Clear Sky Science · en
Point-of-care multiplexed detection of cytokines using a HexaPie electrode array for asthma endotyping
Why testing asthma with spit could change home health
Asthma affects hundreds of millions of people, yet doctors still struggle to match each patient with the right treatment. This study introduces a small electronic sensor, called HexaPie, that reads immune signals from a simple saliva sample instead of blood. By measuring several inflammation-related molecules at once, it could one day let people monitor their asthma—and other immune problems—quickly and painlessly at home or in a clinic. 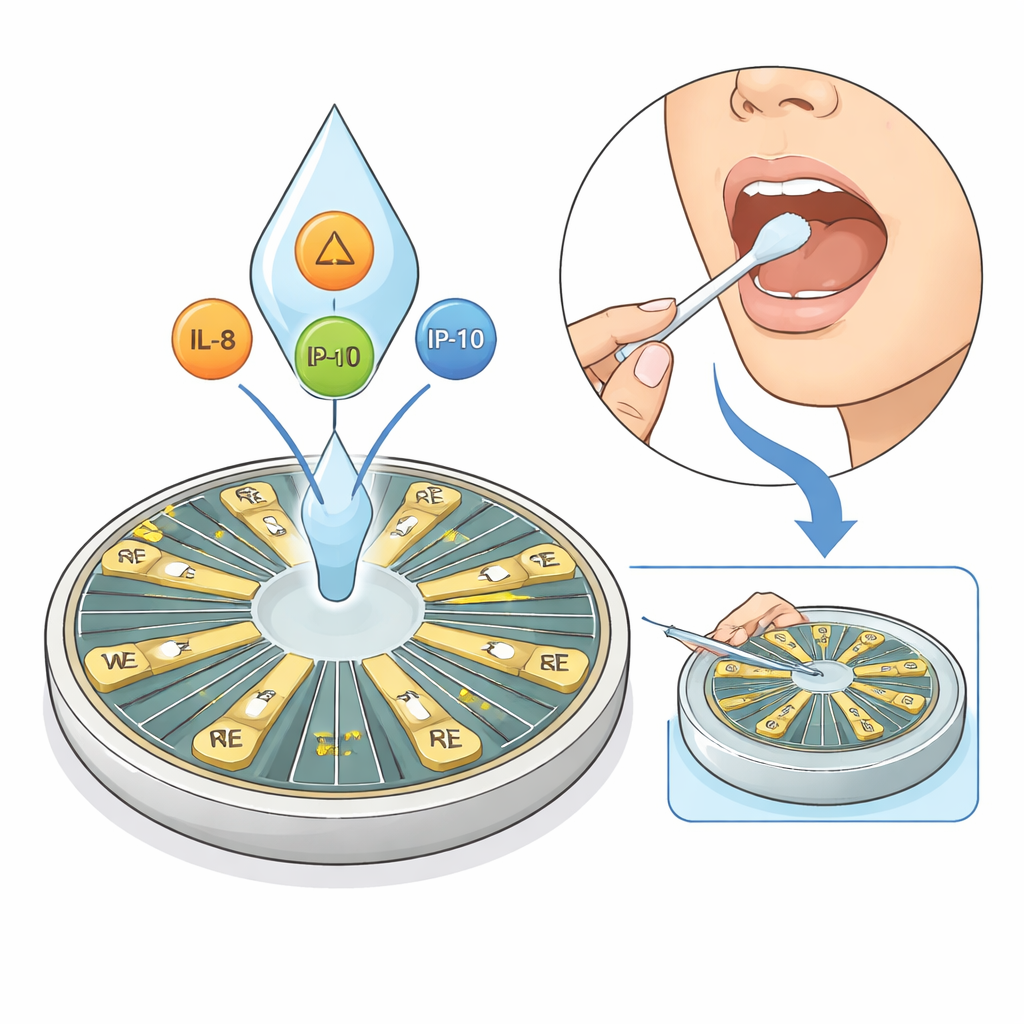
A new way to listen to the immune system
Asthma is not a single disease but a collection of different “types” driven by distinct immune pathways. Traditional tests often focus on one marker at a time and usually require blood draws and centralized laboratory machines, which are slow and inconvenient for frequent checks. The HexaPie system aims to solve this by spotting three key immune messengers in saliva—IL-8, IL-10, and IP-10—that together sketch a broader picture of airway inflammation, including difficult-to-treat, non‑eosinophilic forms of asthma. Because saliva collection is noninvasive and simple, it is well suited to repeated monitoring outside of large hospitals.
A tiny gold array that senses without extra chemicals
HexaPie is built on a custom printed circuit board that hosts six pairs of tiny gold electrodes arranged like slices of a pie. Each working electrode is coated with a different antibody that specifically latches onto one of the target cytokines, while the paired reference electrode stays bare. Instead of using added dyes or redox chemicals, the device relies on a label‑free electrical method: when cytokines in saliva bind to their antibodies, they subtly rearrange charged particles and water at the metal surface. This changes the electrical double layer—a thin region right at the interface—and the resulting shift in electrical impedance can be read out directly, allowing simultaneous measurement of all three cytokines from a single, small droplet.
Putting the design through virtual and laboratory tests
Before building full devices, the team used computer simulations to check that the HexaPie geometry would distribute electric fields and currents in a controlled way between each working and reference electrode. The models showed that the applied voltage stays well confined near the sensing regions and that current flows mainly across the intended gaps—conditions needed for stable, repeatable readings. In the lab, the researchers then verified the chemistry on gold electrodes by adding each layer—linker, antibody, and antigen—while tracking standard electrochemical signals. As more biological material covered the surface, electron flow slowed and impedance increased in predictable steps, confirming that the antibody layers formed correctly and could capture their targets. 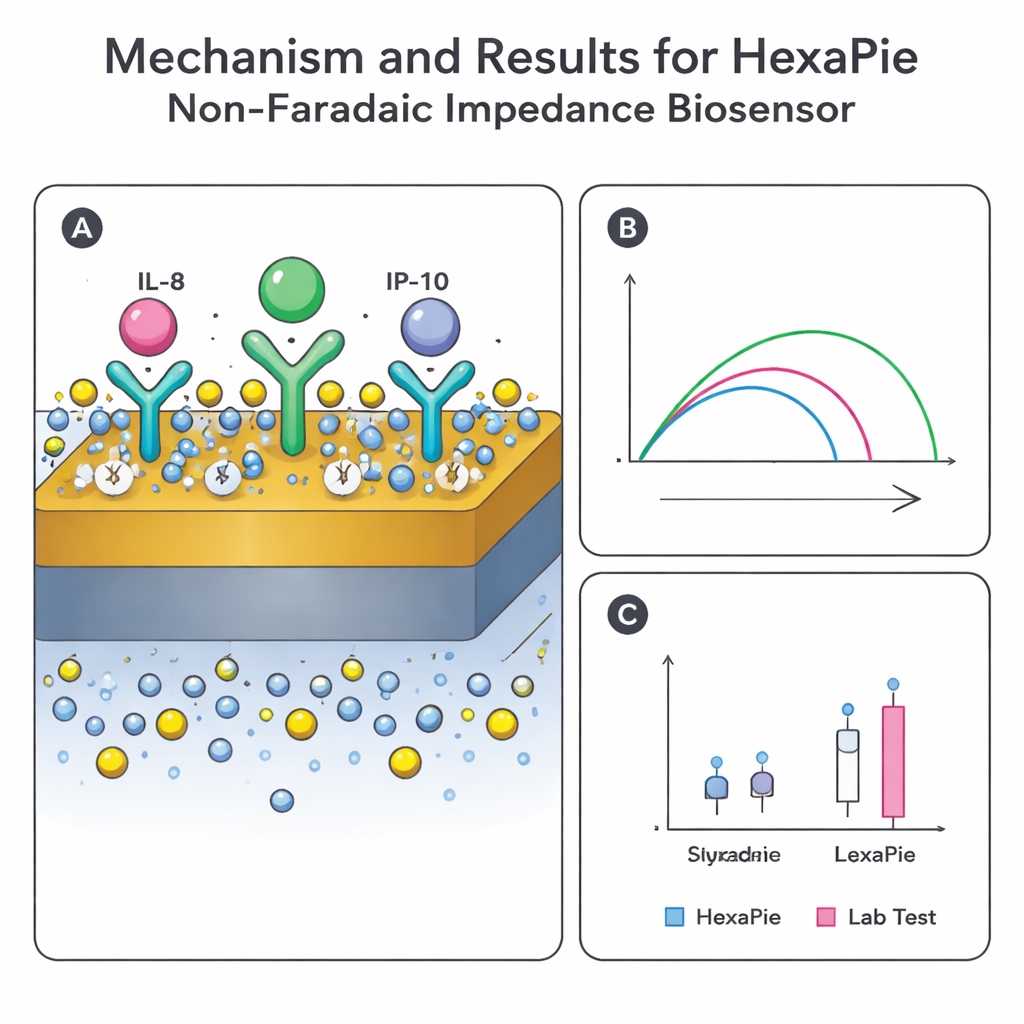
How well the sensor measures real saliva samples
To see whether HexaPie could give trustworthy numbers in practice, the authors spiked known amounts of the three cytokines into pooled human saliva and measured the resulting impedance changes. The signals shifted in an orderly, concentration‑dependent way, and by fitting the data the team built calibration curves that convert electrical readings into cytokine levels. The device reached low detection limits—down to trillionths of a gram per milliliter for some markers—within the ranges reported for people with inflamed airways. Repeated measurements on the same chip and across different chips varied by less than 15 percent, meeting accepted clinical standards. When the researchers added common saliva components such as albumin, uric acid, and vitamin C at high levels, the sensor’s responses hardly budged, indicating that everyday interferents are unlikely to confuse the readings.
Stacking up against hospital‑grade tests
The ultimate test was comparing HexaPie with established laboratory platforms. Saliva from people with asthma (during flare‑ups and in stable phases) and from healthy smokers and non‑smokers was analyzed in parallel using HexaPie, an automated immunoassay system (ELLA), and a bead‑based multiplex test (Luminex). Although this small study did not find clear group‑to‑group differences in cytokine levels—likely due to timing and sample size—the new sensor’s values for IL‑8 and IP‑10 closely matched those from both reference methods, with only tiny average offsets. IL‑10, which is naturally scarce and tricky to measure, showed more disagreement among platforms, underscoring a broader challenge in reliably detecting such low‑abundance molecules in saliva.
What this means for everyday care
For non‑specialists, the takeaway is that HexaPie brings laboratory‑style immune measurements to a compact, reagent‑free device that works directly on saliva. It reliably tracks clinically meaningful rises in three asthma‑related cytokines without added labels or complex sample handling, and its readings agree well with gold‑standard tests for two of them. While more work is needed to refine IL‑10 detection and to validate the system in larger patient groups, this technology points toward a future where people with asthma and other inflammatory diseases could monitor their immune status quickly and painlessly at the point of care, helping doctors tailor treatments to each person’s unique immune profile.
Citation: Churcher, N.K.M., Rizvi, F.Z., Qureshi, A. et al. Point-of-care multiplexed detection of cytokines using a HexaPie electrode array for asthma endotyping. npj Biosensing 3, 16 (2026). https://doi.org/10.1038/s44328-026-00081-2
Keywords: asthma monitoring, saliva biosensor, cytokine detection, point-of-care diagnostics, electrochemical impedance