Clear Sky Science · en
A micro-organ based microfluidic biosensor for continuous monitoring of glucose levels in vivo
Why this matters for people with diabetes
For people living with type 1 diabetes, today’s “artificial pancreas” systems still need constant attention—announcing meals, handling alarms, and worrying about low blood sugar at night. This study explores a radically different kind of glucose sensor that does not rely on a single enzyme or a computer equation, but instead borrows a tiny living piece of the body’s own sugar-control machinery: clusters of pancreatic cells called islets. By letting these mini-organs do what evolution has optimized them to do, the researchers aim to build smarter, safer glucose monitors that could one day run more independently in the background.
A tiny organ as a living sensor
Most current biosensors detect chemicals using purified molecules or single cell lines. They respond to one specific substance and then pass the raw signal to software that tries to interpret it. The authors argue that this approach ignores a powerful built-in “computer”: micro-organs, like pancreatic islets, that naturally sense many signals at once and turn them into a coordinated response. Each islet contains several hormone-producing cell types that constantly talk to each other. Together they keep blood sugar within a narrow, safe range, reacting not only to glucose but also to amino acids, gut and stress hormones, and the body’s recent history of highs and lows. This built-in complexity, they suggest, could be harnessed to produce richer, more reliable information than a simple glucose-oxidase sensor.
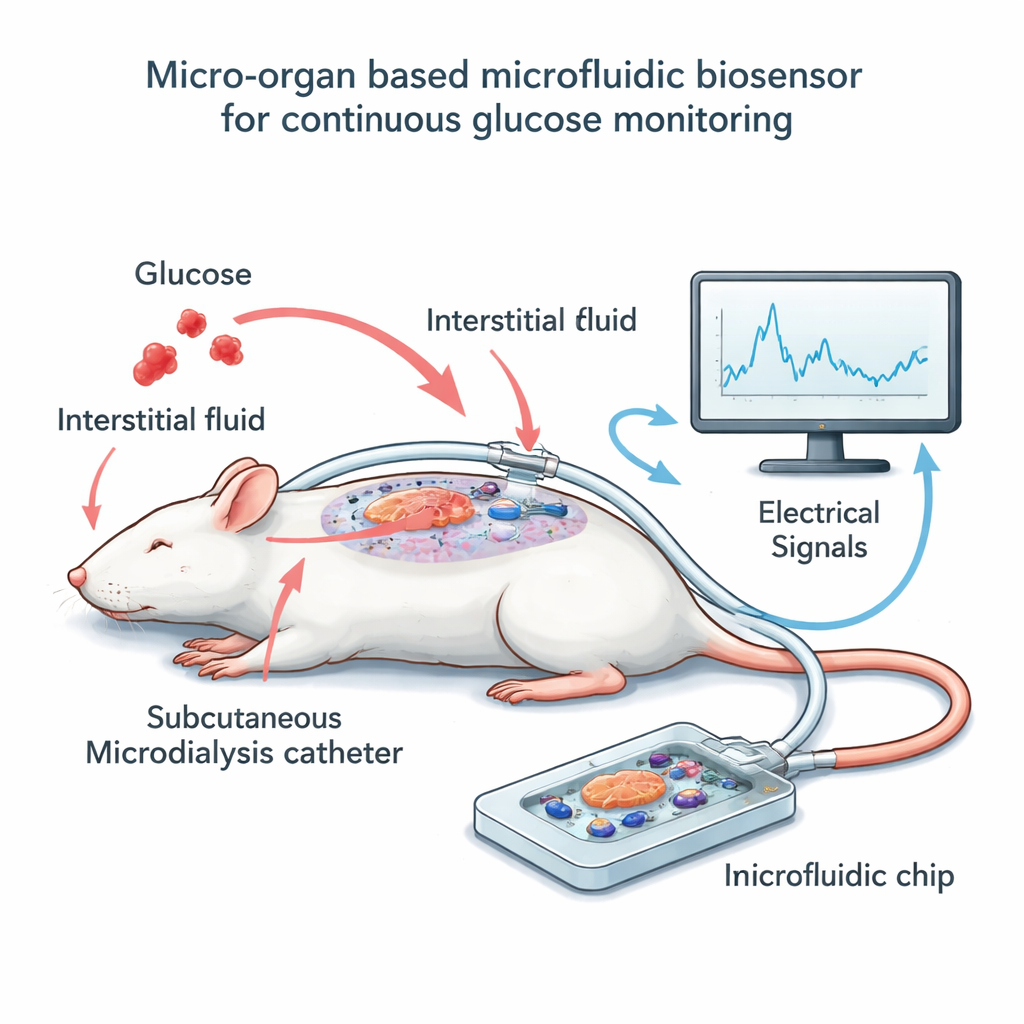
Building the chip-and-catheter system
To test this idea, the team built a small device outside the body that houses a few dozen mouse islets on top of a grid of microscopic electrodes. Instead of bathing these cells directly in blood, they used a technique called microdialysis: a thin flexible catheter sits just under the skin of anesthetized rats and slowly collects the surrounding tissue fluid, which closely tracks blood sugar but with a short delay. A low, constant flow carries this fluid through a narrow channel over the islets on the chip. As glucose levels in the dialysate rise and fall, the islets change their electrical activity, which the electrodes capture as slow, wave-like signals. In parallel, the researchers periodically measured glucose in the rats’ blood and in the dialysate, allowing them to line up the electrical readout with actual sugar levels over time.
How the living sensor responded to changing sugar
First, the scientists tested whether islets on the electrodes could respond to real blood components rather than simple lab solutions. They exposed them to human and rat serum with different glucose levels and saw clear, graded changes in both the frequency and size of their electrical waves. The responses were strong enough to distinguish small steps in sugar concentration across the range relevant for diabetes. They then moved to rat experiments, giving an injection of glucose to raise blood sugar and later injecting insulin to bring it down. After accounting for the time it takes glucose to move from blood to tissue fluid and through the tubing, they found that the frequency of the islets’ slow electrical signals rose and fell almost in lockstep with blood sugar. Statistical analysis across several animals showed a remarkably tight, repeatable relationship between signal frequency and glucose level, while signal amplitude, though informative, was somewhat more variable.
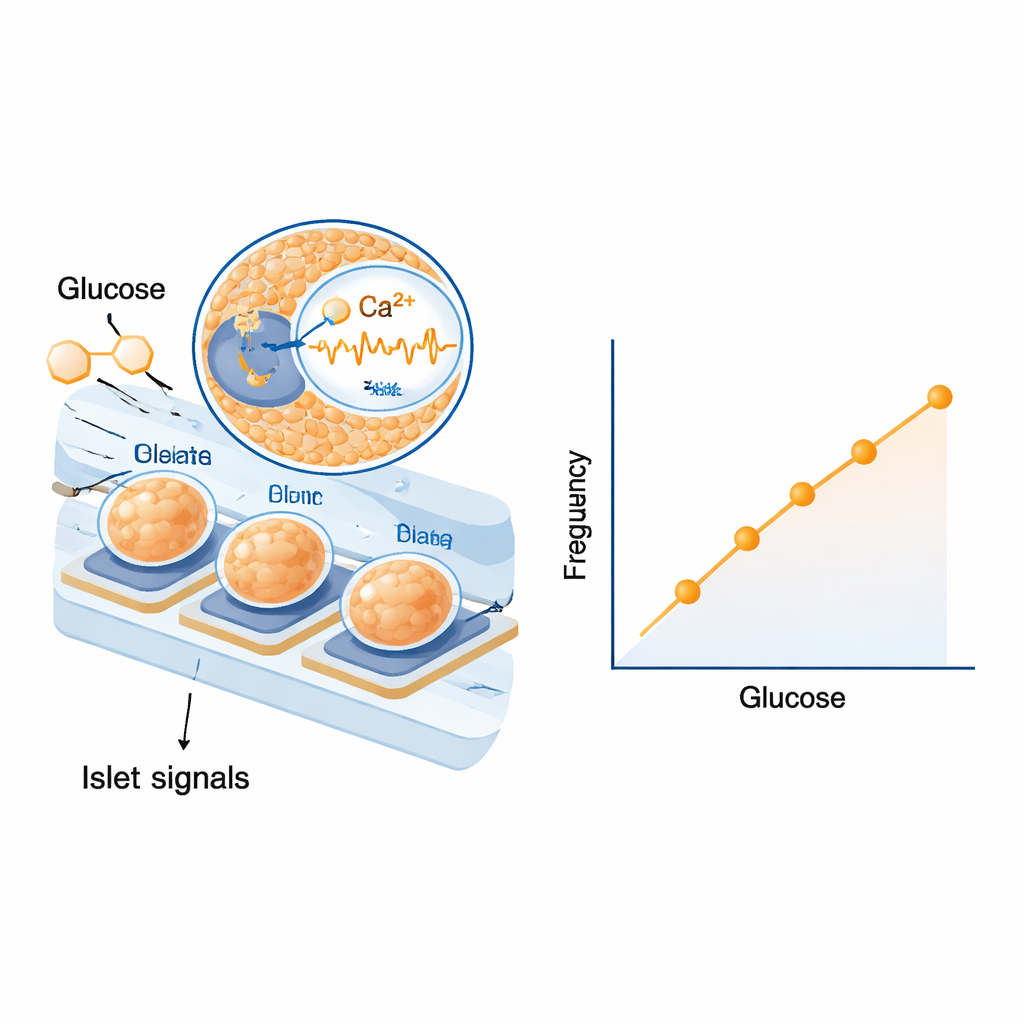
Built-in safety features from biology
An important advantage of using whole islets, rather than a single molecule, is that their behavior already includes safety mechanisms shaped by evolution. Earlier work, and hints from this study, show that islets react more strongly when glucose is falling than when it is rising—a form of “hysteresis” that helps protect against dangerous hypoglycemia. The electrical patterns are also shaped by neighboring cell types that amplify or dampen insulin release depending on the body’s needs. Because the chip simply listens to these integrated signals without adding dyes or genetic modifications, it can record for days without harming the cells. The authors note technical challenges ahead, such as avoiding air bubbles, ensuring long-term microdialysis performance, and deciding what kind of human or stem cell–derived islets should eventually be used. Still, computer simulations using an established diabetes model suggest that such an islet-based sensor could match or even surpass today’s glucose monitors under difficult conditions.
What this could mean for future diabetes care
This work shows that a handful of pancreatic micro-organs, kept alive on a small chip and connected to the body by a thin catheter, can track blood sugar continuously in living animals by their natural electrical chatter. For a non-specialist, the key message is that instead of asking a simple chemical sensor to measure glucose and then forcing a computer to guess what that means for the body, this approach listens directly to the same type of living tissue that normally decides how much insulin to release. If refined and made practical for human use, such living biosensors could become the core of truly autonomous artificial pancreas systems—devices that quietly anticipate the body’s needs and reduce the everyday burden of diabetes management.
Citation: Puginier, E., Pirog, A., de Gannes, F.P. et al. A micro-organ based microfluidic biosensor for continuous monitoring of glucose levels in vivo. npj Biosensing 3, 12 (2026). https://doi.org/10.1038/s44328-025-00077-4
Keywords: continuous glucose monitoring, artificial pancreas, pancreatic islets, microfluidic biosensor, type 1 diabetes