Clear Sky Science · en
Adipose tissue–derived MFG-E8 promotes hepatic inflammation and fibrosis through macrophage activation in a mouse MASH model
Why belly fat can hurt your liver
Many people think of extra belly fat as just a matter of appearance, but it can quietly damage the liver. This study explores how fat around the organs in the abdomen helps drive a serious liver condition called metabolic dysfunction–associated steatohepatitis (MASH). The researchers identify a protein released from fat tissue that travels to the liver and turns up inflammation and scarring, helping explain why people with large amounts of visceral fat are at higher risk of liver failure and liver cancer, even when their liver fat level looks similar to others.
A hidden messenger from fat to liver
Doctors have long observed that people with a lot of deep belly fat are more likely to develop inflamed, scarred livers than people with the same amount of fat inside the liver but less visceral fat. That pattern suggested that fat tissue must be sending chemical messages to the liver, but the key messengers were unknown. In this work, scientists used mice fed a high-fat diet and a rapid, diabetes-like MASH model to scan for proteins that are strongly produced by visceral fat and found in higher levels in the blood during disease. One protein stood out: MFG-E8, a milk fat–related protein known for helping immune cells recognize and clear dying cells.
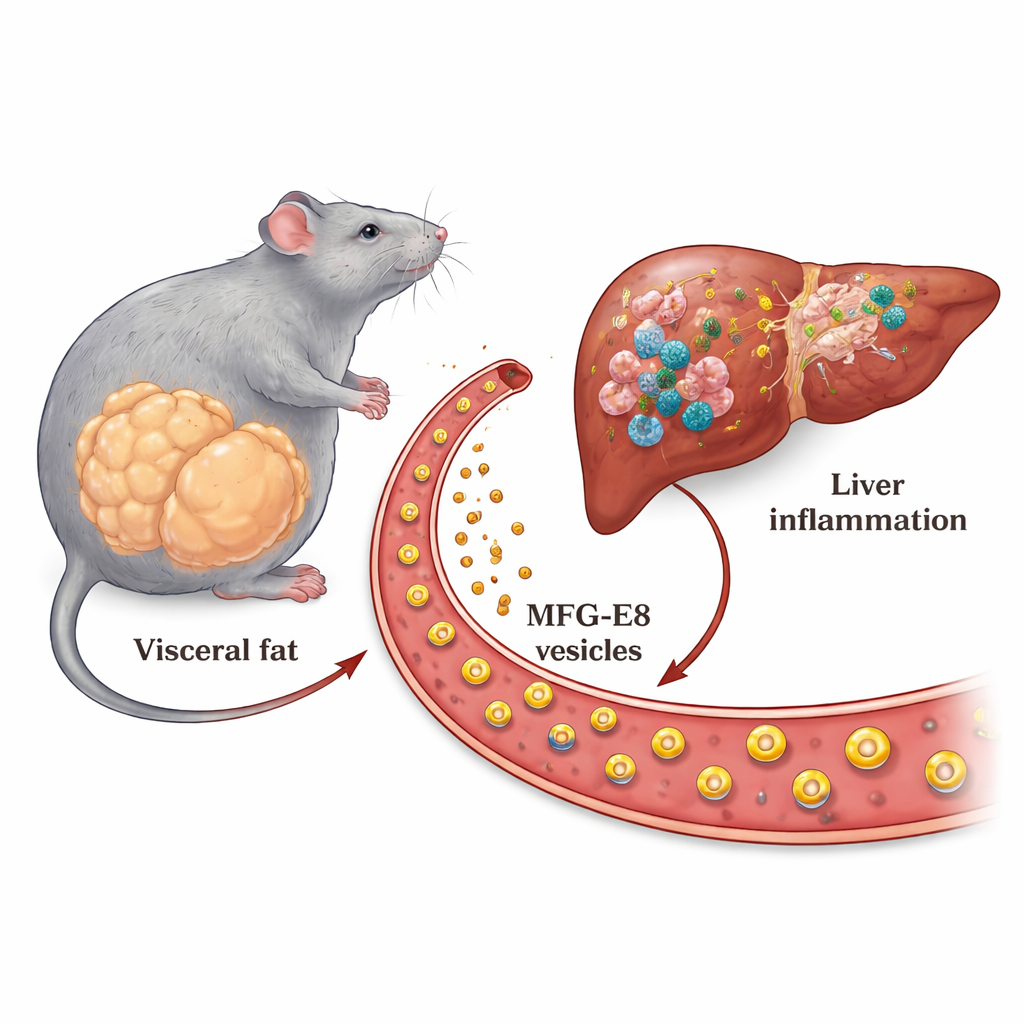
Fat cells crank up MFG-E8 in obesity
The team first asked where MFG-E8 is coming from in diseased animals. They showed that mice on a high-fat diet, and especially mice in the accelerated MASH model, had much higher levels of the Mfge8 gene in their belly fat pads, but not in the liver itself. As fat cells enlarged, they increasingly produced this protein. By separating fat tissue into fat cells and support cells, and by using bone marrow transplant experiments, the researchers demonstrated that the main source of the extra MFG-E8 was the fat cells rather than immune cells. They also confirmed that more MFG-E8 was circulating in the blood, attached to tiny particles called extracellular vesicles, which can act as delivery vehicles to distant organs.
Blocking the signal eases liver damage
To test whether this protein actually worsens liver disease, the scientists used mice that completely lack the Mfge8 gene. When these knockout animals were put through the same MASH-inducing regimen, they developed similar amounts of fat in the liver, but far less inflammation, fewer injured and ballooned liver cells, and much less scar tissue. Immune cell markers and fibrosis-related genes in the liver were sharply reduced, and the animals’ liver enzyme levels in the blood were lower, indicating less damage. Under the microscope, there were far fewer "crown-like" clusters of immune cells encircling dead liver cells, and the overall fibrotic area was smaller. These mice also survived longer than normal mice in the same model, suggesting that removing MFG-E8 helps protect against life‑threatening liver complications.
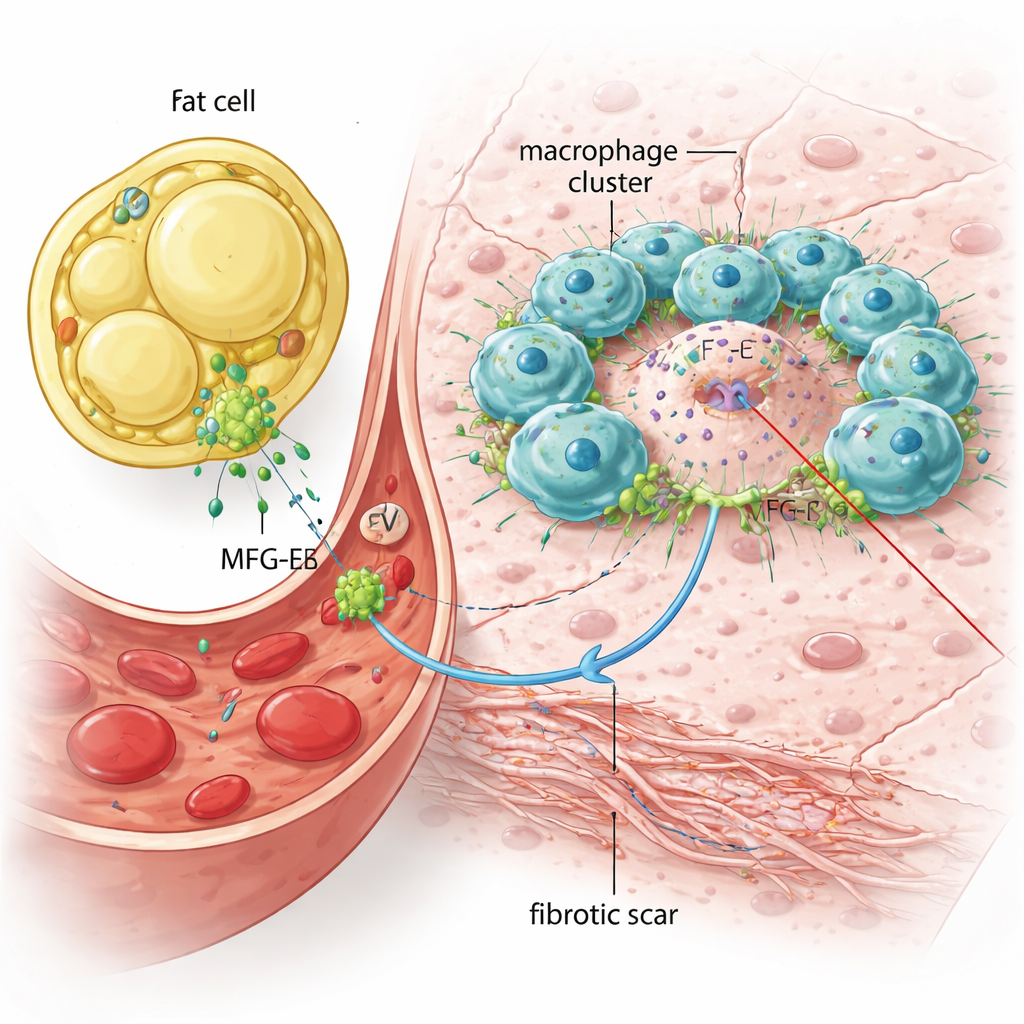
Restoring the signal brings back inflammation
The group then asked whether adding MFG-E8 back could restore disease features. They purified vesicles from cultured immune cells that either did or did not contain MFG-E8 and injected them into Mfge8‑deficient mice in the early MASH phase. Vesicles carrying MFG-E8 selectively boosted inflammatory and scarring genes in the liver and increased the number of immune cell clusters around dying liver cells, while vesicles lacking the protein did not. In a separate dish experiment, they grew mouse macrophages together with dying liver cells. The dead liver cells triggered an inflammatory response, and adding purified MFG-E8 made this response stronger—but only when the immune cells could physically touch the dying cells. When the two cell types were separated by a membrane that blocked direct contact, MFG-E8’s boost largely disappeared, suggesting the protein acts as a physical bridge that tightens the interaction between macrophages and damaged liver cells.
What this means for people with fatty liver
Putting these findings together, the authors propose that, in obesity, oversized fat cells release large amounts of MFG-E8 on vesicles into the bloodstream. This protein then accumulates in the liver, where it helps immune cells cluster around dying liver cells and amplifies inflammatory and scarring signals. Over time, this process can push a relatively silent fatty liver into dangerous MASH with fibrosis and higher risk of cirrhosis and liver cancer. While MFG-E8 can play helpful roles in tissue cleanup and repair in other settings, this study suggests that, in the context of obesity, it becomes part of a harmful fat–liver communication loop. Understanding and eventually interrupting this loop could lead to new ways to protect the liver in people with excess visceral fat.
Citation: Kuroda, M., Nomura, K., Wada, A. et al. Adipose tissue–derived MFG-E8 promotes hepatic inflammation and fibrosis through macrophage activation in a mouse MASH model. npj Metab Health Dis 4, 8 (2026). https://doi.org/10.1038/s44324-026-00099-0
Keywords: fatty liver disease, visceral fat, liver fibrosis, inflammation, MFG-E8