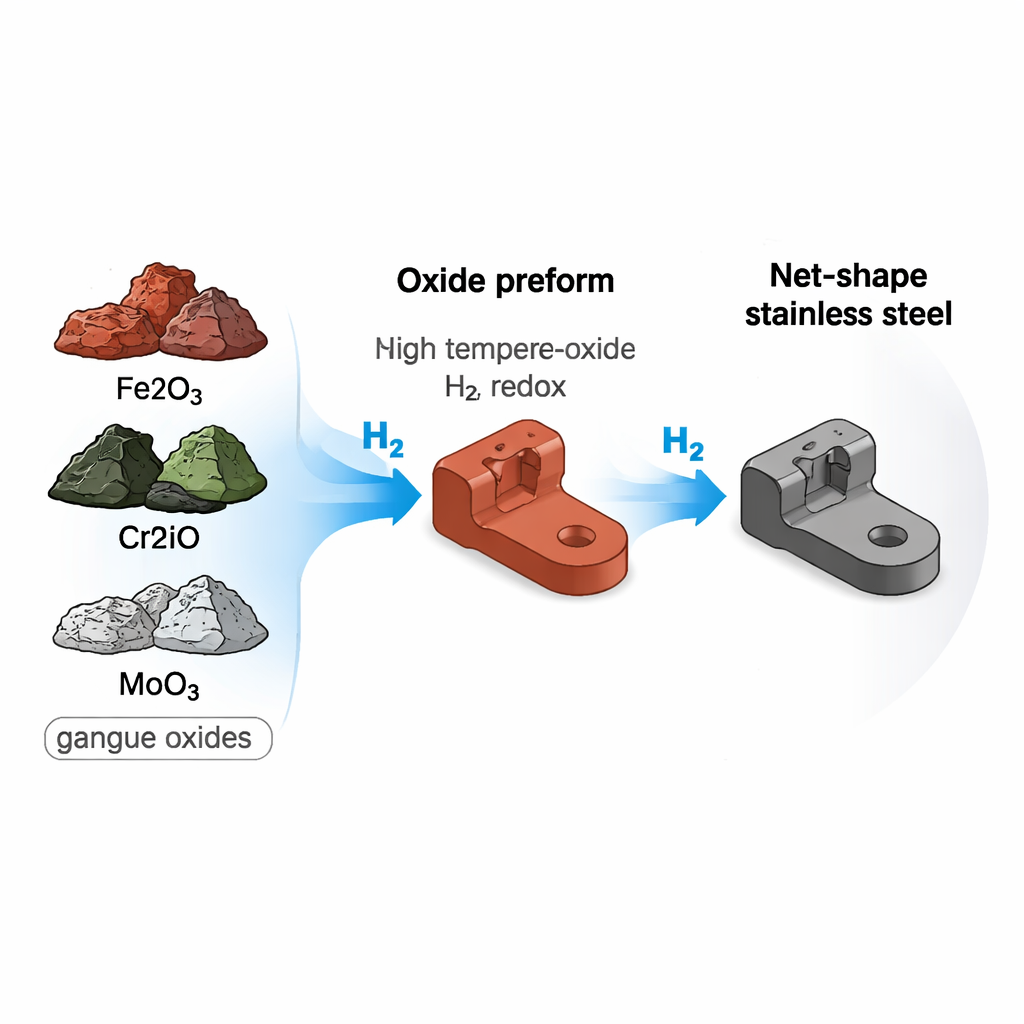
Hydrogen as a Cleaner Chemical Sculptor
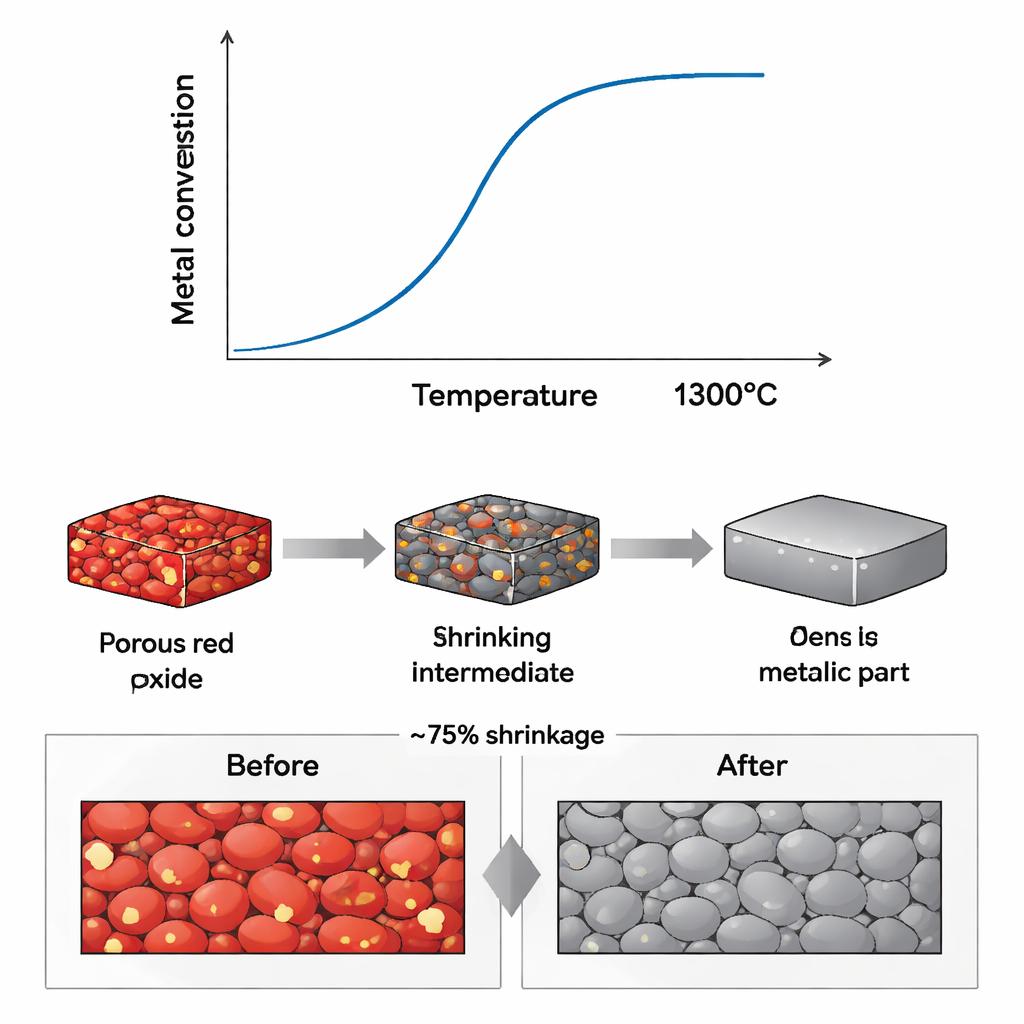
In the furnace, hydrogen acts like a chemical sculptor: it pulls oxygen out of the oxide mixture, forming water vapor instead of carbon dioxide. Careful measurements of weight loss as the sample is heated reveal how this transformation unfolds. By about 700 degrees Celsius, most of the oxygen has already been removed; by 1300 degrees, all four metals are fully “liberated” from their oxides and blended into a single stainless-steel phase. X-ray measurements confirm that the original mix of oxide crystals gives way to a uniform metallic structure, where iron, chromium, nickel, and molybdenum atoms share a common crystal arrangement typical of austenitic stainless steels used in cookware and chemical equipment.
From Chunky Preform to Precise Metal Part
To test whether this chemistry can be used for real components, the team cast the oxide mixture into the shape of a suspension bearing, a mechanical part with practical engineering features. After hydrogen treatment, the part shrank by about three-quarters in volume but did so evenly in all directions, preserving the original design lines. This means engineers can compensate for the shrinkage simply by scaling up the starting mold. Microscopy images show a dense, crack-free metal with a fine, even distribution of the main alloy elements. Tiny pockets of the original “gangue” oxides—based on silicon and aluminum—remain, mainly along pores and interfaces, where they could weaken the metal. This suggests that while full purification may not be essential, controlling or reducing these impurities will be important for demanding applications.
Unraveling the Hidden Order of Chemical Changes
Behind the scenes, the oxide mixture does not reduce all at once; there is a specific sequence. Thermodynamic calculations reveal that nickel oxide and molybdenum oxide convert to metal at relatively low temperatures, followed by progressive reduction of iron and finally chromium oxide, which is traditionally hard to reduce with hydrogen alone. The study shows that intermediate compounds formed between iron and chromium oxides help pull chromium across the finish line, especially once some metallic iron is already present. In effect, the mix of oxides cooperates to lower the barriers for the most stubborn components, allowing the whole blend to become stainless steel under conditions milder than expected from standard textbook diagrams.
What This Means for Greener Metalmaking
For non-specialists, the take-home message is that it may be possible to turn ore-like powders straight into near-final stainless-steel parts using hydrogen, skipping several energy-intensive steps and greatly reducing emissions. The process still faces challenges—such as managing shrinkage and leftover mineral impurities—but the proof-of-concept shows that “rocks to ready parts” is no longer just an idea. If developed further, this approach could contribute to low-carbon steel production, shorten supply chains, and ultimately make the metal world we rely on more sustainable.
Citation: Yang, M., Kannan, R., Keshavarz, M.K. et al. Hydrogen-based ore-to-part manufacturing of near-net-shape stainless steel. npj Adv. Manuf. 3, 9 (2026). https://doi.org/10.1038/s44334-026-00069-w
Keywords: hydrogen steelmaking, stainless steel, additive manufacturing, ore reduction, low-carbon metallurgy