Clear Sky Science · en
Development and preclinical results of a transcatheter aortic valve implant with novel polymeric leaflets
Why a new kind of heart valve matters
Heart valve disease is common in older adults and is increasingly treated without open-heart surgery, using folded replacement valves that doctors thread into the heart through blood vessels. Today’s valves are usually made from treated animal tissue, which can wear out, calcify, and fail over time—especially in younger, more active patients. This study explores a different approach: a transcatheter aortic valve made with advanced synthetic materials that aim to last longer, resist damage, and remain kinder to the blood flowing past them.
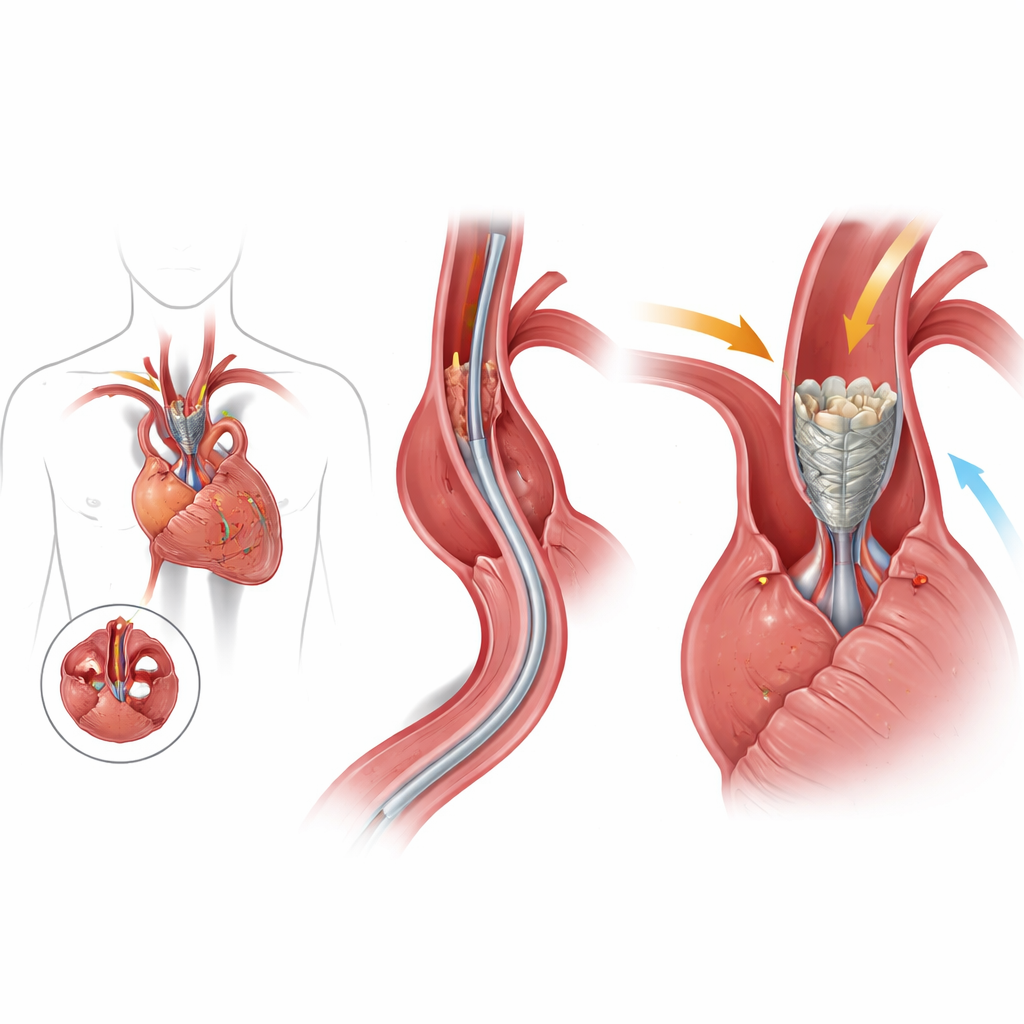
A gentler route to fixing a tight heart valve
The aortic valve sits at the exit of the heart’s main pumping chamber and opens and closes with every heartbeat. When it becomes stiff and narrow, the heart has to work dangerously hard to push blood out. Surgeons have long been able to replace this valve, but surgery requires opening the chest and using a heart-lung machine. In the past two decades, transcatheter aortic valve implantation (TAVI) has changed that by letting doctors deliver a folded valve through an artery and expand it inside the old one. While this has been a breakthrough for older and high‑risk patients, current TAVI valves still rely on animal tissue leaflets that can deteriorate, limiting how confidently they can be offered to younger people.
A valve built from smart plastics and memory metal
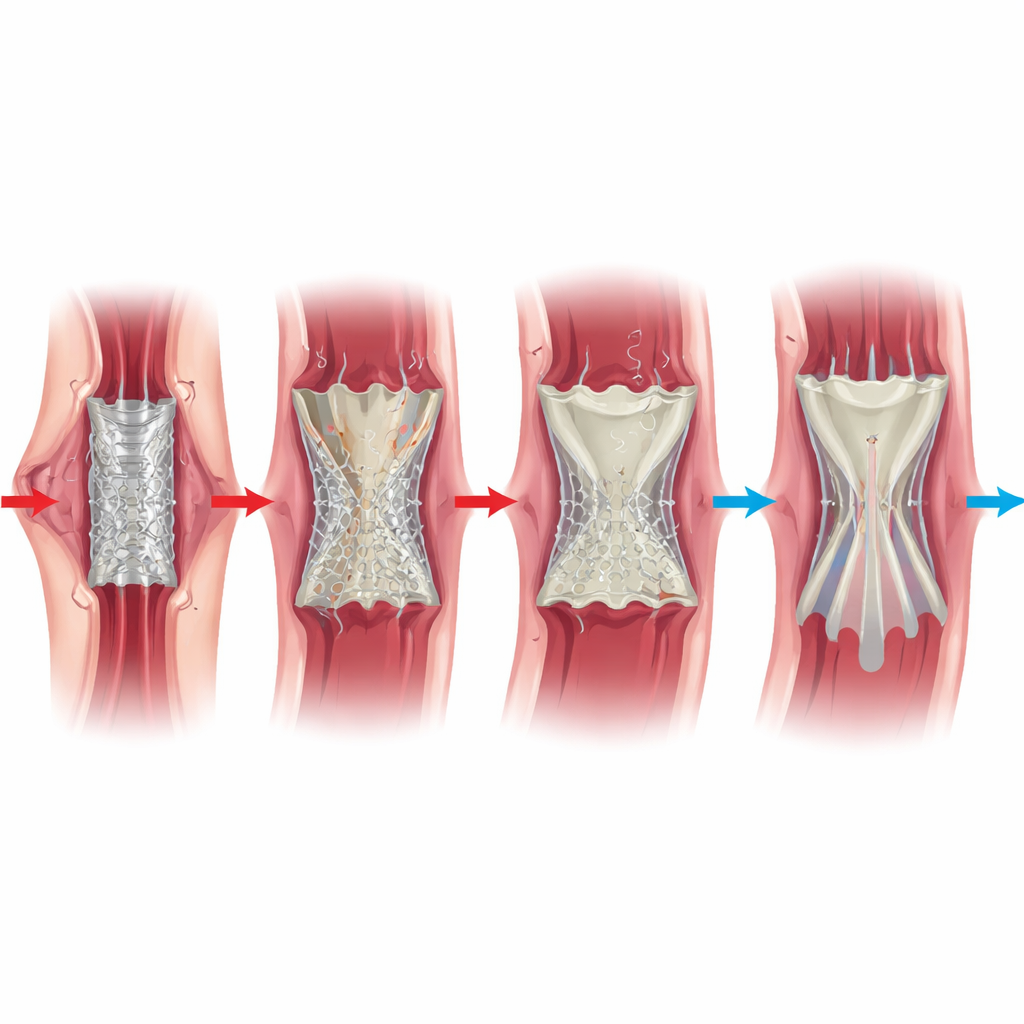
The team designed a new TAVI device that replaces the animal tissue leaflets with thin, flexible "polymeric" ones made from a specialized silicone‑based polyurethane (called LifePolymer) and mounts them on a self‑expanding frame of nitinol, a metal alloy that naturally springs back to a preset shape. The frame has an hourglass profile to keep space for the heart’s own arteries, and its cells are coated in the same polymer to soften contact with blood. Around the base, a porous skirt of electrospun polymer is added to help the valve seal against the native tissue and reduce leaks around the edges. This design aims to pair the strength and fatigue resistance of engineered materials with the smooth blood flow of a healthy natural valve.
Putting the new valve through demanding bench tests
Before heading into animals, the researchers pushed the valve through extensive laboratory testing meant to mimic years of use. They cycled the nitinol frame 200 million times—roughly five years of heartbeats—under stressful conditions and saw no fractures, cracks, or changes in shape. In a pulsating flow system that imitates the beating heart, the valve allowed generous forward flow with pressure differences and backflow well within international performance standards. High‑speed flow imaging showed smooth jets of blood through the center, with very little swirling or stagnation near the leaflets or at the joints between them, areas where clots often begin. Computer‑tracked particles washed out efficiently, suggesting a low tendency to trigger clot formation. Standard safety tests found no evidence that the materials damaged cells, blood, or DNA, or triggered immune or allergic reactions.
Testing the valve in living hearts
To see how the valve behaved in a living circulation, the team implanted it into the aortic position of nine sheep, a commonly used model for heart valves because their hearts beat with similar pressures to humans and their tissues calcify rapidly. Six animals had successful valve placement and were followed for 90 days. Ultrasound scans showed that the valves opened and closed freely, maintained good blood flow, and produced little to no leakage through the center or around the sides. Blood tests stayed within normal ranges, with no signs of red‑blood‑cell damage or organ stress. When the animals were humanely euthanized and their hearts examined, the polymer leaflets were still smooth and flexible, showing no calcification, no tears, and no overgrowth of scar tissue that might have begun to block flow. The porous outer skirt had begun to knit gently with surrounding tissue, helping anchor the device without interfering with leaflet motion.
What this could mean for future patients
Taken together, these early results suggest that a transcatheter valve built from advanced polymers on a self‑expanding memory-metal frame can provide strong, stable support and healthy blood flow while remaining friendly to blood and surrounding tissue—at least over the first few months. If longer‑term studies confirm that the polymer leaflets truly resist wear and calcification better than animal tissue, such devices could last longer and be safer for younger people who might otherwise face multiple valve replacements over a lifetime. The work does not yet prove long‑term benefit in humans, but it lays the groundwork for next‑stage testing and hints at a future where minimally invasive heart valve repair could combine durability with gentle interaction with the body.
Citation: Stanfield, J.R., Johnson, G., Belais, N. et al. Development and preclinical results of a transcatheter aortic valve implant with novel polymeric leaflets. npj Cardiovasc Health 3, 14 (2026). https://doi.org/10.1038/s44325-026-00112-x
Keywords: transcatheter aortic valve, polymeric heart valve, aortic stenosis, biocompatible materials, cardiovascular implants