Clear Sky Science · en
Enteric nervous system in exercise physiology: a microbiota-neural interface
Why Your Gut Nerves Matter When You Work Out
Anyone who has sprinted to the finish of a race or pushed through a hard workout knows that the gut often weighs in—sometimes with cramps, nausea, or an urgent dash to the bathroom. This review article argues that a hidden “second brain” in the gut, called the enteric nervous system, helps explain why some people thrive on exercise while others struggle, and how your gut microbes and gut nerves may shape performance, recovery, and long-term health.
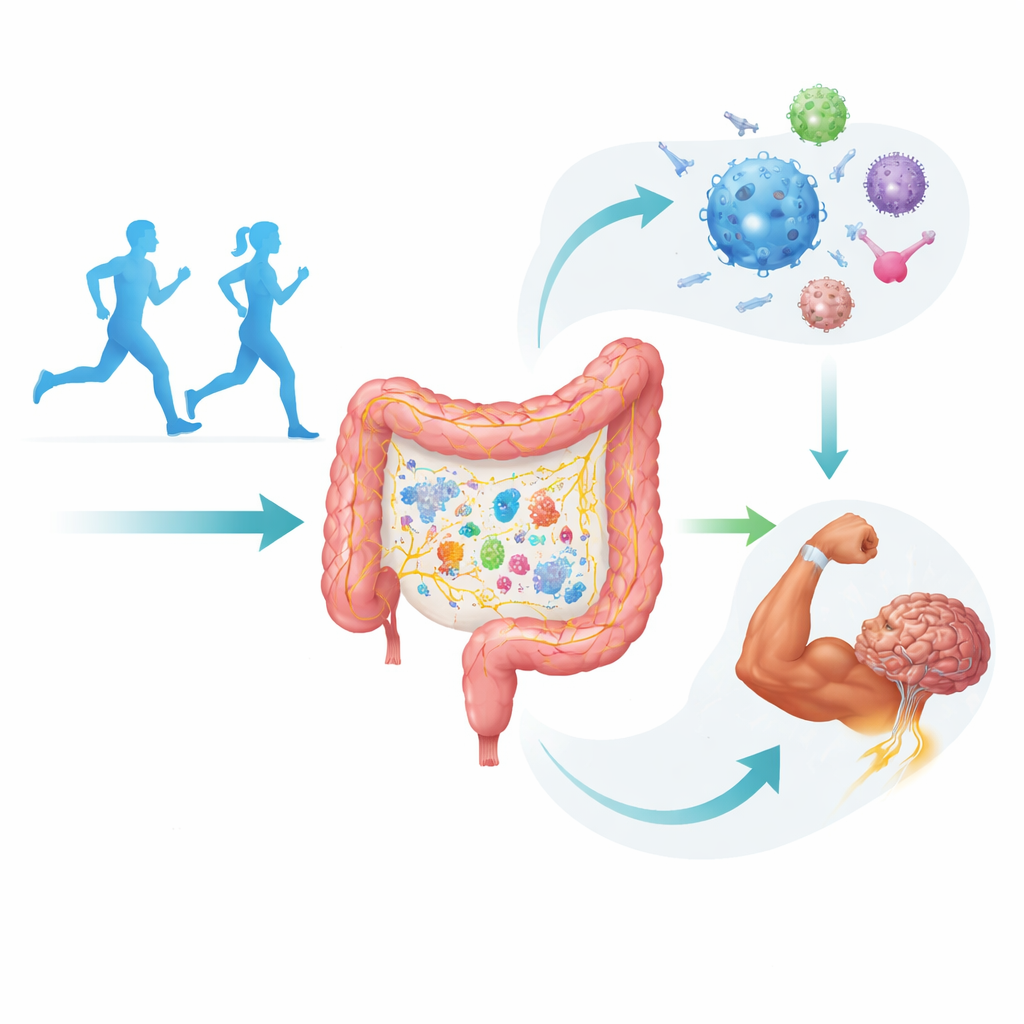
The Missing Link in Exercise Responses
Scientists have long known that regular physical activity improves metabolism, immunity, and brain function, yet people respond very differently to the same training plan. Recent research has highlighted gut microbes and their chemical byproducts as important players in these differences, especially over weeks and months. However, many exercise-related gut changes—such as discomfort and barrier “leakiness”—appear within 30 to 60 minutes, far faster than most microbial chemicals can act. The authors propose that the gut’s own nervous system, which can react in seconds, fills this timing gap and works hand-in-hand with microbes to shape both rapid symptoms and longer-term adaptation.
The Second Brain at Work During Exercise
The enteric nervous system is a dense web of neurons and support cells embedded in the gut wall. During a workout, blood is diverted away from the digestive tract, mechanical strain rises, and stress hormones surge. Enteric sensory cells detect stretching and flow almost instantly and can speed up or slow down gut movements, adjust secretions, and tighten or loosen the intestinal barrier. These rapid, local adjustments change how quickly food moves, how much oxygen reaches the gut lining, and how watery or sticky the environment is—all factors that reshape the living space and resources available to microbes during and after each exercise bout.
Talking Nerves, Immune Cells, and Microbes
The review emphasizes that gut nerves do not act alone. When strenuous exercise temporarily stresses the gut lining, immune cells must respond quickly but not overreact. Enteric neurons release signal molecules that tune the activity of macrophages, mast cells, and innate lymphoid cells, helping balance repair and inflammation. Microbial products such as short-chain fatty acids, tryptophan-derived compounds, and the calming transmitter GABA adjust how excitable these neural and immune circuits are in the first place—effectively setting the “gain” on the system. In turn, nerve-driven changes in mucus release, fluid flow, and barrier tightness reshape microbial niches, favoring some species and constraining others. This ongoing three-way conversation helps determine whether a tough session leads to smooth recovery or lingering gut upset and inflammation.
From Gut Feelings to Muscle and Motivation
Signals arising in the gut do not stop at the intestinal wall. Some microbial molecules enter the bloodstream and slowly influence muscle metabolism, insulin sensitivity, and mitochondria. Others act faster by engaging enteric sensory neurons and vagus nerve pathways that connect to brain regions controlling effort, motivation, and fatigue. In animal studies, certain gut microbes boost exercise willingness by triggering reward circuits in the brain via cannabinoid-like signals. The authors suggest that rapid nerve-based signaling, layered on top of slower circulating chemicals, could explain why two people with similar fitness and similar microbiota can experience very different levels of exertion, pacing, and performance during the same workout.
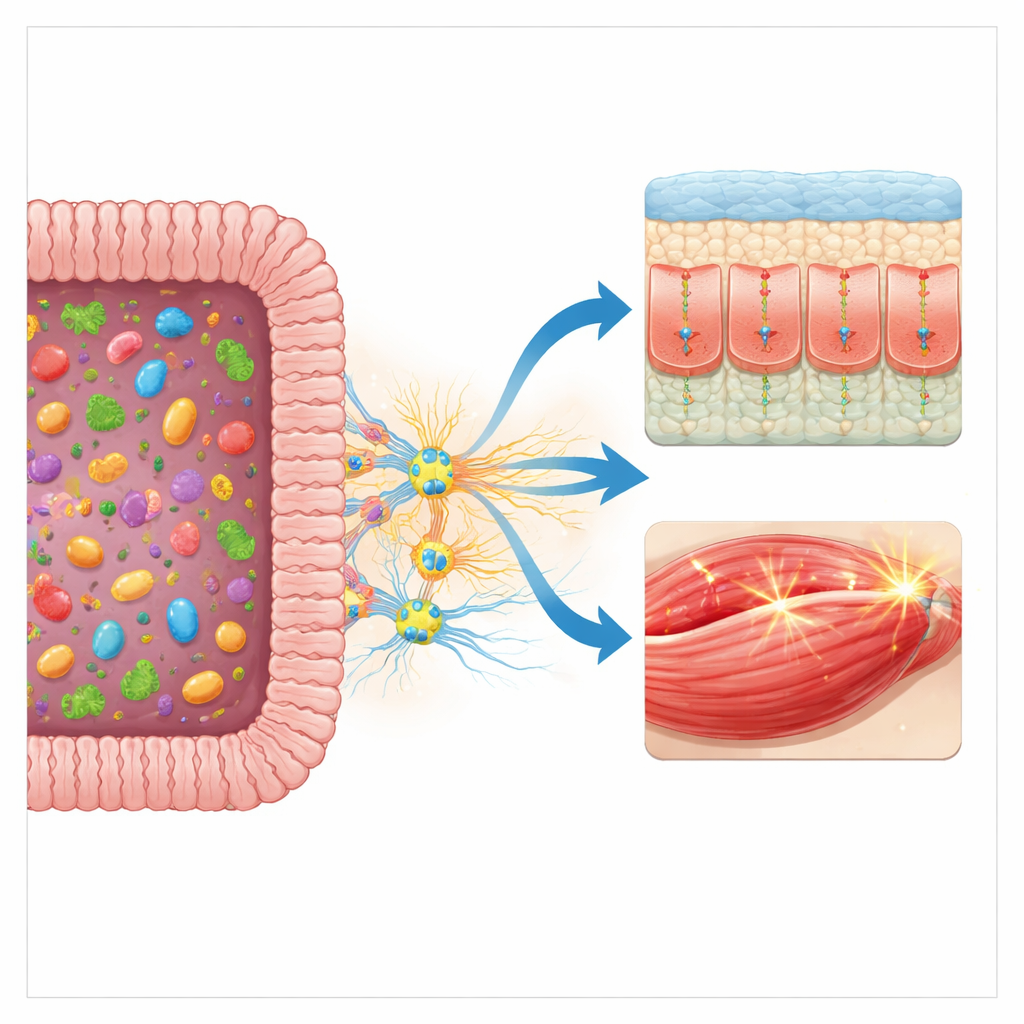
Different Gut-Nerve “Types” and Future Possibilities
To make sense of this variability, the authors introduce the idea of “neuro-enteric phenotypes”—shifting patterns in how an individual’s gut nerves weigh stress, microbial cues, and immune signals. Some states, dominated by stress-related drive, may lead to fast transit, fragile barriers, and more gut symptoms under heavy training. Others, supported by soothing pathways involving molecules like vasoactive intestinal peptide and protective microbial products, may promote steadier motility, stronger barriers, and better tolerance. These states are not fixed: early-life experiences, diet, stress, aging, and training history can all mold the gut nervous system. Although direct measurements in humans are challenging, the authors outline approaches that combine gut permeability tests, stool chemistry, heart rate variability, and symptom tracking to indirectly probe these patterns during exercise.
What This Means for Everyday Athletes
Overall, the article concludes that the gut’s nervous system is a central hub linking exercise, microbes, immunity, and whole-body performance across timescales from minutes to months. Rather than viewing the microbiome or muscles in isolation, the authors argue for an integrated “gut-nerve-microbe-muscle” framework. In the long run, decoding individual neuro-enteric phenotypes could support more personalized training plans and nutrition strategies—helping people reduce gut distress, enhance recovery, and gain more health benefits from the same amount of exercise.
Citation: Chen, HL., Huang, JT., Guo, JJ. et al. Enteric nervous system in exercise physiology: a microbiota-neural interface. npj Metab Health Dis 4, 12 (2026). https://doi.org/10.1038/s44324-026-00106-4
Keywords: enteric nervous system, exercise physiology, gut microbiome, gut-brain axis, intestinal barrier