Clear Sky Science · en
Microfluidic nanomagnetically isolated neuron- and astrocyte-derived extracellular vesicles to differentiate Lewy body and Alzheimer’s disease
Why this matters for families and doctors
Dementia is an umbrella term that covers several brain diseases with similar symptoms but very different underlying causes. Two of the most common, Alzheimer’s disease and Lewy body disease, can look nearly the same in the clinic, yet respond differently to drugs and carry different risks. Today, the only way to know with certainty which disease a person had is to examine the brain after death. This study describes a new kind of blood test that reads tiny messages released from brain cells, with the goal of telling these two dementias apart while people are still alive.
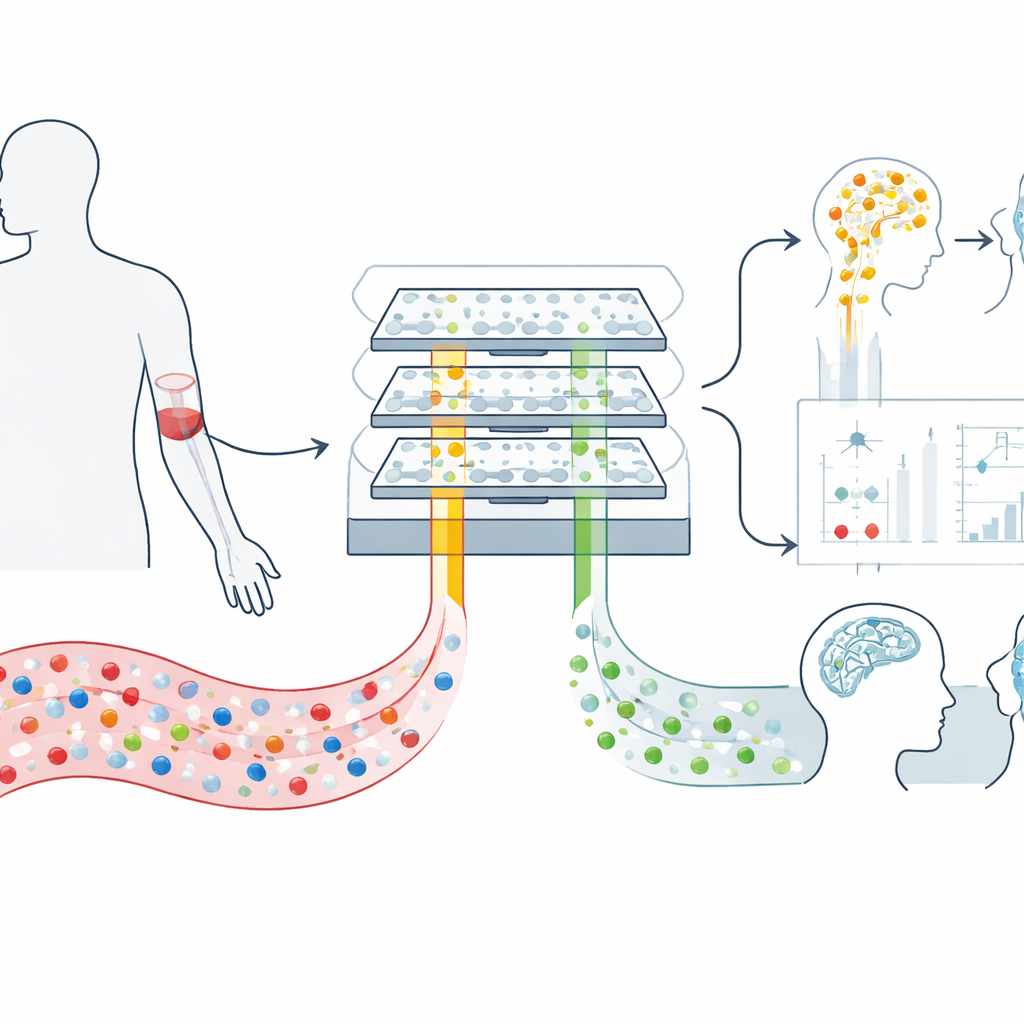
Tiny parcels traveling from brain to blood
Our brains constantly shed microscopic bubbles called extracellular vesicles into the bloodstream. These vesicles are like padded envelopes sent out by nerve cells and their support cells, carrying bits of genetic material and proteins that reflect what is happening inside the brain. Because they can cross the blood–brain barrier and survive in circulation, they offer a rare, minimally invasive window into brain biology. The challenge is that a milliliter of blood contains hundreds of billions of vesicles from the entire body, so brain-derived ones are vastly outnumbered and hard to pull out with standard lab methods.
A magnetic chip that sorts brain signals
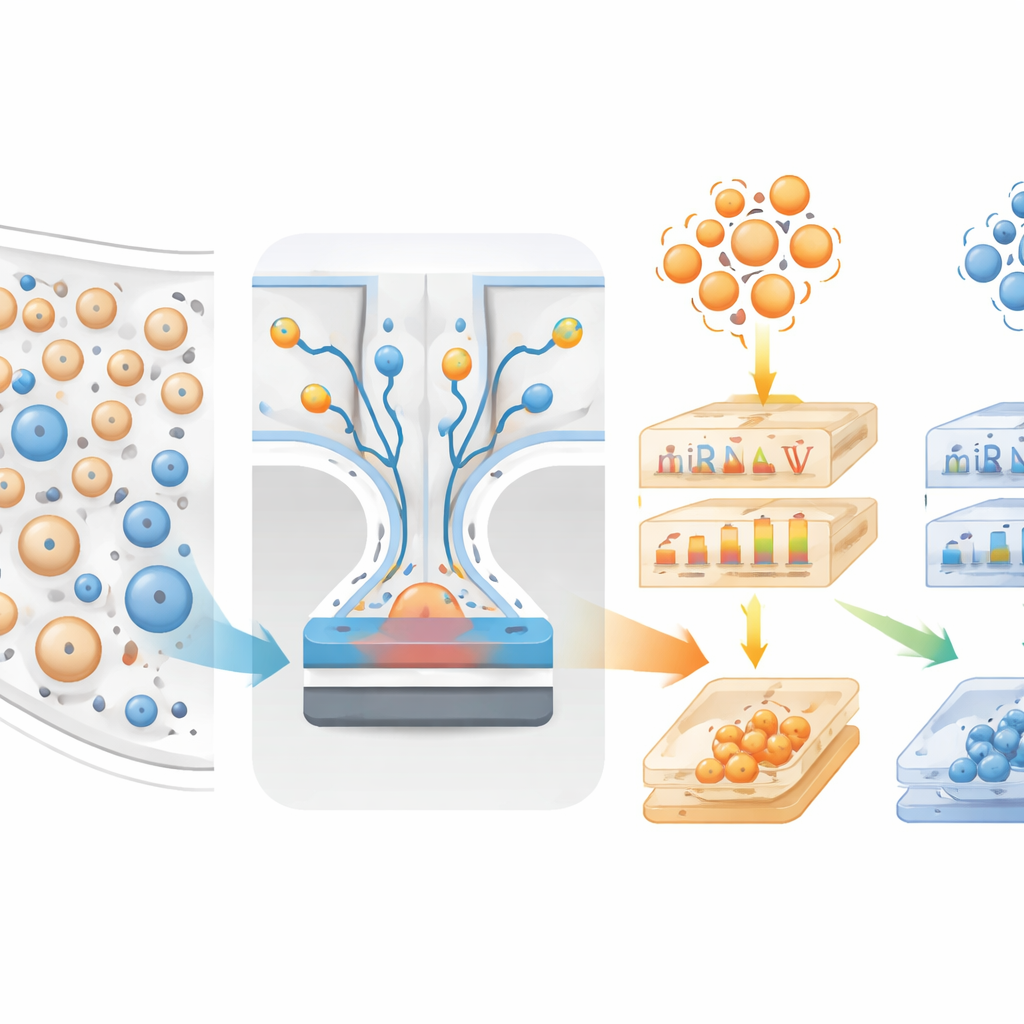
To tackle this, the researchers built a microfluidic device called mTENPO that combines a credit-card–sized chip with a strong magnet. Before a blood sample enters the chip, vesicles are tagged with magnetic nanoparticles using antibodies that latch onto surface proteins typically found on nerve-cell vesicles (GluR2) or astrocyte vesicles (GLAST). As the tagged mixture flows through millions of tiny pores in the chip, the magnetic field pulls the strongly tagged brain vesicles toward the pore walls, holding them back while the rest wash through. In this way, the team can independently enrich two vesicle populations—those mainly from neurons and those mainly from astrocytes—directly from a small plasma sample.
Reading the molecular fingerprints
From 137 volunteers whose brain diagnoses were confirmed after death, the team isolated these two vesicle types and also measured classic Alzheimer’s blood proteins such as different forms of beta-amyloid and tau. They then sequenced short regulatory RNAs (miRNAs) inside the vesicles and asked which ones differed between people with pure Lewy body disease, pure Alzheimer’s, mixed pathologies, or no dementia. Multiple miRNAs carried by neuron-derived and astrocyte-derived vesicles, along with several tau-related proteins in plasma, showed distinct patterns between Lewy body and Alzheimer’s groups. When the researchers mapped the genes targeted by these miRNAs, they found strong links to pathways involved in cell survival, inflammation, and protein handling—processes already implicated in neurodegeneration.
Building a powerful combined blood test
Because no single marker cleanly separated the diseases, the team used machine-learning tools to assemble a compact panel of the most informative signals drawn from all three compartments: neuron vesicles, astrocyte vesicles, and blood proteins. The resulting 15-feature panel included 14 specific vesicle miRNAs plus two phosphorylated tau proteins. Tested with repeated cross-validation to reduce overfitting, this multi-marker panel distinguished Lewy body disease from Alzheimer’s with about 95% accuracy and an area under the curve of 0.96—substantially better than any individual measure alone. People whose brains showed mixtures of pathologies tended to have intermediate marker levels, suggesting that such panels may one day help recognize mixed disease instead of forcing an either–or label.
From proof of concept to future clinic use
The study is an early but important demonstration that brain-cell vesicles in blood can carry enough detailed information to separate major dementia types. The work still needs to be confirmed in larger, more diverse groups and in patients followed over time, and the authors emphasize current limits in pinpointing the exact cell of origin for every vesicle. Nonetheless, the combination of a scalable magnetic chip and a carefully chosen multimarker panel offers a roadmap for future blood tests that could guide treatment choices, improve clinical trials, and give families much clearer answers about the specific disease behind a loved one’s dementia.
Citation: Yang, S.J., Lin, A.A., Shen, H. et al. Microfluidic nanomagnetically isolated neuron- and astrocyte-derived extracellular vesicles to differentiate Lewy body and Alzheimer’s disease. npj Biosensing 3, 19 (2026). https://doi.org/10.1038/s44328-026-00086-x
Keywords: extracellular vesicles, Lewy body disease, Alzheimer’s disease, blood-based biomarkers, microfluidic diagnostics