Clear Sky Science · en
Surface Transmon Resonance (STR): a handheld nanogap biosensor for real-time, label-free molecular binding kinetics
Why a Tiny Electronic Sensor Matters for Your Health
Modern medicine relies on tests that detect proteins and other molecules in your blood to diagnose disease, guide treatments, and monitor health. Today, many of the most accurate tests use bulky, expensive optical machines that live in centralized laboratories. This article introduces a new kind of palm‑sized electronic sensor that can read out the same kind of molecular binding events in real time, without fluorescent labels or large optical setups. If such sensors can be made cheap and portable, they could move sophisticated diagnostics from specialized labs to clinics, ambulances, and even home devices.
A New Way to “Listen” to Molecules
The researchers present a technology they call Surface Transmon Resonance (STR), an electronic biosensor that borrows ideas from quantum computing hardware. Instead of using light, STR relies on high‑frequency radio waves sent through a tiny circuit that includes a nanoscale gap between two metal lines. When molecules stick to the surfaces inside this gap, they subtly change how the circuit resonates, much like adding weight to a guitar string changes its pitch. A low‑cost handheld instrument called a nano vector network analyzer measures these shifts in resonance phase and frequency, producing curves very similar to those from surface plasmon resonance (SPR), the optical “gold standard” for studying how biomolecules bind.

Beating a Fundamental Roadblock in Electronic Sensing
Electronic biosensors usually struggle in salty, water‑based solutions like blood, because dissolved ions form a screening layer that hides molecular charges from the sensing electrode. This effect, known as Debye screening, has limited many transistor‑based biosensors. STR tackles this problem by operating at hundreds of megahertz, a regime where the ions cannot keep up with the rapidly oscillating electric field. As a result, the screening layer weakens and the field can probe a thin layer of molecules at the surface more directly. The sensor’s design focuses the electric field into a nanometer‑scale gap, whose size is comparable to typical proteins, so even a thin molecular layer takes up a significant fraction of the sensing volume and produces a measurable resonance shift.
Watching Proteins Bind in Real Time
To show that STR can do serious biochemical work, the team studied a classic test pair: bovine serum albumin (BSA), a well‑known protein, and antibodies that recognize it. First they flowed a buffer solution through a microfluidic channel over the sensor to get a baseline, then injected BSA to coat the gold surface inside the gap, and finally introduced anti‑BSA antibodies at different concentrations. The sensor tracked how its resonance frequency shifted over time as antibodies attached and later detached when clean buffer was reintroduced. Because the nanogap is so small, binding events have a large effect compared with a control device that has a 10‑micrometer‑wide gap, confirming that STR responds mainly to surface‑bound molecules rather than bulk liquid changes. By fitting the binding and unbinding curves, the authors extracted association and dissociation rates, and an overall affinity constant that closely matched values measured independently with an SPR instrument.
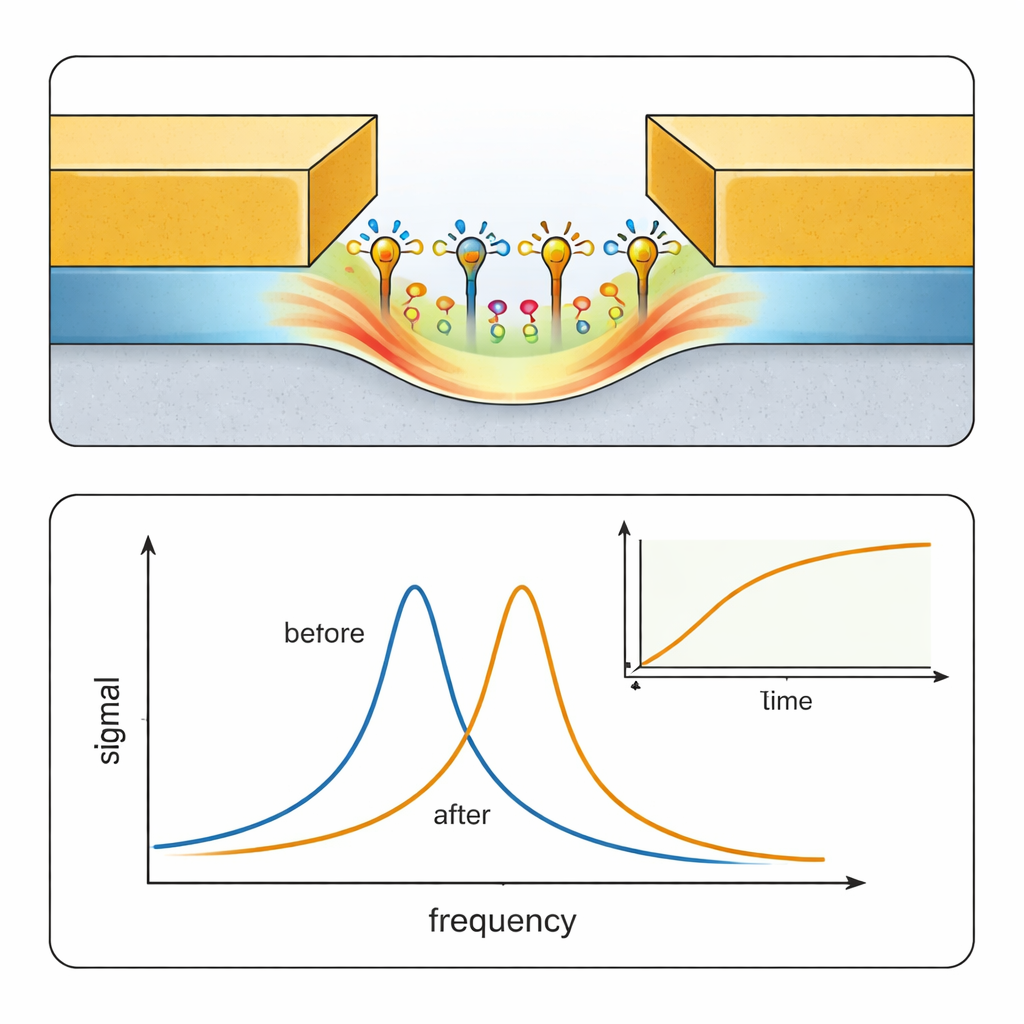
Performance That Rivals Optical Lab Equipment
Beyond simple detection, the authors quantified how sensitive STR is. They showed that tiny shifts in resonance frequency correspond to very small changes in the electrical properties of the solution near the surface, and determined a protein detection limit of about 7 nanomolar for the tested antibody. That performance is comparable to several advanced nanoplasmonic SPR sensors reported in the literature. Importantly, this was achieved using a prototype built for portability and low cost, not for ultimate sensitivity. The main noise source was the handheld analyzer itself, and the authors outline straightforward paths to improvement, such as increasing signal power, boosting the sharpness (Q‑factor) of the resonance, narrowing the gap further, and refining surface chemistry and electronics integration.
From Lab Benchtop to Pocket‑Sized Diagnostics
To make STR practical at scale, the team also developed a wafer‑level fabrication approach that can produce arrays of nanogap sensors using techniques compatible with mainstream microelectronics. They envision future versions in which the sensing structures and radio‑frequency circuits are integrated on the same chip, potentially forming the heart of portable or even wearable diagnostic tools. Because STR delivers the kind of real‑time binding curves and quantitative kinetic data usually restricted to large optical instruments, it could bring laboratory‑grade molecular analysis to many more settings. For non‑specialists, the takeaway is that this work moves us closer to handheld devices capable of tracking how specific biomolecules bind and interact in real time—opening the door to faster, more accessible, and more personalized medical testing.
Citation: Chantigian, B.K., Oh, SH. Surface Transmon Resonance (STR): a handheld nanogap biosensor for real-time, label-free molecular binding kinetics. npj Biosensing 3, 15 (2026). https://doi.org/10.1038/s44328-026-00080-3
Keywords: biosensor, molecular diagnostics, nanotechnology, radio frequency sensing, label-free detection