Clear Sky Science · en
Fast and sensitive multiplexed diagnostic system enabled by real-time solid-phase PCR assay
Why faster virus tests matter
When someone shows up with fever and a cough, doctors often need to know quickly which virus is to blame—flu, COVID-19, or something else entirely. Today’s lab tests can be very accurate, but they are often slow, expensive, and designed to look for only a few germs at a time. This paper describes a new kind of “lab on a chip” that can spot several different respiratory viruses at once, within about 20 minutes, using a compact and relatively simple device.
A tiny lab on a plastic chip
The researchers built a microfluidic chip—about the size of a postage stamp—that guides drops of liquid through narrow channels. Into this chip, a health worker could load a patient sample, such as material from a nasal swab. The chip then handles three major steps that are usually done with separate instruments: cleaning up the genetic material from the sample, copying it millions of times using a method called PCR, and reading out which viruses are present. Because everything happens automatically inside the same device, the number of manual steps, the risk of contamination, and the need for skilled staff are all greatly reduced.

Turning a PCR test into a pixel map
A key innovation lies in how the device reads the genetic signals. Instead of assigning a different color dye to each virus—which quickly becomes impractical when testing many targets—the team attaches short DNA “catchers” for many viruses onto a flat surface inside the chip, arranged as a tiny grid. All of the viruses share the same fluorescent label, but each catcher occupies its own physical spot. During the test, copied genetic fragments from the sample bind to their matching spots like puzzle pieces finding their correct place. A simple camera looks down at the surface and measures how bright each spot becomes over time, turning the result into a map of glowing dots that reveals which viruses are present and how much of each there is.
Two rooms, one fast temperature cycle
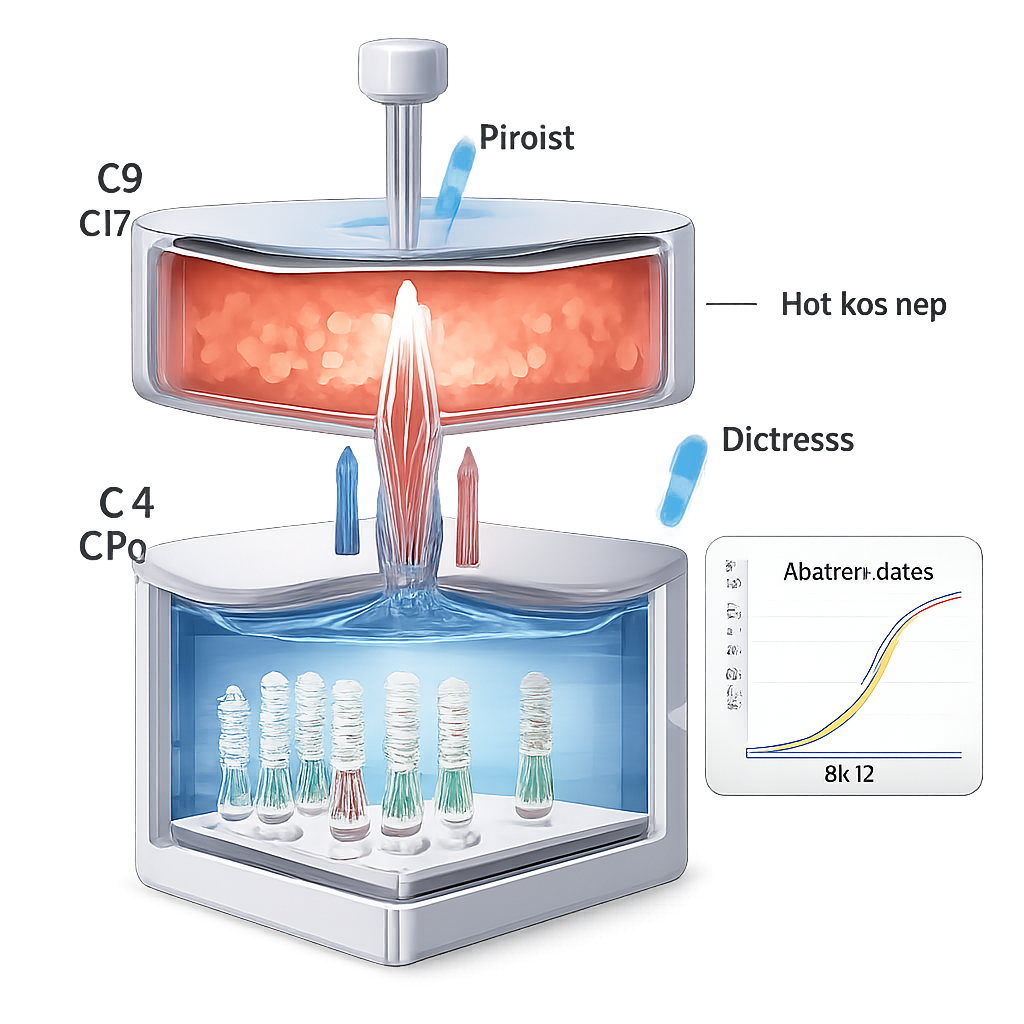
Conventional PCR machines heat and cool a bulk metal block over and over, which takes time and requires bulky heaters, fans, and careful temperature control. In this new system, the authors sidestep that problem by splitting the process between two permanent temperature “rooms” on the chip. One chamber is kept hot to separate DNA strands, while the other is kept warm to let new strands form and stick to the catcher spots. A flexible roof over the hot chamber is pressed and released by a mechanical pin, shuttling the reaction liquid back and forth through a narrow channel. Because only the liquid moves and the solid parts stay at constant temperature, each cycle is completed in seconds, and the overall test finishes much faster than in standard machines.
Cleaning up the signal for clear answers
Another challenge with many fluorescent tests is background glow from unused dye molecules floating in the liquid, which can swamp the signal from the surface-bound spots. The authors solve this by timing when they read the fluorescence: after each heating and cooling cycle, the liquid containing the free dye is pulled away into the hot chamber, temporarily leaving the catcher surface almost dry. At that moment, the camera records the brightness of each spot with minimal interference. Over 40 cycles, the signals from virus-specific spots rise in a way that can be quantified, while control spots remain stable. In trials using synthetic RNA for five different respiratory viruses—including SARS-CoV-2, influenza A and B, rhinovirus, and parainfluenza—the chip could reliably detect as few as 10 copies of viral genetic material per reaction.
Toward rapid, multi-virus checks at the point of care
To a non-specialist, the main message is that the authors have combined several clever engineering ideas—a tiny valve, a two-room temperature design, and a surface covered with virus-specific catchers—into a single compact device that can quickly and sensitively test for multiple respiratory viruses at once. Because it uses only one fluorescent color and avoids complex optics and large heating blocks, the system could be cheaper and easier to shrink into portable instruments. While the present work used prepared viral samples rather than real patient swabs, it points toward future point-of-care tools that could deliver broad, accurate diagnoses in minutes, helping clinicians choose the right treatment and respond faster during outbreaks.
Citation: Seder, I., Téllez, R.C., Zhang, J. et al. Fast and sensitive multiplexed diagnostic system enabled by real-time solid-phase PCR assay. npj Biosensing 3, 17 (2026). https://doi.org/10.1038/s44328-026-00082-1
Keywords: multiplex PCR, microfluidic diagnostics, solid-phase PCR, respiratory viruses, point-of-care testing