Clear Sky Science · en
Beta 2 adrenergic receptor agonists as a treatment for metabolic dysfunction-associated steatohepatitis (MASH)
Why this liver study matters
Fatty liver disease tied to obesity and type 2 diabetes is now one of the most common reasons people need liver transplants. Yet there are only a couple of approved drugs, and they help only some patients. This study asks a surprising question: could a familiar asthma medicine, already used safely by millions, also help protect the liver from this silent, progressive damage?
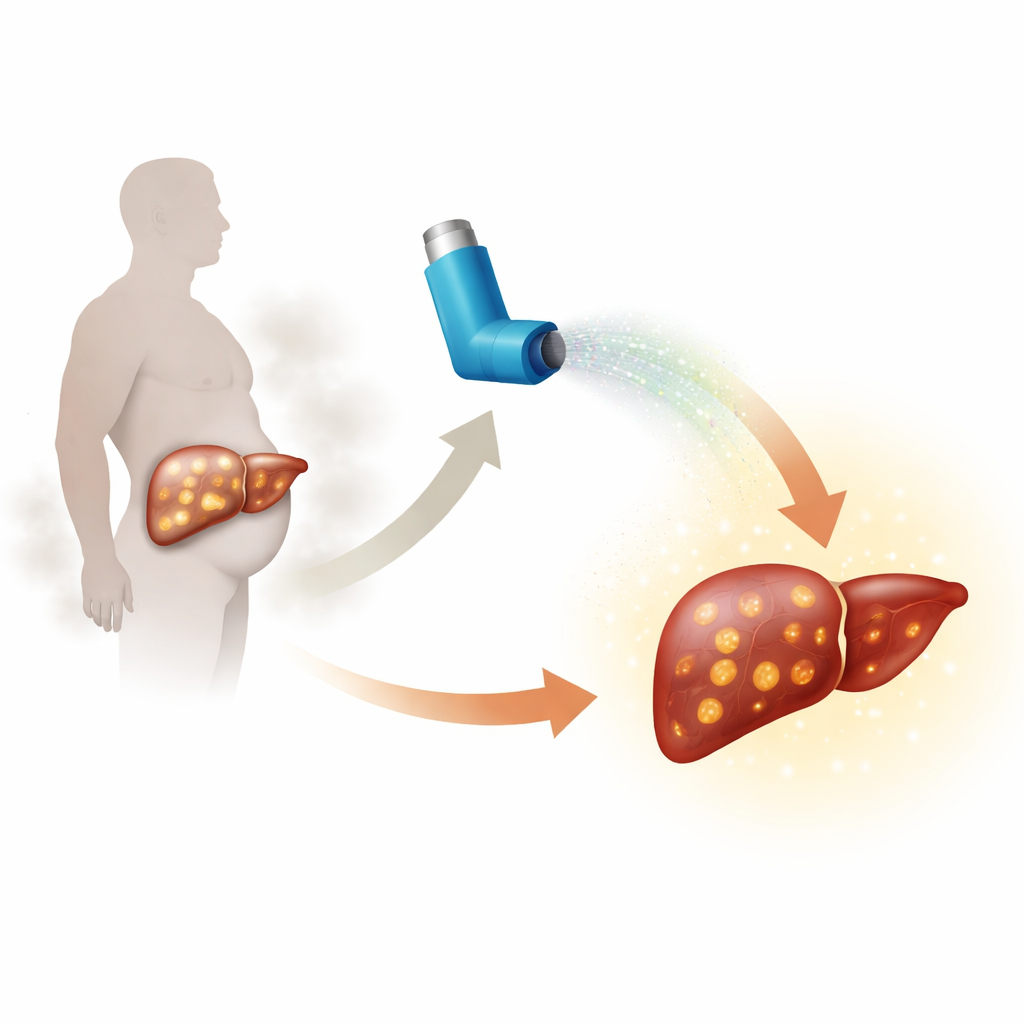
A common illness with high stakes
The condition the researchers focus on is metabolic dysfunction–associated steatohepatitis, or MASH. In MASH, the liver becomes swollen with fat, inflamed, and gradually scarred. People with type 2 diabetes are especially at risk, and many go on to develop cirrhosis, liver cancer, or kidney failure. Current drugs target hormones and inflammation but leave many patients without good options. Because earlier work showed that a drug called formoterol—a long‑acting beta‑2 adrenergic receptor agonist, widely used in inhalers for lung disease—could protect the kidneys in diabetes, the team wondered if it might also rescue an overworked, fatty liver.
Testing an asthma drug in a fatty liver model
The researchers first turned to mice fed a high‑fat diet that reliably causes fatty liver. After several months on this diet, the mice received either formoterol or a placebo injection for four weeks. Under the microscope, livers from untreated animals were packed with fat droplets, while those from the formoterol group showed a striking reduction in fat and lower overall disease scores. Chemical analysis confirmed that major fat types, especially triglycerides and related molecules, were reduced. At the same time, imaging of liver tissue revealed more mitochondria—the tiny power plants inside cells—in treated animals, and protein measurements showed increases in key components of the cell’s energy‑producing machinery.
Looking inside human liver cells
To see whether these effects translated to human liver tissue, the team used HepaRG cells, a human liver‑like cell line. When these cells were soaked in abundant dietary fats, they ballooned with fat droplets. Adding formoterol largely prevented this buildup, even though the cells were still exposed to the same amount of fat. Using an oxygen‑sensing device, the scientists found that formoterol boosted the cells’ energy use, especially the portion directly linked to making ATP, the cell’s energy currency. Treated cells ran their mitochondria near full capacity, suggesting a shift toward burning fuel more efficiently rather than simply storing it as fat.
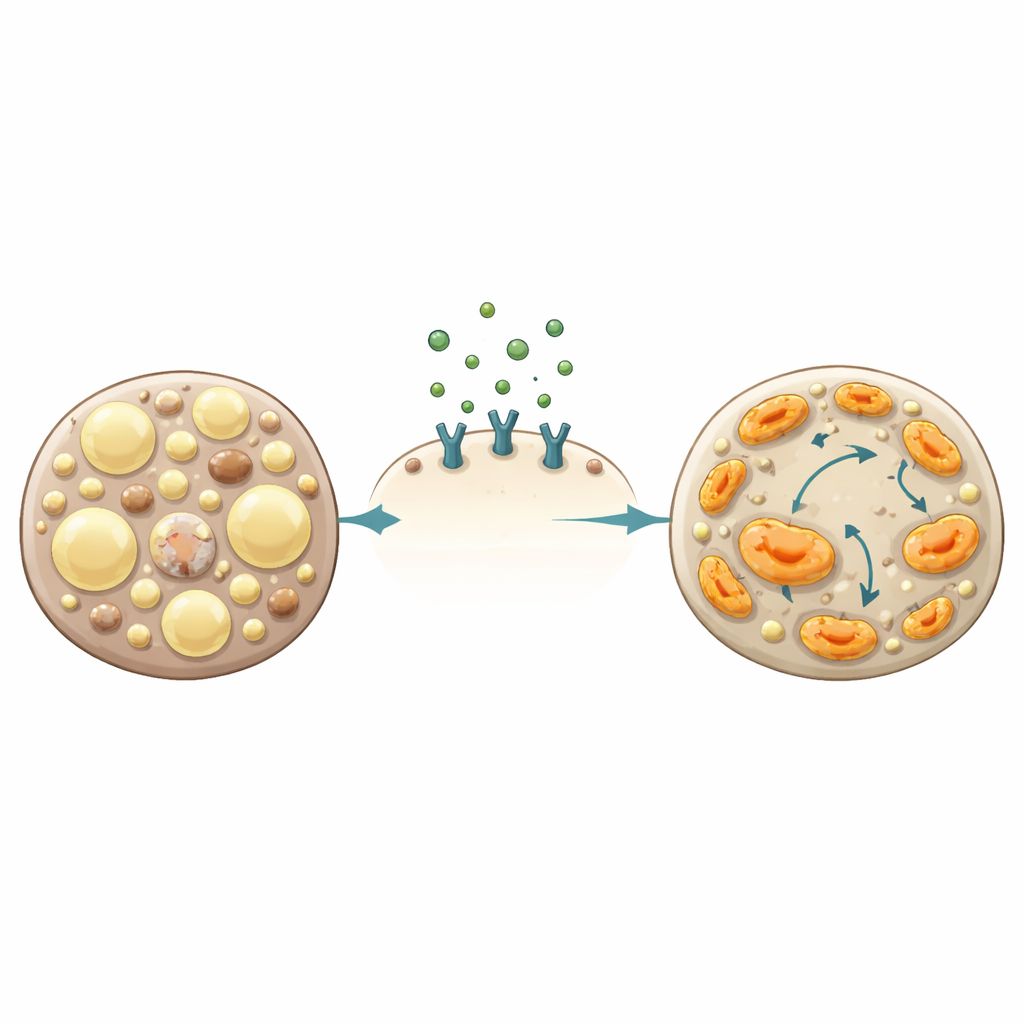
Rewiring cell programs and dampening stress
The team then examined which genes were turned on or off in mouse livers and human liver cells after formoterol treatment. In both systems, the drug enhanced programs related to energy production and amino acid handling, while dialing down those linked to inflammation, scarring, and fat synthesis. One standout change was a strong increase in a urea‑cycle enzyme that is often reduced in fatty liver disease, hinting that overall liver metabolic fitness was restored. Genes involved in building and remodeling the structural “scaffolding” around cells were suppressed in mice, consistent with a potential benefit against future scarring, even though visible fibrosis had not yet appeared in this particular model.
Signals from the clinic
Finally, the researchers analyzed health records from nearly 60,000 adults with MASH in a large multicenter database. They compared people who had been prescribed long‑acting beta‑2 agonists (usually for lung conditions like chronic obstructive pulmonary disease) with similar patients who had never received these drugs. After carefully balancing the groups for age, other illnesses, and liver disease severity, those taking these medicines had lower rates of cirrhosis, fluid buildup in the abdomen, serious infections in that fluid, bleeding from enlarged veins in the esophagus, kidney‑liver failure, and overall death during the follow‑up period.
What this could mean for patients
Taken together, the animal, cell, and human data point in the same direction: drugs like formoterol may help an overburdened fatty liver by building more and better‑working mitochondria, reducing fat accumulation, and calming harmful inflammation and scarring signals. Because these medications are already widely used and relatively inexpensive, they could potentially be repurposed more quickly than brand‑new drugs—if future clinical trials confirm that they are effective and safe at doses appropriate for liver disease. The authors are already running such a trial in people with diabetes, kidney disease, and fatty liver, raising the possibility that a familiar inhaler medicine could one day become part of the toolkit against a major metabolic liver epidemic.
Citation: Winkler, B.S., Stayer, K.M., Rao, A.K. et al. Beta 2 adrenergic receptor agonists as a treatment for metabolic dysfunction-associated steatohepatitis (MASH). npj Metab Health Dis 4, 14 (2026). https://doi.org/10.1038/s44324-026-00108-2
Keywords: fatty liver disease, type 2 diabetes, formoterol, mitochondria, beta-2 agonists