Clear Sky Science · en
Carbon nanotube microelectrode arrays enable scalable and accessible electrophysiological recordings of cerebral organoids
Listening In on Miniature Human Brains
Scientists are increasingly growing tiny, simplified versions of the human brain in the lab, called cerebral organoids. These living “mini-brains” could transform how we study brain development, neurological diseases, and new medicines. But to truly understand what these organoids are doing, researchers need to eavesdrop on their electrical activity—the language of the brain. This paper introduces a new, affordable platform that makes it much easier to record these signals from many organoids at once, bringing large-scale brain-on-a-chip experiments closer to everyday lab reality.
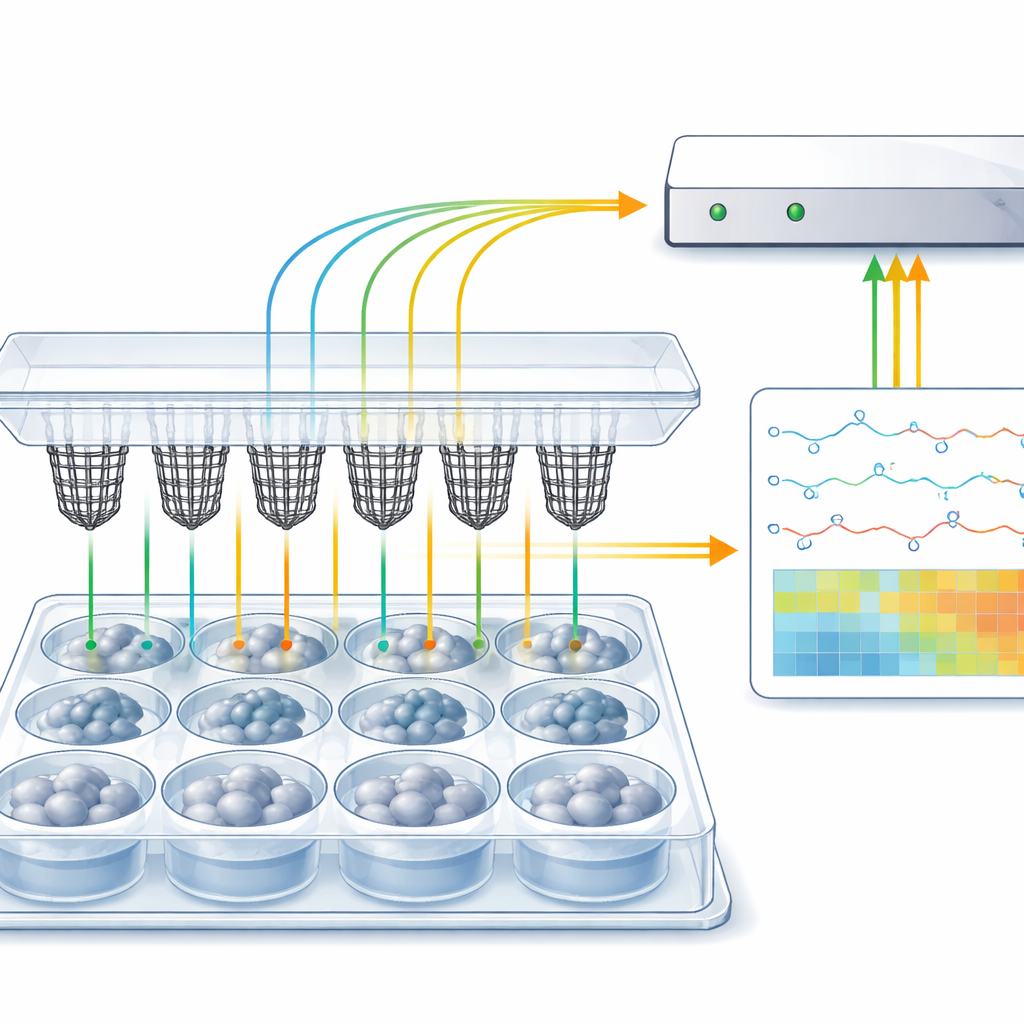
Why Measuring Mini-Brain Activity Is Hard
Cerebral organoids mimic key features of the human brain, including complex cell types and spontaneous electrical firing. However, every organoid is somewhat different, and this natural variability means scientists must study large numbers to draw reliable conclusions. Existing tools for measuring electrical activity, like microelectrode arrays and fine-tipped probes, are usually built for flat cell cultures, are expensive, and often require specialised cleanroom manufacturing and custom culture setups. Three-dimensional electrode grids exist, but they are difficult to make, low-throughput, and rarely fit smoothly into the standard plastic plates most biology labs already use.
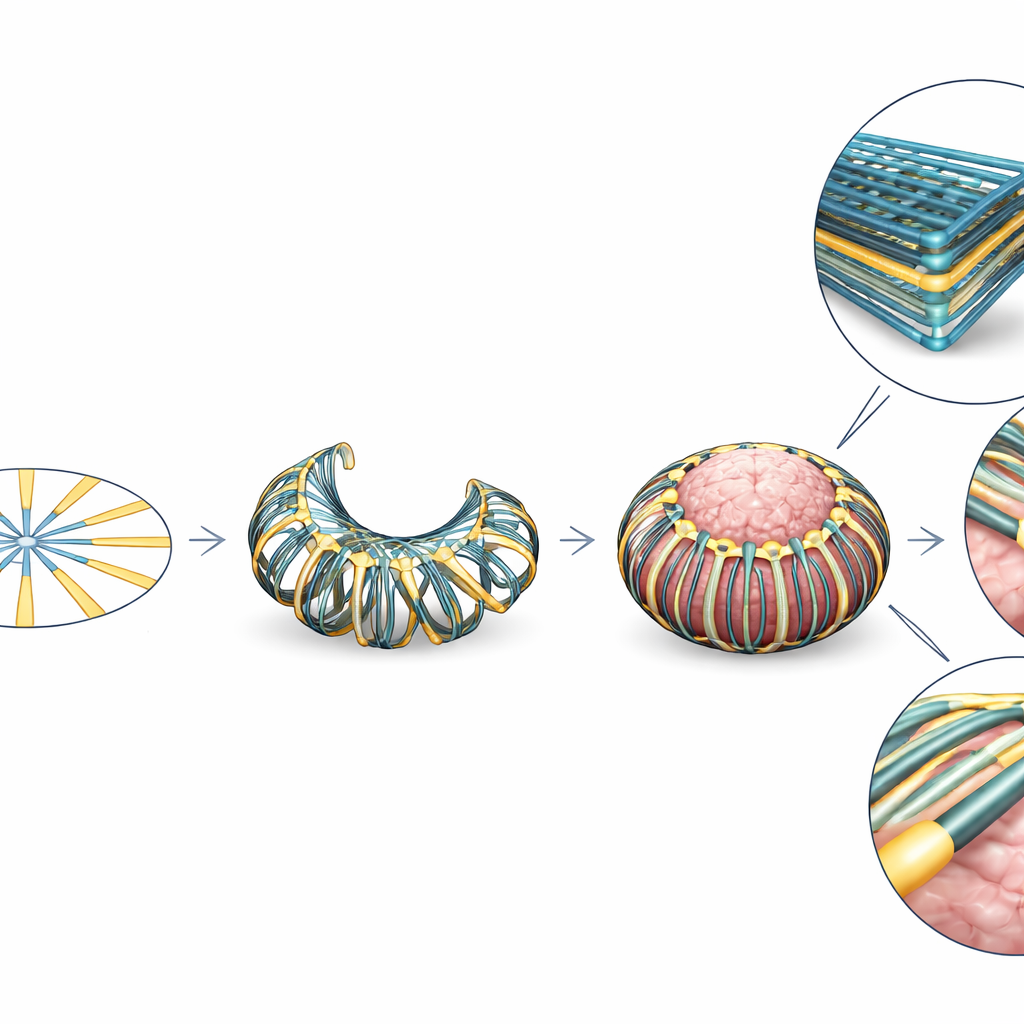
A Basket That Gently Hugs the Brain
The authors present a new device called CAMEO (Conformal Array for Monitoring Electrophysiology of Organoids). Each CAMEO starts life as a flat, cartwheel-shaped pattern of twelve thin electrode “spokes.” During assembly, this cartwheel is transformed into a basket-like structure that hangs from the lid of a standard six-well culture plate. When an organoid is pipetted into a well, the flexible spokes bend inward and gently wrap around its surface, forming a net that conforms to the organoid without trapping or damaging it. The basket shape positions electrodes all around the organoid in three dimensions, while the lid connects to off-the-shelf recording systems through a thin printed circuit board. Multiple CAMEOs on one lid allow parallel recordings from many organoids.
New Materials for Cheaper, Tougher Sensors
Instead of using costly metals like gold or platinum, CAMEO electrodes are made from films of single-walled carbon nanotubes embedded in a soft polymer. The team developed a bulk process in which pristine nanotubes are dissolved in a strong acid and then self-assemble into free-standing, centimetre-scale films at the liquid surface. Because this approach avoids harsh vibration and surfactants that usually damage nanotubes, the resulting sheets retain high electrical conductivity, flexibility, and strength at a fraction of the cost of gold. Laser cutting and simple lamination steps on tattoo paper replace traditional microfabrication, enabling dozens of devices to be produced in parallel without a cleanroom. Tests show that these nanotube electrodes maintain stable resistance under repeated bending and display lower electrical impedance and better charge transfer than gold, properties that improve signal quality for tiny neural spikes.
Proving the Platform Works
The researchers first validated that CAMEO devices can detect very small, well-defined test signals passed through salty solution, preserving the waveform shape despite the low voltages involved. They then moved to real organoids. Human cortical organoids were grown using established recipes and later transferred into a brain-supportive medium known to enhance neuronal firing. After an hour resting inside the CAMEO baskets, the organoids produced clear electrical spikes of 10–100 microvolts—typical for neuronal activity—while control wells without organoids remained quiet. Adding glutamate, a major excitatory brain chemical, or high potassium levels caused spikes to increase, confirming that the recorded signals behaved like genuine neuronal responses.
Spotting Disease Signatures at Scale
To showcase the power of high-throughput recording, the team studied organoids derived from people with Angelman Syndrome, a rare neurodevelopmental disorder caused by loss of a gene called UBE3A in neurons. They recorded signals from 34 organoids—both neurotypical and Angelman types—grown in parallel. The Angelman organoids showed significantly smaller spike amplitudes than controls, echoing earlier findings from single-cell experiments but now observed in intact three-dimensional tissue. Overall, the platform captured activity from 74 organoids across different experiments, representing the largest electrophysiology dataset for cerebral organoids reported so far and highlighting its ability to handle biologically diverse samples.
What This Means for Future Brain Research
This study demonstrates that low-cost, flexible carbon nanotube baskets integrated into ordinary culture plates can reliably record electrical activity from many miniature human brains at once. By combining affordability, robustness, and compatibility with everyday lab workflows, CAMEO lowers a major barrier to large-scale functional studies of brain organoids. In practical terms, it opens the door to more systematic investigations of brain development, drug responses, and genetic disorders, where statistically meaningful sample sizes are essential. As the platform is refined for long-term recordings and paired with advanced data analysis, it could become a standard tool for mapping how complex neural networks emerge and falter in health and disease.
Citation: Mishra, N., Kaveti, R., Liu, P. et al. Carbon nanotube microelectrode arrays enable scalable and accessible electrophysiological recordings of cerebral organoids. npj Biosensing 3, 20 (2026). https://doi.org/10.1038/s44328-026-00088-9
Keywords: brain organoids, electrophysiology, microelectrode arrays, carbon nanotubes, Angelman syndrome